
(a)
Interpretation:
When the given compound is heated, ethene gas is evolved and a product with the formula
Concept introduction:
The Diels-Alder reaction is reversible at high temperature, and this process is called a retro Diels-Alder reaction. The mechanism for this reaction is as the reverse of a Diels-Alder mechanism i.e. the entire six-membered ring is break-down into the corresponding diene and the dienophile. The aromatic proton given signal range from
Answer to Problem 24.82P
The structure of
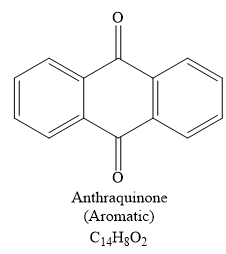
Explanation of Solution
The given compound is
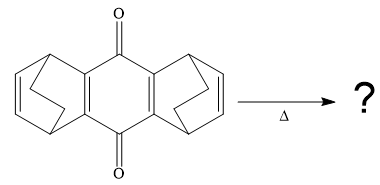
So when this compound is heated, ethene gas is evolved and a product with formula
The signal in
The product of the retro Diels-Alder reaction is also aromatic and matched with the given spectra and also ethene gas is evolved. so the above product is the correct one.
When the given compound is heated, ethene gas is evolved and a product with the formula
(b)
Interpretation:
When the given compound is heated, ethene gas is evolved and a product with the formula
Concept introduction:
The Diels-Alder reaction is reversible at high temperature and this process is called a retro Diels-Alder reaction. The mechanism for this reaction is as the reverse of a Diels-Alder mechanism i.e. the entire six-membered ring is broken down into the corresponding diene and the dienophile. The aromatic proton given signal range from
Answer to Problem 24.82P
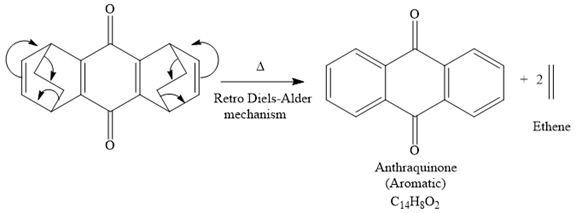
The mechanism for the given reaction is
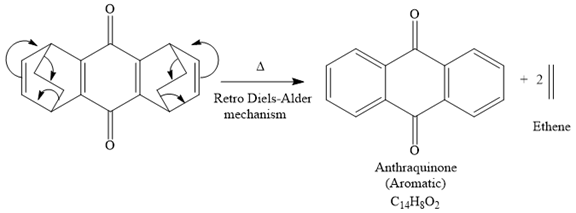
Explanation of Solution
The given compound is

So when this compound is heated, ethene gas is evolved and product with formula
The signal in
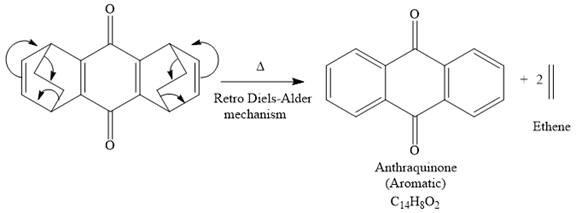
The product of the retro Diels-Alder reaction is also aromatic and matched with the given spectra and also ethene gas is evolved. So the above product and the mechanism (retro Diels-Alder mechanism) is the correct one.
When the given compound is heated, ethene gas is evolved and a product with the formula
(c)
Interpretation:
When the given compound is heated, ethene gas is evolved and a product with the formula
Concept introduction:
The Diels-Alder reaction is reversible at high temperature and this process is called a retro Diels-Alder reaction. The mechanism for this reaction is as the reverse of a Diels-Alder mechanism i.e. the entire six-membered ring is break-down into the corresponding diene and the dienophile. The aromatic proton given signal range from
Answer to Problem 24.82P
The main driving force that favours the product in the given reaction is the formation of a more stable aromatic compound.
Explanation of Solution
The given reaction is
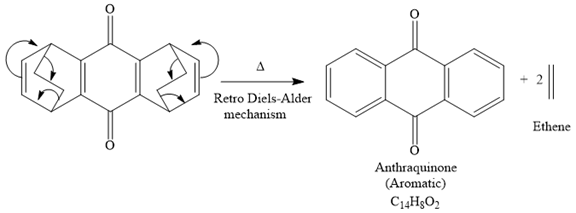
It is noticed that the starting material in the given reaction is overall non-aromatic and also possesses strain due to middle ethylene groups on both sides, but the product is aromatic and strain-free one. Since aromatic compounds are more stable than the non-aromatic compounds this is a key state and the main driving force for the given reaction.
When the given compound is heated, ethene gas is evolved and a product with the formula
Want to see more full solutions like this?
Chapter 24 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 2. Predict the product(s) that forms and explain why it forms. Assume that any necessary catalytic acid is present. .OH HO H₂N OHarrow_forwardconsider the rate of the reaction below to be r. Whats the rate after each reaction? Br + NaCN CN + NaBr a. Double the concentration of alkyl bromide b. Halve the concentration of the electrophile & triple concentration of cyanide c. Halve the concentration of alkyl chloridearrow_forwardPredict the organic reactant that is involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forward
- What are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forwardWhat is the organic molecule X of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forwardWhat are is the organic molecule X and product Y of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forward
- At 300 K, in the decomposition reaction of a reactant R into products, several measurements of the concentration of R over time have been made (see table). Without using graphs, calculate the order of the reaction. t/s [R]/(mol L-1) 0 0,5 171 0,16 720 0,05 1400 0,027arrow_forwardPredict the organic products that form in the reaction below, and draw the skeletal ("line") structures of the missing organic products. Please include all steps & drawings & explanations.arrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forward
- What are the products of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forwardWhat would happen if you added the HCI to the Grignard reagent before adding benzophenone? Draw a reaction mechanism to support your answer.arrow_forwardAt 300 K, in the decomposition reaction of a reactant R into products, several measurements of the concentration of R over time have been made (see table). Calculate the order of the reaction. t/s [R]/ (mol L-1) 0 0,5 171 0,16 720 0,05 1400 0,027arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





