
Concept explainers
(a)
Interpretation:
The products expected when D-ribose is reacted with dilute ![]() is to be stated.
is to be stated.
Concept introduction:
The
Answer to Problem 24.35AP
The product obtained when D-ribose is reacted with dilute ![]() is shown below.
is shown below.
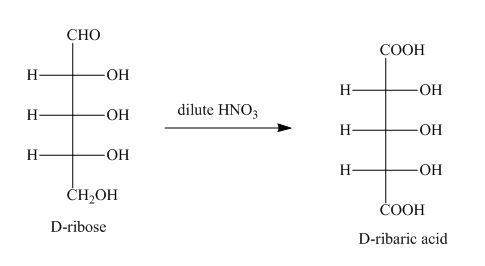
Explanation of Solution
The product obtained when D-ribose is reacted with dilute ![]() is shown below.
is shown below.

Figure 1
The oxidation of D-ribose into D-ribaric acid occurs in the presence of dilute nitric acid.
The product obtained when D-ribose is reacted with dilute ![]() is shown in Figure 1.
is shown in Figure 1.
(b)
Interpretation:
The products expected when D-ribose is reacted with ![]() is to be stated.
is to be stated.
Concept introduction:
Kiliani-Fischer process is is the reaction pathway by which an aldose is extended by one carbon unit. The first step of this reaction is the attack of the cyanide group on the carbonyl carbon of the aldehyde group resulting in the formation of the cyanohydrins. The cyanohydrins thus formed is reduced to imine with catalytic hydrogenation. The imine thus formed can easily be hydrolyzed by into aldose and ammonium ion.
Answer to Problem 24.35AP
The products obtained when D-ribose is reacted with ![]() are shown below.
are shown below.
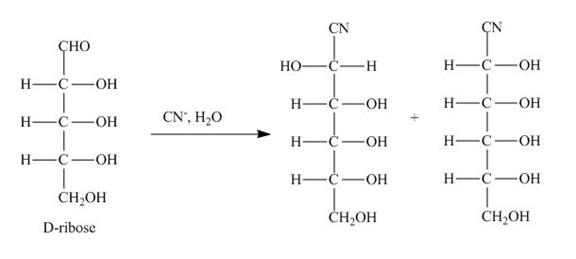
Explanation of Solution
The products obtained when D-ribose is reacted with ![]() are shown below.
are shown below.

Figure 2
The D-ribose is converted into cyanohydrins by the nucleophilic attack of the cyanide group on the carbonyl carbon of the aldehyde group.
The products obtained when D-ribose is reacted with ![]() are shown in Figure 2.
are shown in Figure 2.
(c)
Interpretation:
The products expected when the product of part (b) is reacted with ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction:
Kiliani-Fischer process is is the reaction pathway by which an aldose is extended by one carbon unit. The first step of this reaction is the attack of the cyanide group on the carbonyl carbon of the aldehyde group resulting in the formation of the cyanohydrins. The cyanohydrins thus formed is reduced to imine with catalytic hydrogenation. The imine thus formed can easily be hydrolyzed by into aldose and ammonium ion.
Answer to Problem 24.35AP
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown below.
are shown below.
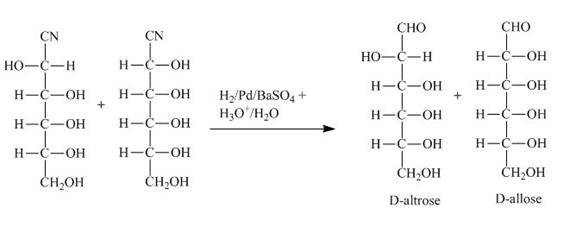
Explanation of Solution
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown below.
are shown below.

Figure 3
The product of part (b) is the cyanohydrin of D-ribose which is then converted into the extended aldose, altrose and allose. The catalytic hydrogenation of cyanohydrin into imine is done by ![]() . The imine then formed is hydrolyzed into the altrose and allose by
. The imine then formed is hydrolyzed into the altrose and allose by ![]() .
.
The products obtained when the product of part (b) is reacted with ![]() and
and ![]() are shown in Figure 3.
are shown in Figure 3.
(d)
Interpretation:
The products expected when D-ribose is reacted with ![]() is to be stated.
is to be stated.
Concept introduction:
A monosaccharide is converted into cyclic acetals on reaction with alcohols in the presence of acidic conditions. The hydroxide group right to the oxygen atom in the pyranose ring structure is methylated and result in the formation of acetal.
Answer to Problem 24.35AP
The product obtained when D-ribose is reacted with ![]() is shown below.
is shown below.
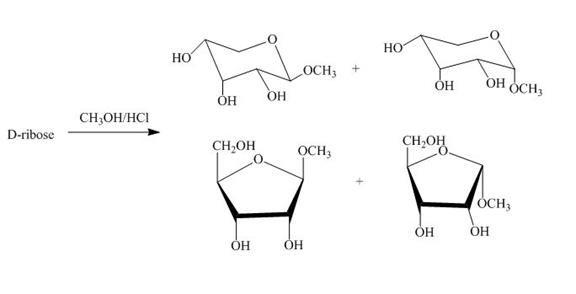
Explanation of Solution
The product obtained when D-ribose is reacted with ![]() is shown below.
is shown below.
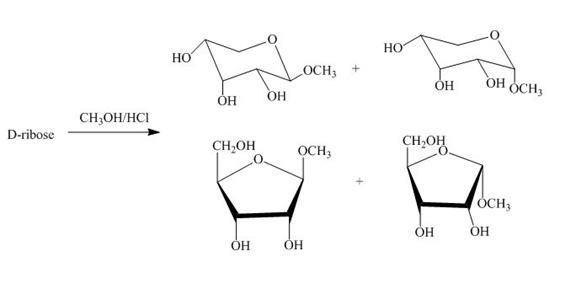
Figure 4
The D-ribose on reaction with methanol and hydrochloric acid is converted into the acetal. The acetal formed is found in both forms alpha and beta regardless of the configuration of D-ribose.
The product obtained when D-ribose is reacted with ![]() is shown in Figure 4.
is shown in Figure 4.
(e)
Interpretation:
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is to be stated.
is to be stated.
Concept introduction:
The methylation of the hydroxyl group of sugars is an important reaction. The methylation of hydroxyl groups is done with the help of methylating agent dimethyl sulfate in the presence of strong base sodium hydroxide.
Answer to Problem 24.35AP
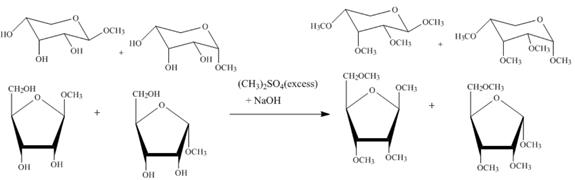
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown below.
is shown below.
Explanation of Solution
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown below.
is shown below.

Figure 5
The four products of part (d) are alkylated in the strong base sodium hydroxide. The sodium hydroxide takes up the acidic proton of alcohol groups and converts them to alkoxide ion form. This alkoxide ion then attacks on the dimethyl sulfate (also a methylating agent) and take up the methyl group simultaneously eliminating the methyl sulfate group.
The product obtained when the product of part (d) is reacted with ![]() (excess) and
(excess) and ![]() is shown in Figure 5.
is shown in Figure 5.
Want to see more full solutions like this?
Chapter 24 Solutions
EBK ORGANIC CHEMISTRY
- An expression for the root mean square velocity, vrms, of a gas was derived. Using Maxwell’s velocity distribution, one can also calculate the mean velocity and the most probable velocity (mp) of a collection of molecules. The equations used for these two quantities are vmean=(8RT/πM)1/2 and vmp=(2RT/M)1/2 These values have a fixed relationship to each other.(a) Arrange these three quantities in order of increasing magnitude.(b) Show that the relative magnitudes are independent of the molar mass of the gas.(c) Use the smallest velocity as a reference for establishing the order of magnitude and determine the relationship between the larger and smaller values.arrow_forwardThe reaction of solid dimethylhydrazine, (CH3)2N2H2, and liquefied dinitrogen tetroxide, N2O4, has been investigated for use as rocket fuel. The reaction produces the gases carbon dioxide (CO2), nitrogen (N2), and water vapor (H2O), which are ejected in the exhaust gases. In a controlled experiment, solid dimethylhydrazine was reacted with excess dinitrogen tetroxide, and the gases were collected in a closed balloon until a pressure of 2.50 atm and a temperature of 400.0 K were reached.(a) What are the partial pressures of CO2, N2, and H2O?(b) When the CO2 is removed by chemical reaction, what are the partial pressures of the remaining gases?arrow_forwardOne liter of chlorine gas at 1 atm and 298 K reacts completely with 1.00 L of nitrogen gas and 2.00 L of oxygen gas at the same temperature and pressure. A single gaseous product is formed, which fills a 2.00 L flask at 1.00 atm and 298 K. Use this information to determine the following characteristics of the product:(a) its empirical formula;(b) its molecular formula;(c) the most favorable Lewis formula based on formal charge arguments (the central atom is N);(d) the shape of the molecule.arrow_forward
- How does the square root mean square velocity of gas molecules vary with temperature? Illustrate this relationship by plotting the square root mean square velocity of N2 molecules as a function of temperature from T=100 K to T=300 K.arrow_forwardDraw product B, indicating what type of reaction occurs. F3C CF3 NH2 Me O .N. + B OMearrow_forwardBenzimidazole E. State its formula. sState the differences in the formula with other benzimidazoles.arrow_forward
- Draw product A, indicating what type of reaction occurs. F3C CN CF3 K2CO3, DMSO, H₂O2 Aarrow_forward19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forwardpls helparrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


