
Concept explainers
Name the following compounds:
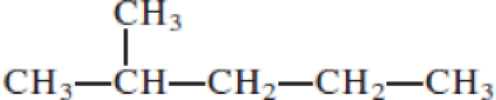
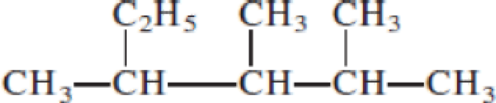
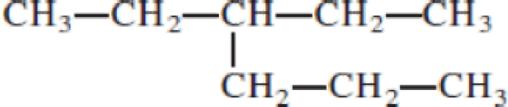
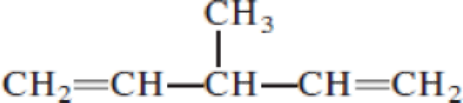
- (a) CH3─C≡C─CH2─CH3
a)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the substituent should get least possible number.
- Write the name of the compound; the parent name written as last part of the name. The name of the substituents is written as prefix and a hypen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Explanation of Solution
Given structure:
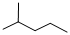
Predict the longest continuous chain of carbon atoms:
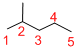
The longest continuous carbon chain of carbon atoms contains five carbons and the parent name is PENTANE. The Suffix ‘ane’ represents the structure contains only single bonds.
Predict substituents and its location:
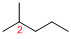
At each Carbon of 2 one methyl group is present. Hence, the structure containing one substituents, then prefix ‘2-methyl’ is added before the parent name. Hyphen separates the number that the substituent attached with carbon.
IUPAC name: 2-methylpentane.
b)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the substituent should get least possible number.
- Write the name of the compound; the parent name written as last part of the name. The name of the substituents is written as prefix and a hypen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Explanation of Solution
Given structure:
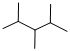
Predict the longest continuous chain of carbon atoms:
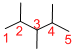
The longest continuous carbon chain of carbon atoms contains five carbons and the parent name is PENTANE. The Suffix ‘ane’ represents the structure contains only single bonds.
Predict substituents and its location:
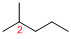
At each Carbon of 2, 3, and 4 one methyl group is present. Hence, the structure containing three identical substituents, then prefix ‘2,3,4-trimethyl’ is added before the parent name. Hyphen separates the number that the substituent attached with carbon.
IUPAC name: 2,3,4-trimethylpentane.
c)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the substituent should get least possible number.
- Write the name of the compound; the parent name written as last part of the name. The name of the substituents is written as prefix and a hyphen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Explanation of Solution
Given structure:
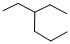
Predict the longest continuous chain of carbon atoms:
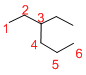
The longest continuous carbon chain of carbon atoms contains six carbons and the parent name is HEXANE. The Suffix ‘ane’ represents the structure contains only single bonds.
Predict substituents and its location:

At each Carbon of 3 one ethyl group is present. Hence, the structure containing one substituent, then prefix ‘3-ethyl’ is added before the parent name. Hyphen separates the number that the substituent attached with carbon.
IUPAC name: 3-ethylhexane.
d)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the substituent should get least possible number.
- Write the name of the compound; the parent name written as last part of the name by replacing ‘ane’ to ‘ene’. The name of the substituents is written as prefix and a hyphen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Explanation of Solution
Given structure:
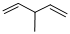
Predict the longest continuous chain of carbon atoms:
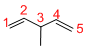
The longest continuous carbon chain of carbon atoms contains five carbons and the parent name is 1,4-PENTDIENE. The Suffix ‘diene’ represents the structure contains two double bonds at C-1 and C-4.
Predict substituents and its location:
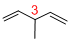
At each Carbon of 3 one methyl group is present. Hence, the structure containing one substituent, then prefix ‘3-methyl’ is added before the parent name. Hyphen separates the number that the substituent attached with carbon.
IUPAC name: 3-methyl-1,4-pentadiene.
e)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the substituent should get least possible number.
- Write the name of the compound; the parent name written as last part of the name by replacing ‘ane’ to ‘yne’. The name of the substituents is written as prefix and a hypen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Explanation of Solution
Given structure:

Predict the longest continuous chain of carbon atoms:
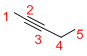
The longest continuous carbon chain of carbon atoms contains five carbons and the parent name is PENT-2-YNE. The Suffix ‘2-yne’ represents the structure contains one triple bond at C-2.
Predict substituents and its location:
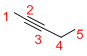
No substituents present and no prefix can be written. Hence, the name is,
IUPAC name: Pent-2-yne.
f)
Interpretation:
The given structure has to be named.
Concept introduction:
IUPAC Nomenclature for alkene:
- The longest continuous chain of carbon atoms is identified.
- The substituent groups attached to the parent chain is identified. A substituent group contains group of atoms attached to the carbon atom of the chain.
- While numbering the longest chain, the double bond should get least possible number.
- Write the name of the compound; the parent name written as last part of the name by replacing ‘ane’ to ‘ene’. The name of the substituents is written as prefix and a hypen separates the number that the substituents attached with carbon. More than one substituent should be written in alphabetical order.
Geometric isomers of Alkenes:
Cis-isomer: When two particular atoms (group of atoms) are adjacent to each other, the alkene is known as cis-isomer.
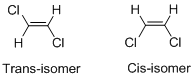
Trans-isomer: When two particular atoms (group of atoms) are across from each other, the alkene is known as cis-isomer.
Explanation of Solution
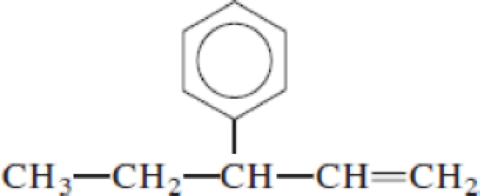
Given structure:
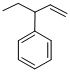
Predict the longest continuous chain of carbon atoms:
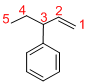
The longest continuous carbon chain of carbon atoms contains five carbons and the parent name is PENT-1-ENE. The Suffix ‘1-ene’ represents the structure contains one double bond at C-1.
Predict substituents and its location:

At each Carbon of 3 one phenyl group is present. Hence, the structure containing one substituent, then prefix ‘3-phenyl’ is added before the parent name. Hyphen separates the number that the substituent attached with carbon.
IUPAC name: 3-phenylpent-1-ene.
Want to see more full solutions like this?
Chapter 24 Solutions
Chemistry
- For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Molecule Inductive Effects O donating O withdrawing O no inductive effects Resonance Effects Overall Electron-Density ○ donating ○ withdrawing O no resonance effects O electron-rich O electron-deficient O similar to benzene Cl O donating O withdrawing ○ donating ○ withdrawing O no inductive effects O no resonance effects O Explanation Check O electron-rich O electron-deficient similar to benzene X © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessarrow_forwardIdentifying electron-donating and For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Molecule Inductive Effects NH2 ○ donating NO2 Explanation Check withdrawing no inductive effects Resonance Effects Overall Electron-Density ○ donating O withdrawing O no resonance effects O donating O withdrawing O donating withdrawing O no inductive effects Ono resonance effects O electron-rich electron-deficient O similar to benzene O electron-rich O electron-deficient O similar to benzene olo 18 Ar 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forwardRank each of the following substituted benzene molecules in order of which will react fastest (1) to slowest (4) by electrophilic aromatic substitution. Explanation Check Х (Choose one) OH (Choose one) OCH3 (Choose one) OH (Choose one) © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward
- Assign R or S to all the chiral centers in each compound drawn below porat bg 9 Br Brarrow_forwarddescrive the energy levels of an atom and howan electron moces between themarrow_forwardRank each set of substituents using the Cahn-Ingold-Perlog sequence rules (priority) by numbering the highest priority substituent 1.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





