
CHEMISTRY:CENTRAL SCIENCE-W/MOD.ACCESS
14th Edition
ISBN: 9780134809694
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 100IE
A typical amino acid with one amino group and one
- Suggest the forms of the amino acid at low PH and at high pH.
- Amino acids generally have two pKa values, one in the range of 2 to 3 and the other in the range of 9 to 10. Serine, for example, has pKa values of 2.19 and 9.21. Using species such as acetic acid and ammonia as models, suggest the origin of the two pKa values.
- Glutamic acid is an amino acid that has three pKa’s: 2.10, 4.07, and 9.47. Draw the structure of glutamic acid, and assign each pKa to the appropriate part of the molecule.
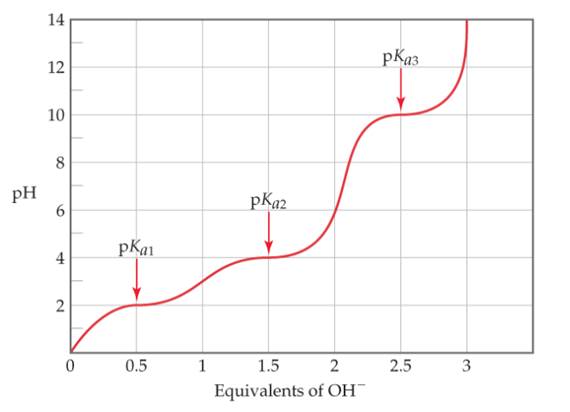
- An unknown amino acid is titrated with strong base, producing the following titration curve. Which amino acids are likely candidates for the unknown?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Redraw the molecule below as a skeletal ("line") structure. Be sure to use wedge and dash bonds if necessary to accurately
represent the direction of the bonds to ring substituents.
Cl.
Br
Click and drag to start drawing a
structure.
: ☐
☑
P
K
m
Choose the best reagents to complete the following reaction.
L
ZI
0
Problem 4 of 11
A
1. NaOH
2. CH3CH2CH2NH2
1. HCI
B
OH
2. CH3CH2CH2NH2
DII
F1
F2
F3
F4
F5
A
F6
C
CH3CH2CH2NH2
1. SOCl2
D
2. CH3CH2CH2NH2
1. CH3CH2CH2NH2
E
2. SOCl2
Done
PrtScn
Home
End
FA
FQ
510
*
PgUp
M
Submit
PgDn
F11
None
Chapter 24 Solutions
CHEMISTRY:CENTRAL SCIENCE-W/MOD.ACCESS
Ch. 24.2 - Prob. 24.1.1PECh. 24.2 - Prob. 24.1.2PECh. 24.2 - How many hydrogen atoms are in 2, 2-...Ch. 24.2 - Prob. 24.2.2PECh. 24.3 - Prob. 24.3.1PECh. 24.3 - Prob. 24.3.2PECh. 24.3 - Prob. 24.4.1PECh. 24.3 - Prob. 24.4.2PECh. 24.3 - Prob. 24.5.1PECh. 24.3 - Prob. 24.5.2PE
Ch. 24.4 - Prob. 24.6.1PECh. 24.4 - Prob. 24.6.2PECh. 24.7 - Prob. 24.7.1PECh. 24.7 - Practice Exercise 2 Name the dipeptide and give...Ch. 24.7 - How many chiral carbon atoms are there in the...Ch. 24.7 - Prob. 24.8.2PECh. 24 - Prob. 1DECh. 24 - Prob. 1ECh. 24 - Prob. 2ECh. 24 - Prob. 3ECh. 24 - Prob. 4ECh. 24 - Prob. 5ECh. 24 - Prob. 6ECh. 24 - Prob. 7ECh. 24 - Prob. 8ECh. 24 - Prob. 9ECh. 24 - Prob. 10ECh. 24 - Prob. 11ECh. 24 - Prob. 12ECh. 24 - Prob. 13ECh. 24 - Prob. 14ECh. 24 - Prob. 15ECh. 24 - Prob. 16ECh. 24 - Prob. 17ECh. 24 - Prob. 18ECh. 24 - Prob. 19ECh. 24 - Prob. 20ECh. 24 - Prob. 21ECh. 24 - Prob. 22ECh. 24 - Prob. 23ECh. 24 - Prob. 24ECh. 24 - Prob. 25ECh. 24 - Prob. 26ECh. 24 - Prob. 27ECh. 24 - Prob. 28ECh. 24 - Prob. 29ECh. 24 - Prob. 30ECh. 24 - Prob. 31ECh. 24 - Prob. 32ECh. 24 - Prob. 33ECh. 24 - Prob. 34ECh. 24 - Prob. 35ECh. 24 - Prob. 36ECh. 24 - Prob. 37ECh. 24 - Prob. 38ECh. 24 - Prob. 39ECh. 24 - Describe the intermediate that is thought to form...Ch. 24 - Prob. 41ECh. 24 - Prob. 42ECh. 24 - Prob. 43ECh. 24 - Prob. 44ECh. 24 - Prob. 45ECh. 24 - Prob. 46ECh. 24 - Prob. 47ECh. 24 - Prob. 48ECh. 24 - Prob. 49ECh. 24 - Prob. 50ECh. 24 - Prob. 51ECh. 24 - Prob. 52ECh. 24 - Prob. 53ECh. 24 - Prob. 54ECh. 24 - Prob. 55ECh. 24 - Prob. 56ECh. 24 - Prob. 57ECh. 24 - Prob. 58ECh. 24 - Prob. 59ECh. 24 - Prob. 60ECh. 24 - Prob. 61ECh. 24 - Prob. 62ECh. 24 - Prob. 63ECh. 24 - Prob. 64ECh. 24 - Prob. 65ECh. 24 - Prob. 66ECh. 24 - Prob. 67ECh. 24 - Prob. 68ECh. 24 - Prob. 69ECh. 24 - Prob. 70ECh. 24 - Prob. 71ECh. 24 - Prob. 72ECh. 24 - Prob. 73ECh. 24 - Prob. 74ECh. 24 - Prob. 75ECh. 24 - Prob. 76ECh. 24 - Prob. 77ECh. 24 - Prob. 78ECh. 24 - Prob. 79ECh. 24 - Prob. 80ECh. 24 - Prob. 81AECh. 24 - Prob. 82AECh. 24 - Prob. 83AECh. 24 - Prob. 84AECh. 24 - Prob. 85AECh. 24 - Prob. 86AECh. 24 - Prob. 87AECh. 24 - Prob. 88AECh. 24 - Prob. 89AECh. 24 - Prob. 90AECh. 24 - Prob. 91AECh. 24 - Prob. 92AECh. 24 - Prob. 93AECh. 24 - Prob. 94AECh. 24 - Prob. 95IECh. 24 - Prob. 96IECh. 24 - Prob. 97IECh. 24 - Prob. 98IECh. 24 - Prob. 99IECh. 24 - A typical amino acid with one amino group and one...Ch. 24 - Prob. 101IECh. 24 - Prob. 102IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Add curved arrows to the reactants in this reaction. A double-barbed curved arrow is used to represent the movement of a pair of electrons. Draw curved arrows. : 0: si H : OH :: H―0: Harrow_forwardConsider this step in a radical reaction: Br N O hv What type of step is this? Check all that apply. Draw the products of the step on the right-hand side of the drawing area below. If more than one set of products is possible, draw any set. Also, draw the mechanism arrows on the left-hand side of the drawing area to show how this happens. O primary Otermination O initialization O electrophilic O none of the above × ☑arrow_forwardNonearrow_forward
- Can I get a drawing of what is happening with the orbitals (particularly the p orbital) on the O in the OH group? Is the p orbital on the O involved in the ring resonance? Why or why not?arrow_forward1) How many monochlorination products-including stereochemistry- are there for the molecule below:arrow_forwardSelect an amino acid that has and N-H or O-H bond in its R-group (you have 8 to choose from!). Draw at least two water molecules interacting with the R-group of the amino acid.arrow_forward
- Is this aromatic?arrow_forwardCHEM2323 E Tt PS CH03 Draw and name all monobromo derivatives of pentane, C5H11Br. Problem 3-33 Name: Draw structures for the following: (a) 2-Methylheptane (d) 2,4,4-Trimethylheptane Problem 3-35 (b) 4-Ethyl-2,2-dimethylhexane (e) 3,3-Diethyl-2,5-dimethylnonane (c) 4-Ethyl-3,4-dimethyloctane 2 (f) 4-Isopropyl-3-methylheptane KNIE>arrow_forwardProblem 3-42 Consider 2-methylbutane (isopentane). Sighting along the C2-C3 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. Problem 3-44 Construct a qualitative potential-energy diagram for rotation about the C-C bond of 1,2-dibromoethane. Which conformation would you expect to be most stable? Label the anti and gauche conformations of 1,2- dibromoethane. Problem 3-45 Which conformation of 1,2-dibromoethane (Problem 3-44) would you expect to have the largest dipole moment? The observed dipole moment of 1,2-dibromoethane is µ = 1.0 D. What does this tell you about the actual conformation of the molecule?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY