
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
12th Edition
ISBN: 9781119766919
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 22, Problem 42P
Interpretation Introduction
Interpretation:
The structure of the given compound is to be determined, based on the
Concept introduction:
Alditols are the sugar alcohols formed by the addition of hydrogen to a monosaccharide. The reduction of aldoses or ketoses through sodium borohydride (
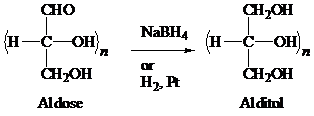
The
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the general properties of equilibrium constants
At a certain temperature, the equilibrium constant K for the following reaction is 0.84:
H2(g) + 2(g) 2 HI(g)
=
Use this information to complete the following table.
Suppose a 34. L reaction vessel is filled with 0.79 mol of HI.
What can you say about the composition of the mixture in the
vessel at equilibrium?
There will be very little H2 and 12.
☐ x10
There will be very little HI.
Neither of the above is true.
What is the equilibrium constant for the following reaction? Be
sure your answer has the correct number of significant digits.
2 HI(g)
H₂(9)+12(9)
K =
What is the equilibrium constant for the following reaction? Be
sure your answer has the correct number of significant digits.
2 H2(g)+212(9)
4 HI(g)
K = ☐
☑
Predicting the qualitative acid-base properties of salts
Consider the following data on some weak acids and weak bases:
base
acid
Κα
Kb
name
formula
name
formula
hydrocyanic acid
- 10
HCN
4.9 × 10
pyridine
C₂H₂N 1.7 × 10 9
acetic acid
HCH3CO2 1.8 × 10
5
hydroxylamine HONH2 1.1 × 10¯
8
Use this data to rank the following solutions in order of increasing pH. In other words, select a '1' next to the solution that will have the lowest pH, a '2' next to
the solution that will have the next lowest pH, and so on.
0.1 M KCN
solution
pH
choose one ✓
0.1 M C5H5NHCI
choose one ✓
0.1 M NaCH3CO2
choose one ✓
0.1 M HONH3Br
✓ choose one
1 (lowest)
2
3
4 (highest)
For this question please solve the first question. Please explain your thought process, the steps you took, and how you would tackle a similar problem. Thank you for your help!
Chapter 22 Solutions
ORGANIC CHEMISTRY-WILEYPLUS ACCESS PKG.
Ch. 22 - Prob. 1PPCh. 22 - Prob. 2PPCh. 22 - Prob. 3PPCh. 22 - Prob. 4PPCh. 22 - Prob. 5PPCh. 22 - Prob. 6PPCh. 22 - Prob. 7PPCh. 22 - Prob. 8PPCh. 22 - Practice Problem 22.9 What products would you...Ch. 22 - Prob. 10PP
Ch. 22 - Prob. 11PPCh. 22 - Prob. 12PPCh. 22 - Prob. 13PPCh. 22 - Prob. 14PPCh. 22 - Prob. 15PPCh. 22 - Prob. 16PPCh. 22 - Prob. 17PPCh. 22 - Prob. 18PPCh. 22 - Prob. 19PPCh. 22 - Prob. 20PCh. 22 - Prob. 21PCh. 22 - Prob. 22PCh. 22 - Prob. 23PCh. 22 - Prob. 24PCh. 22 - Prob. 25PCh. 22 - Prob. 26PCh. 22 - Prob. 27PCh. 22 - Prob. 28PCh. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - Prob. 31PCh. 22 - Prob. 32PCh. 22 - Prob. 33PCh. 22 - Prob. 34PCh. 22 - Prob. 35PCh. 22 - Prob. 36PCh. 22 - Prob. 37PCh. 22 - Prob. 38PCh. 22 - Arbutin, a compound that can be isolated from the...Ch. 22 - Prob. 40PCh. 22 - Prob. 41PCh. 22 - Prob. 42PCh. 22 - Prob. 43PCh. 22 - 22.44 The following reaction sequence represents...Ch. 22 - 22.45
The NMR data for the two anomers...Ch. 22 - Shikimic acid is a key biosynthetic intermediate...
Knowledge Booster
Similar questions
- Part C A solution that is 0.040 M in HCIO4 and 0.046 M in HCI Express your answer numerically to two decimal places. ΜΕ ΑΣΦ ? pH = Submit Request Answer Part D A solution that is 1.08% HCl by mass (with a density of 1.01 g/mL) Express your answer numerically to three decimal places. ΜΕ ΑΣΦ -> 0 ? pH =arrow_forwardPredict the equilibrium arrows for the following reaction:*see imagearrow_forwardProvide the missing information for each of the two following reacitons: *see imagearrow_forward
- Draw an example of the following functional groups: *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. A protecting group will be needed. *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. *see imagearrow_forward
- Provide the missing information for each of the four reactions: *see imagearrow_forward6. Chlorine dioxide (CIO) is used as a disinfectant in municipal water-treatment plants. It decomposes in a first-order reaction with a rate constant of 14 s. How long would it take for an initial concentration of 0.06 M to decrease to 0.02 M? [6 pts]arrow_forwardIf possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ẞ carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. en HO OHarrow_forward
- Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the intermediate and product of this hydrohalogenation reaction. Include all lone pairs and charges as appropriate. Br Select to Draw 51°F Sunny esc F1 HBr Select to Draw 1,2-hydride shift Br Select to Draw Q Search F2 F3 F4 1 2 # # 3 DII L F5 F6 F tA $ % Λarrow_forwardplease help i cant find the article to even startarrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole