
Chemistry (7th Edition)
7th Edition
ISBN: 9780321943170
Author: John E. McMurry, Robert C. Fay, Jill Kirsten Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 22, Problem 22.13P
Interpretation Introduction
Interpretation:
To justify why diborane structure is not like ethane structure.
Concept introduction:
Diborane the combination of hydrogen and boron which has the chemical formula 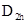 structure which has terminal four and couple of bridging hydrogen atoms. This bond type is referred as ‘banana bond’.
structure which has terminal four and couple of bridging hydrogen atoms. This bond type is referred as ‘banana bond’.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
7. Help
11. Help
8. Help
Chapter 22 Solutions
Chemistry (7th Edition)
Ch. 22 - Prob. 22.1PCh. 22 - Prob. 22.2ACh. 22 - Prob. 22.3PCh. 22 - Prob. 22.4CACh. 22 - Prob. 22.5PCh. 22 - Prob. 22.6PCh. 22 - Prob. 22.7ACh. 22 - Prob. 22.8CPCh. 22 - Prob. 22.9ACh. 22 - Prob. 22.10P
Ch. 22 - Prob. 22.11PCh. 22 - Prob. 22.12PCh. 22 - Prob. 22.13PCh. 22 - Prob. 22.14PCh. 22 - Prob. 22.15PCh. 22 - Prob. 22.16PCh. 22 - Prob. 22.17CPCh. 22 - Prob. 22.18CACh. 22 - Prob. 22.19PCh. 22 - Prob. 22.20PCh. 22 - Prob. 22.21PCh. 22 - Conceptual PRACTICE 22.22 Look at the location of...Ch. 22 - Prob. 22.23ACh. 22 - Prob. 22.24CPCh. 22 - Prob. 22.25PCh. 22 - Prob. 22.26PCh. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Prob. 22.30PCh. 22 - Prob. 22.31PCh. 22 - Prob. 22.32PCh. 22 - Prob. 22.33CPCh. 22 - Prob. 22.34CPCh. 22 - Prob. 22.35CPCh. 22 - Prob. 22.36CPCh. 22 - Prob. 22.37CPCh. 22 - Prob. 22.38CPCh. 22 - Prob. 22.39CPCh. 22 - Prob. 22.40CPCh. 22 - Prob. 22.41CPCh. 22 - Prob. 22.42CPCh. 22 - Prob. 22.43CPCh. 22 - Prob. 22.44CPCh. 22 - Prob. 22.45CPCh. 22 - Prob. 22.46CPCh. 22 - Prob. 22.47CPCh. 22 - Prob. 22.48SPCh. 22 - Prob. 22.49SPCh. 22 - Prob. 22.50SPCh. 22 - Prob. 22.51SPCh. 22 - Prob. 22.52SPCh. 22 - Prob. 22.53SPCh. 22 - Prob. 22.54SPCh. 22 - Prob. 22.55SPCh. 22 - Prob. 22.56SPCh. 22 - Prob. 22.57SPCh. 22 - Prob. 22.58SPCh. 22 - Prob. 22.59SPCh. 22 - Prob. 22.60SPCh. 22 - Prob. 22.61SPCh. 22 - Prob. 22.62SPCh. 22 - Prob. 22.63SPCh. 22 - Prob. 22.64SPCh. 22 - Prob. 22.65SPCh. 22 - Prob. 22.66SPCh. 22 - Prob. 22.67SPCh. 22 - Prob. 22.68SPCh. 22 - Prob. 22.69SPCh. 22 - Prob. 22.70SPCh. 22 - Prob. 22.71SPCh. 22 - Prob. 22.72SPCh. 22 - Prob. 22.73SPCh. 22 - Prob. 22.74SPCh. 22 - Prob. 22.75SPCh. 22 - Prob. 22.76SPCh. 22 - Prob. 22.77SPCh. 22 - Prob. 22.78SPCh. 22 - Prob. 22.79SPCh. 22 - Prob. 22.80SPCh. 22 - Prob. 22.81SPCh. 22 - Prob. 22.82SPCh. 22 - Prob. 22.83SPCh. 22 - Prob. 22.84SPCh. 22 - Prob. 22.85SPCh. 22 - Magnesium metal is produced by electrolysis of...Ch. 22 - Prob. 22.87SPCh. 22 - Prob. 22.88SPCh. 22 - Prob. 22.89SPCh. 22 - Prob. 22.90SPCh. 22 - Prob. 22.91SPCh. 22 - Prob. 22.92SPCh. 22 - Prob. 22.93SPCh. 22 - Prob. 22.94SPCh. 22 - Prob. 22.95SPCh. 22 - Prob. 22.96SPCh. 22 - Prob. 22.97SPCh. 22 - Prob. 22.98SPCh. 22 - Prob. 22.99SPCh. 22 - Prob. 22.100SPCh. 22 - Prob. 22.101SPCh. 22 - Prob. 22.102SPCh. 22 - Prob. 22.103SPCh. 22 - Prob. 22.104SPCh. 22 - Prob. 22.105SPCh. 22 - Prob. 22.106SPCh. 22 - Prob. 22.107SPCh. 22 - Prob. 22.108SPCh. 22 - Prob. 22.109SPCh. 22 - Prob. 22.110SPCh. 22 - Prob. 22.111SPCh. 22 - Prob. 22.112SPCh. 22 - Prob. 22.113SPCh. 22 - Prob. 22.114SPCh. 22 - Prob. 22.115SPCh. 22 - Prob. 22.116SPCh. 22 - Prob. 22.117SPCh. 22 - Prob. 22.118SPCh. 22 - Prob. 22.119SPCh. 22 - Prob. 22.120SPCh. 22 - Prob. 22.121SPCh. 22 - Prob. 22.122SPCh. 22 - Prob. 22.123SPCh. 22 - Prob. 22.124SPCh. 22 - Prob. 22.125SPCh. 22 - Prob. 22.126SPCh. 22 - Prob. 22.127SPCh. 22 - Prob. 22.128SPCh. 22 - Prob. 22.129SPCh. 22 - Prob. 22.130SPCh. 22 - Prob. 22.131SPCh. 22 - Prob. 22.132SPCh. 22 - Prob. 22.133SPCh. 22 - Prob. 22.134SPCh. 22 - Prob. 22.135SPCh. 22 - Prob. 22.136SPCh. 22 - Prob. 22.137SPCh. 22 - Prob. 22.138SPCh. 22 - Prob. 22.139SPCh. 22 - Prob. 22.140SPCh. 22 - Prob. 22.141SPCh. 22 - Prob. 22.142SPCh. 22 - Prob. 22.143SPCh. 22 - Prob. 22.144SPCh. 22 - Prob. 22.145SPCh. 22 - Prob. 22.146SPCh. 22 - Prob. 22.147SPCh. 22 - Prob. 22.148SPCh. 22 - Prob. 22.149SPCh. 22 - Prob. 22.150SPCh. 22 - Prob. 22.151SPCh. 22 - Prob. 22.152SPCh. 22 - Prob. 22.153SPCh. 22 - Prob. 22.154SPCh. 22 - Prob. 22.155SPCh. 22 - Prob. 22.156SPCh. 22 - Prob. 22.157SPCh. 22 - Prob. 22.158CPCh. 22 - Prob. 22.159CPCh. 22 - Prob. 22.160CPCh. 22 - Prob. 22.161CPCh. 22 - Prob. 22.162CPCh. 22 - Prob. 22.163CPCh. 22 - Prob. 22.164CPCh. 22 - Prob. 22.165CPCh. 22 - Prob. 22.166CPCh. 22 - Prob. 22.167CPCh. 22 - Prob. 22.168CPCh. 22 - Prob. 22.169CPCh. 22 - Prob. 22.170CPCh. 22 - Prob. 22.171CPCh. 22 - Prob. 22.172CPCh. 22 - Prob. 22.173CPCh. 22 - Prob. 22.174CPCh. 22 - Prob. 22.175MPCh. 22 - Prob. 22.176MPCh. 22 - Prob. 22.177MPCh. 22 - Prob. 22.178MPCh. 22 - Prob. 22.179MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following are descriptions of possible starting material for this reaction? H ? trace acid an ester a ketone an imine an aldehyde a carboxylic acid an enamine a primary amine a secondary amine a tertiary aminearrow_forwardNonearrow_forwardWhat are the reagents needed for this and the third structure I only got the top right structure rightarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY