
Concept explainers
Complete the following concept map to review some of the concepts of gas exchange.
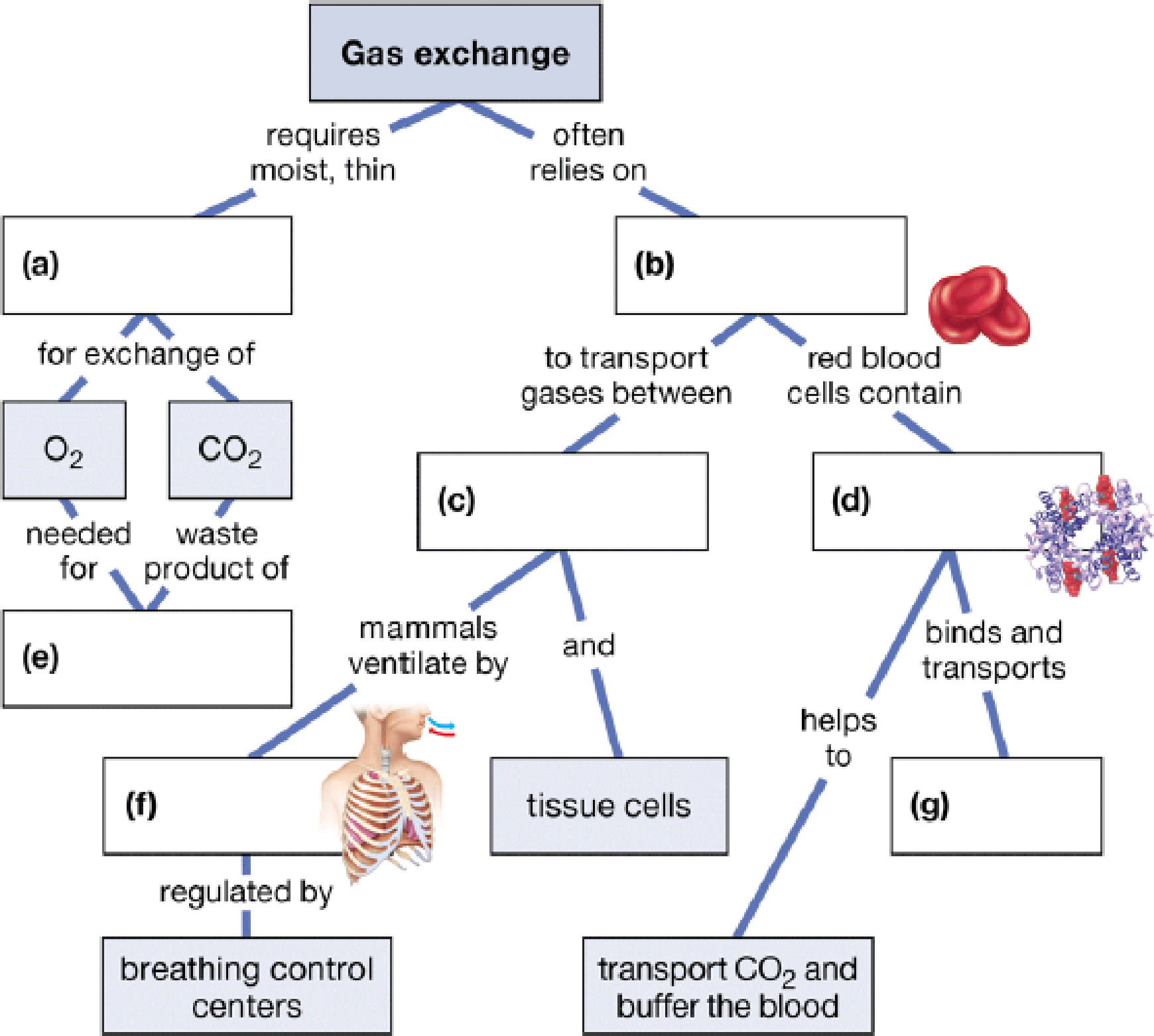
To complete: The given map to review some of the concepts of gas exchange.
Introduction:
The respiratory system is a biological system that ensures the transport of air by the process of inhalation and exhalation, that is, in and out the body with the help of the lungs.
Answer to Problem 1CC
Fig.1 shows the completed map to review some of the concepts of gas exchange.
Pictorial representation: Some of the concepts related to gas exchange are presented in the concept map given in Fig.1.
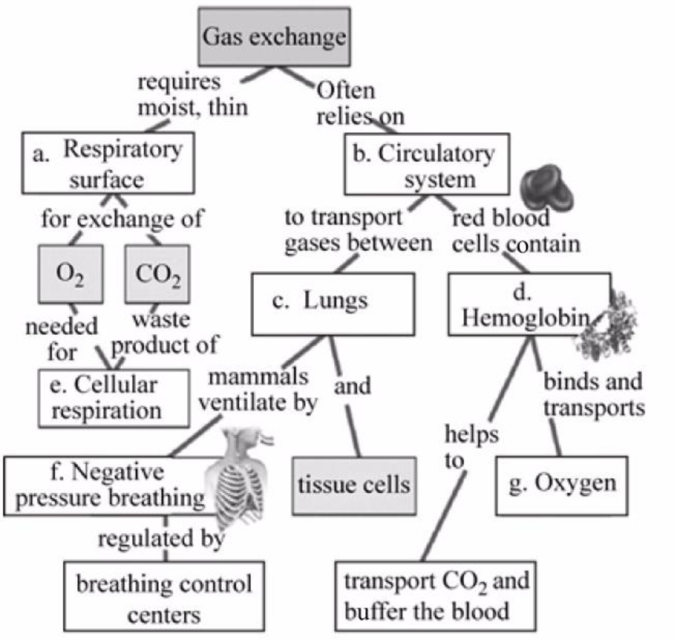
Fig.1: Concepts of gas exchange.
Explanation of Solution
a.
Correct answer: Respiratory surface.
For gas exchange to occur, the respiratory surface should be thin and moist. Hence, the correct answer is the respiratory surface.
b.
Correct answer: Circulatory system.
Gas exchange relies on the circulatory system as the exchanged oxygen and carbon dioxide travel in the blood to the heart and different body cells. Hence, the correct answer is the circulatory system.
c.
Correct answer: Lungs
It is the main organ for the exchange of gases consisting of air sacs that carry out gaseous exchange and the blood carries the exchanged gas to the tissue cells. Hence, the correct answer is the lungs.
d.
Correct answer: Hemoglobin.
Hemoglobin is a blood pigment present inside the red blood cell, which helps in the transport of oxygen and carbon dioxide by binding with it and also maintains the pH of the blood. Hence, the correct answer is hemoglobin.
e.
Correct answer: Cellular respiration.
In cellular respiration, oxygen is used to convert glucose into carbon dioxide, water, and energy in the form of ATP. Hence, the correct answer is cellular respiration.
f.
Correct answer: Negative pressure breathing.
This type of breathing is regulated by the “breathing control center of the brain”. In negative pressure breathing, the air pressure inside the lungs drops during the contraction of the diaphragm, and the lower portion of the chest cavity rises; this allows the movement of air from outside to inside. Hence, the correct answer is negative pressure breathing.
g.
Correct answer: Oxygen.
Hemoglobin pigment in the blood binds with the oxygen present in the lungs, and this oxygen is utilized to form energy. Hence, the correct answer is oxygen.
Want to see more full solutions like this?
Chapter 22 Solutions
Campbell Biology: Concepts & Connections, Books a la Carte Plus Mastering Biology with eText -- Access Card Package (8th Edition)
- Calculate pressure using Dalton's law of partial pressures. A gas mixture is made up of N2 (6.91 g), 02 (7.95 g), and H2 (0.850 g). The mixture has a volume of 27.1 L at 27 °C. Calculate the partial pressure of each gas in the mixture and the total pressure of the gas mixture. PN2 = P02 PH = H2 = Ptotal atm atm atm atmarrow_forwardGas moves from an area of ________ partial pressure to an area of ________ partial pressure. low; high low; low high; high high; lowarrow_forwarddefine the equilibrium constant KP for the chemical equilibrium ofideal-gas mixtures?arrow_forward
- Using the information below, calculate the Oxygen Diffusion Driving Force (mmHg), which is the pressure gradient that drives O₂ out of the alveoli and into the blood (calculated as PAO₂- PVO₂): Barometric Pressure at Salt Lake City, UT= 4,226 ft. (1,288 m)→→ 657 mmHg . Estimated mixed-venous PO2 of blood returning to the lungs after leaving the muscle (PvO₂) at rest, measured at Salt Lake City, UT: PvO2 = 36 mmHg O 90 mmHg O 54 mmHg O 36 mmHg O 29 mmHgarrow_forwardUsing the regional barometric pressure info. below, and being careful to consider the vapor pressure in saturated alveolar air, calculate the alveolar oxygen pressure (PAO₂) (mmHg) for the following location: . Salt Lake City, UT= 4,226 ft. (1,288 m)→ 657 mmHg O 90 mmHg O 16 mmHg O 137.5 mmHg O 81 mmHgarrow_forward(a) The partial pressure of CO2 in the lungs can be varied rapidly by the rate and depth of breathing. For example, a common remedy to alleviate hiccups is to increase the concentration of CO2 in the lungs. This can be achieved by holding one’s breath, by very slow and shallow breathing (hypoventilation), or by breathing in and out of a paper bag.Under such conditions, pCO2 in the air space of the lungs rises above normal. Qualitatively explain the effect of these procedures on the blood pH.(b) A common practice of competitive short-distance runners is to breathe rapidly and deeply (hyperventilate) for about half a minute to remove CO2from their lungs just before the race begins. Blood pH may rise to 7.60. Explain why the blood pH increases.(c) During a short-distance run, the muscles produce a large amount of lactic acid (CH2CH(OH)COOH; K2 = 1.38 × 10 –4 M) from their glucose stores. Why might hyperventilation before a dash be useful?arrow_forward
- The human lungs can function satisfactorily up to a limit where the pressure difference between the outside and inside of the lungs is 1/19 of an atmosphere. If a diver uses a snorkel for breathing, how far below the water can she swim? Assume the diver is in saltwater its density is 1047 kg/m. Atmospheric pressure is Pata-1.01x10 Pa and g=9.8 m/s?.arrow_forwardDraw or illustrate how the described result looks like in a tube.A/K, no H2S, gas (+)andK/A, H2S in butt, gas (+)arrow_forwardChoose the three option A, B, Carrow_forward
- If 46.5 mol of an ideal gas occupies 95.5 L at 373 K, what is the pressure of the gas? pressure: I atmarrow_forwardArrange the following according to decreasing oxygen requirement. 1. crab 2. shark 3. clown fish 4. perch 5. hake Select one: O 12345 O 34512 O 54321 O 21543arrow_forwardA typical room is 4.0 m long, 5.0 m wide, and 2.5 m high. What is the total mass of the oxygen in the room assuming that the gas in the room is at STP and that air contains 21% oxygen and 79% nitrogen?arrow_forward
 Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax CollegeBasic Clinical Lab Competencies for Respiratory C...NursingISBN:9781285244662Author:WhitePublisher:Cengage
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax CollegeBasic Clinical Lab Competencies for Respiratory C...NursingISBN:9781285244662Author:WhitePublisher:Cengage





