
EP MICROBIOLOGY:W/DISEASES BY..-MOD.ACC
5th Edition
ISBN: 9780134607894
Author: BAUMAN
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2VI
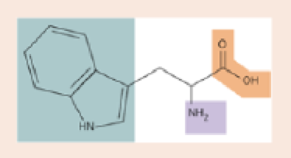
Shown is the amino acid tryptophan. Put the letter “C” at the site of every carbon atom. Label the amino group, the carboxyl group, and the side group.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please answer q3
Respond to the following in a minimum of 175 words:
How might CRISPR-Cas 9 be used in research or, eventually, therapeutically in patients?
What are some potential ethical issues associated with using this technology?
Do the advantages of using this technology outweigh the disadvantages (or vice versa)? Explain your position.
You are studying the effect of directional selection on body height in three
populations (graphs a, b, and c below).
(a) What is the selection differential? Show your calculation. (2 pts)
(b) Which population has the highest narrow sense heritability for height?
Explain your answer. (2 pts)
(c) If you examined the offspring in the next generation in each population,
which population would have the highest mean height? Why? (2 pts)
(a)
Midoffspring height (average height of offspring)
Short
Short
Short
Short
(c)
Short
(b)
Short
Tall
Short
Tall
Short
Short
Tall
Midparent height
(average height of
Mean of population = 65 inches
Mean of breading parents = 70 inches
Mean of population = 65 inches
Mean of breading parents = 70 inches
Mean of population = 65 inches
Mean of breading parents = 70 inches
Chapter 2 Solutions
EP MICROBIOLOGY:W/DISEASES BY..-MOD.ACC
Ch. 2 - Electrons zip around the nucleus at about 5...Ch. 2 - Chlorine and potassium atoms form ionic bonds,...Ch. 2 - Why are decomposition reactions exothermic, that...Ch. 2 - Why does the neutralization of an acid by a base...Ch. 2 - Raw Oysters and Antacids: A Deadly Mix? The highly...Ch. 2 - Why do the cell membranes of microbes living in...Ch. 2 - Prob. 1MCCh. 2 - The atomic mass of an atom most closely...Ch. 2 - One isotope of iodine differs from another in...Ch. 2 - Prob. 4MC
Ch. 2 - Which of the following terms most correctly...Ch. 2 - In water, cations and anions of salts dissociate...Ch. 2 - Prob. 7MCCh. 2 - Which of the following statements about a...Ch. 2 - Proteins are polymers of ___________. a. amino...Ch. 2 - Which of the following are hydrophobic organic...Ch. 2 - Fill in the Blanks 1. The outermost electron shell...Ch. 2 - Fill in the Blanks 2. The type of chemical bond...Ch. 2 - Prob. 3FIBCh. 2 - Prob. 4FIBCh. 2 - Fill in the Blanks 5. Groups of atoms such as NH2...Ch. 2 - Fill in the Blanks 6. The reverse of dehydration...Ch. 2 - Fill in the Blanks 7. Reactions that release...Ch. 2 - Fill in the Blanks 8. All chemical reactions begin...Ch. 2 - Fill in the Blanks 9. The ____________ scale is a...Ch. 2 - Prob. 10FIBCh. 2 - Label a portion of the molecule below; label two...Ch. 2 - Shown is the amino acid tryptophan. Put the letter...Ch. 2 - List three main types of chemical bonds, and give...Ch. 2 - Name five properties of water that are vital to...Ch. 2 - Prob. 3SACh. 2 - What is the difference between atomic oxygen and...Ch. 2 - Explain how the polarity of water molecules makes...Ch. 2 - Prob. 1CTCh. 2 - Prob. 2CTCh. 2 - Two freshmen disagree about an aspect of...Ch. 2 - When an egg white is heated, it changes from...Ch. 2 - Prob. 5CTCh. 2 - The poison glands of many bees and wasps contain...Ch. 2 - Prob. 7CTCh. 2 - Prob. 8CTCh. 2 - The deadly poison hydrogen cyanide has the...Ch. 2 - Triple covalent bonds are stronger and more...Ch. 2 - How can hydrogen bonding between water molecules...Ch. 2 - How can a single molecule of magnesium hydroxide...Ch. 2 - Prob. 13CTCh. 2 - Prob. 14CTCh. 2 - A textbook states that only five nucleotide bases...Ch. 2 - Using the following terms, fill in the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- P You are studying a population of 100 flowers that has two alleles at a locus for flower color, blue (B) and green (G). There are 15 individuals with the BB genotype, 70 individuals with the BG genotype, and 15 individuals with the GG genotype. (a) What are the allele frequencies of B and G in the starting population? Show your calculations. (2 pts) (b) Is this population in Hardy-Weinberg equilibrium? Show your calculations. (3 pts) 12pt v Paragraph BIU UA AV & VT2V f CO Varrow_forwardIn a natural population of outbreeding plants, the variance of the total number of seeds per plant is 16. From the natural population, 20 plants are taken into the laboratory and developed into separate true-breeding lines by self- fertilization-with selection for high, low, or medium number of seeds-for 10 generations. The average variance in the tenth generation in each of the 20 sets is about equal and averages 5.8 across all the sets. Estimate the broad-sense heritability for seed number in this population. (4 pts) 12pt v Paragraph BIUA V V T² v B ① O wordsarrow_forwardIn a natural population of outbreeding plants, the variance of the total number of seeds per plant is 16. From the natural population, 20 plants are taken into the laboratory and developed into separate true-breeding lines by self- fertilization-with selection for high, low, or medium number of seeds-for 10 generations. The average variance in the tenth generation in each of the 20 sets is about equal and averages 5.8 across all the sets. Estimate the broad-sense heritability for seed number in this population. (4 pts) 12pt v Paragraph BI DI T² v ✓ B°arrow_forward
- Question 1 In a population of Jackalopes (pictured below), horn length will vary between 0.5 and 2 feet, with the mean length somewhere around 1.05 feet. You pick Jackalopes that have horn lengths around 1.75 feet to breed as this appears to be the optimal length for battling other Jackalopes for food. After a round of breeding, you measure the offsprings' mean horn length is 1.67. What is the heritability of horns length (h2)? Is Jackalope horn length a heritable trait? (4 pts)? 12pt v Paragraph BIU A ✓arrow_forwardFrequency of allele A1 Question 2 The graph below shows results of two simulations, both depicting the rise in frequency of beneficial allele in a population of infinite size. The selection coefficient and the starting frequency are the same, but in one simulation the beneficial allele is dominant and in the other it is recessive. Neither allele is fixed by 500 generations. 1.0 1 0.8 0.6 0.4 2 0.2 0 0 100 200 300 400 500 Generation (a) Which simulation shows results for a dominant and which shows results for a recessive allele? How can you tell? (4 pts) (b) Neither of the alleles reaches fixation by 500 generations. If given enough time, will both of these alleles reach fixation in the population? Why or why not? (3 pts) 12pt Paragraph BIU AT2v Varrow_forwardQuestion 14 The relative fitnesses of three genotypes are WA/A= 1.0, WA/a = 0.7, and Wa/a = 0.3. If the population starts at the allele frequency p = 0.5, what is the value of p in the next generation? (3 pts) 12pt v V Paragraph B I U D V T² v V V p O words <arrow_forward
- According to a recent study, 1 out of 50,000 people will be diagnosed with cystic fibrosis. Cystic fibrosis can be caused by a mutant form of the CFTR gene (dominant gene symbol is F and mutant is f). A. Using the rate of incidence above, what is the frequency of carriers of the cystic fibrosis allele for CFTR in the US? (3 pts) B. In a clinical study, 400 people from the population mentioned in (A.) were genotyped for BRCA1 Listed below are the results. Are these results in Hardy- Weinberg equilibrium? Use Chi Square to show whether or not they are. (3 pts) BRCA1 genotype # of women 390 BB Bb bb 10 0 12pt Paragraph L BIUAV V T² v Varrow_forwardOutline a method for using apomixis to maintain feminized CannabisAssume apomixis is controlled by a single dominant gene. You can choose the type of apomixis: obligate or facultative, gametophytic or sporophytic. Discuss advantages and disadvantages of your proposed method.arrow_forwardKinetics: One-Compartment First-Order Absorption 1. In vivo testing provides valuable insight into a drug’s kinetics. Assessing drug kinetics following multiple routes of administration provides greater insight than a single route of administration alone. The following data was collected in 250-g rats following bolus IV, oral (PO), and intraperitoneal (ip) administration. Using this data and set of graphs, determine:(calculate for each variable) (a) k, C0, V, and AUC* for the bolus iv data (b) k, ka, B1, and AUC* for the po data c) k, ka, B1, and AUC* for the ip data (d) relative bioavailability for po vs ip, Fpo/Fip (e)absolute ip bioavailability, Fip (f) absolute po bioavailability, Fpoarrow_forward
- 3. A promising new drug is being evaluated in human trials. Based on preliminary human tests, this drug is most effective when plasma levels exceed 30 mg/L. Measurements from preliminary tests indicate the following human pharmacokinetic parameter values: t1/2,elim = 4.6hr, t1/2,abs = 0.34hr, VD = 0.29 L/kg, Foral = 72%. Based on these parameters, estimate the following if a 49 kg woman were to receive a 1000mg oral dose of this drug: (a) Estimate the plasma concentration of the drug at 1hr, 6 hr, and 20hr after taking the drug ( Concentration estimate) (b) Estimate the time for maximum plasma concentration (tmax). (c) Estimate the maximum plasma concentration (Cmax). (d) Estimate the time at which the plasma level first rises above 30 mg/L. (Note this is a trial and error problem where you must guess a time, plug it into the concentration equation, and determine if it is close to 30 mg/L. Hint: based on part (a) it should be apparent that the answer is less than 1hr.) (e)…arrow_forwardList substitutions in your diet you could make to improve it based on what you know now about a balanced diet. For instance, if you like to drink soda, you might substitute skim milk or water for some of the soft drinks you consumed. List the item you wish to replace with the new item and what you hope to accomplish with that substitution. Be sure to choose foods you know that you'd enjoy and you consider more "healthful." If you feel your diet is already balanced, describe how you accomplish your balanced intake and when you began eating this way.arrow_forwardWhich single food item contained you ate for the 3 days with the most sodium?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning


Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning


Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax


Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license