
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305865617
Author: Brown, Iverson, Anslyn, FOOTE
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.57P
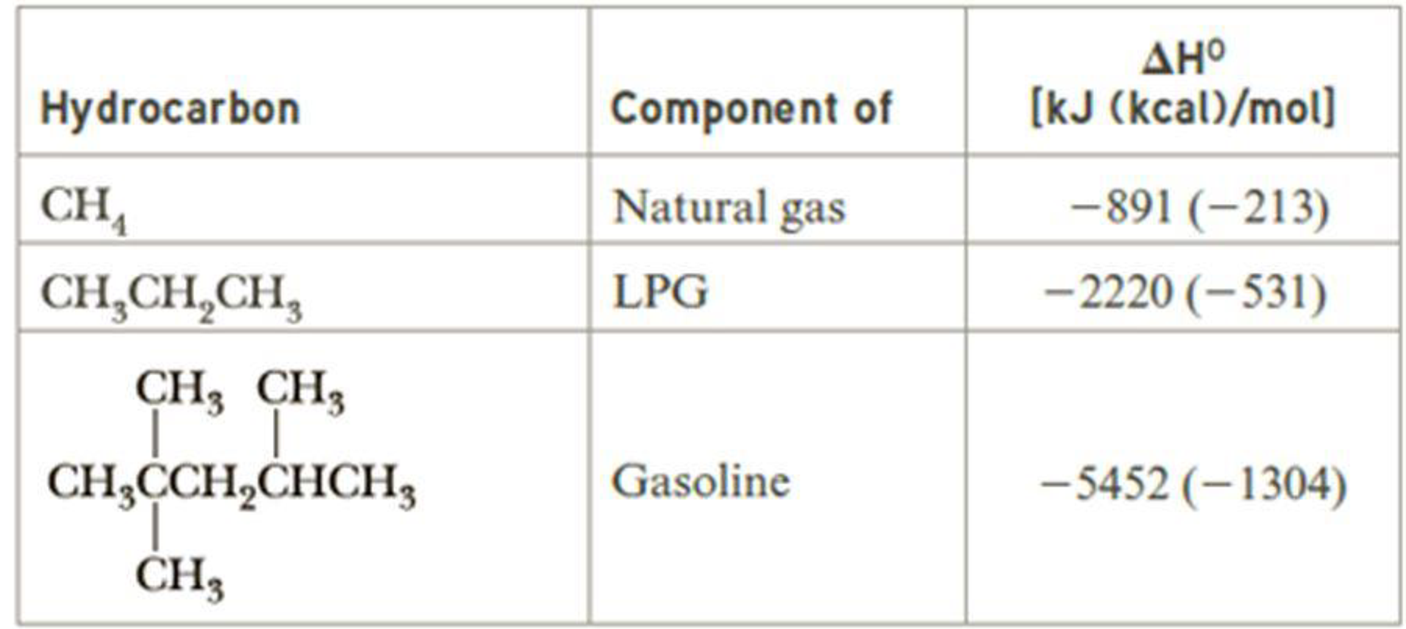
Following are heats of combustion per mole for methane, propane, and 2,2,4-trimeth-ylpentane. Each is a major source of energy. On a gram-for-gram basis, which of these hydrocarbons is the best source of heat energy?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
9. OA. Rank the expected boiling points of the compounds shown below from highest to lowest. Place your answer
appropriately in the box. Only the answer in the box will be graded. (3) points)
OH
OH
بر بد بدید
2
3
There is an instrument in Johnson 334 that measures total-reflectance x-ray fluorescence (TXRF) to do elemental analysis (i.e., determine what elements are present in a sample). A researcher is preparing a to measure calcium content in a series of well water samples by TXRF with an internal standard of vanadium (atomic symbol: V). She has prepared a series of standard solutions to ensure a linear instrument response over the expected Ca concentration range of 40-80 ppm. The concentrations of Ca and V (ppm) and the instrument response (peak area, arbitrary units) are shown below. Also included is a sample spectrum. Equation 1 describes the response factor, K, relating the analyte signal (SA) and the standard signal (SIS) to their respective concentrations (CA and CIS).
Ca, ppm
V, ppm
SCa, arb. units
SV, arb. units
20.0
10.0
14375.11
14261.02
40.0
10.0
36182.15
17997.10
60.0
10.0
39275.74
12988.01
80.0
10.0
57530.75
14268.54
100.0…
A mixture of 0.568 M H₂O, 0.438 M Cl₂O, and 0.710 M HClO are enclosed in a vessel at 25 °C.
H₂O(g) + C₁₂O(g) = 2 HOCl(g)
K = 0.0900 at 25°C
с
Calculate the equilibrium concentrations of each gas at 25 °C.
[H₂O]=
[C₁₂O]=
[HOCI]=
M
Σ
M
Chapter 2 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 2.2 - Do the line-angle formulas in each pair represent...Ch. 2.2 - Draw line-angle formulas for the three...Ch. 2.3 - Write IUPAC names for these alkanes.Ch. 2.4 - Combine the proper prefix, infix, and suffix and...Ch. 2.4 - Write the molecular formula, IUPAC name, and...Ch. 2.4 - Write molecular formulas for each bicycloalkane,...Ch. 2.4 - Prob. 2.7PCh. 2.5 - For 1,2-dichloroethane: (a) Draw Newman...Ch. 2.5 - Following is a chair conformation of cyclohexane...Ch. 2.5 - Draw the alternative chair conformation for the...
Ch. 2.5 - Draw a chair conformation of...Ch. 2.6 - Which cycloalkanes show cis, trans isomerism? For...Ch. 2.6 - Following is a planar hexagon representation for...Ch. 2.6 - Here is one cis,trans isomer of...Ch. 2.6 - Prob. AQCh. 2.6 - Prob. BQCh. 2.6 - Prob. CQCh. 2.7 - Arrange the alkanes in each set in order of...Ch. 2 - Write a line-angle formula for each condensed...Ch. 2 - Write the molecular formula of each alkane.Ch. 2 - Using parentheses and subscripts, provide an even...Ch. 2 - Which statements are true about constitutional...Ch. 2 - Prob. 2.20PCh. 2 - Each member of the following set of compounds is...Ch. 2 - Each of the following compounds is an amine...Ch. 2 - Each of the following compounds is either an...Ch. 2 - Draw structural formulas and write IUPAC names for...Ch. 2 - Draw structural formulas for all of the following....Ch. 2 - Write IUPAC names for these alkanes and...Ch. 2 - Write structural formulas and line-angle formulas...Ch. 2 - Explain why each is an incorrect IUPAC name and...Ch. 2 - For each IUPAC name, draw the corresponding...Ch. 2 - Write the IUPAC name for each compound.Ch. 2 - Prob. 2.31PCh. 2 - Torsional strain resulting from eclipsed CH bonds...Ch. 2 - How many different staggered conformations are...Ch. 2 - Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw a...Ch. 2 - Consider 1-bromo-2-methylpropane and draw the...Ch. 2 - trans-1,4-Di-tert-butylcyclohexane exists in a...Ch. 2 - From studies of the dipole moment of...Ch. 2 - Prob. 2.38PCh. 2 - Following are the alternative chair conformations...Ch. 2 - Prob. 2.40PCh. 2 - Prob. 2.41PCh. 2 - Draw line-angle formulas for the cis and trans...Ch. 2 - Name and draw structural formulas for all...Ch. 2 - Using a planar pentagon representation for the...Ch. 2 - Gibbs free energy differences between...Ch. 2 - Prob. 2.46PCh. 2 - Calculate the difference in Gibbs free energy in...Ch. 2 - Draw the alternative chair conformations for the...Ch. 2 - Use your answers from Problem 2.48 to complete the...Ch. 2 - There are four cis,trans isomers of...Ch. 2 - Draw alternative chair conformations for each...Ch. 2 - 1,2,3,4,5,6-Hexachlorocyclohexane shows cis,trans...Ch. 2 - Prob. 2.53PCh. 2 - What generalization can you make about the...Ch. 2 - What unbranched alkane has about the same boiling...Ch. 2 - Complete and balance the following combustion...Ch. 2 - Following are heats of combustion per mole for...Ch. 2 - Following are structural formulas and heats of...Ch. 2 - Without consulting tables, arrange these compounds...Ch. 2 - Which would you predict to have the larger (more...Ch. 2 - Following are structural formulas for 1,4-dioxane...Ch. 2 - Following is a planar hexagon representation of...Ch. 2 - On the left is a stereorepresentation of glucose...Ch. 2 - Prob. 2.64PCh. 2 - Prob. 2.65P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What units (if any) does the response factor (K) have? Does the response factor (K) depend upon how the concentration is expressed (e.g. molarity, ppm, ppb, etc.)?arrow_forwardProvide the structure, circle or draw, of the monomeric unit found in the biological polymeric materials given below. HO OH amylose OH OH 행 3 HO cellulose OH OH OH Ho HOarrow_forwardWhat units (if any) does K have? Does K depend upon how the concentration is expressed (e.g. molarity, ppm, ppb, etc.)? in calculating the response factorarrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forwardDon't used Ai solution and don't used hand raitingarrow_forwardOA. For the structure shown, rank the bond lengths (labeled a, b and c) from shortest to longest. Place your answer in the box. Only the answer in the box will be graded. (2 points) H -CH3 THe b Нarrow_forward
- Don't used hand raitingarrow_forwardQuizzes - Gen Organic & Biological Che... ☆ myd21.lcc.edu + O G screenshot on mac - Google Search savings hulu youtube google disney+ HBO zlib Homework Hel...s | bartleby cell bio book Yuzu Reader: Chemistry G periodic table - Google Search b Home | bartleby 0:33:26 remaining CHEM 120 Chapter 5_Quiz 3 Page 1: 1 > 2 > 3 > 6 ¦ 5 > 4 > 7 ¦ 1 1 10 8 ¦ 9 a ¦ -- Quiz Information silicon-27 A doctor gives a patient 0.01 mC i of beta radiation. How many beta particles would the patient receive in I minute? (1 Ci = 3.7 x 10 10 d/s) Question 5 (1 point) Saved Listen 2.22 x 107 222 x 108 3.7 x 108 2.22 x 108 none of the above Question 6 (1 point) Listen The recommended dosage of 1-131 for a test is 4.2 μCi per kg of body mass. How many millicuries should be given to a 55 kg patient? (1 mCi = 1000 μСi)? 230 mCiarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forwardQ3: Arrange each group of compounds from fastest SN2 reaction rate to slowest SN2 reaction rate. CI Cl H3C-Cl CI a) A B C D Br Br b) A B C Br H3C-Br Darrow_forwardQ4: Rank the relative nucleophilicity of halide ions in water solution and DMF solution, respectively. F CI Br | Q5: Determine which of the substrates will and will not react with NaSCH3 in an SN2 reaction to have a reasonable yield of product. NH2 Br Br Br .OH Brarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License