
Concept explainers
(a)
Interpretation:
Given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution

The electrostatic potential map shows the molecule with a negative charge concentrated at the center, with positive charge distributed symmetrically around the center. This shows that the individual bond dipoles are all of equal magnitude, and they all point toward the center. The vector addition of these dipoles will be zero because of their symmetric distribution. Therefore, the molecule is nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(b)
Interpretation:
Given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution
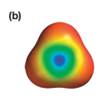
The electrostatic potential map shows a molecule with a negative charge concentrated at the center, with positive charge distributed symmetrically around the center. This shows that the individual bond dipoles are all of equal magnitude, and they all point toward the center. The vector addition of these dipoles will be zero because of their symmetric distribution. Therefore, the molecule is nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(c)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
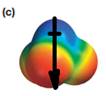
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is downward as shown below.
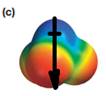
Explanation of Solution
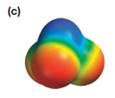
The electrostatic potential map shows a molecule with an asymmetric charge distribution. The positive charge is concentrated on the atom at the top, while the negative charge is distributed on three atoms at the bottom. The individual bond dipoles will therefore not cancel completely. Therefore, the molecule is polar.
The direction of the molecular dipole will be downward, as shown below by the black arrow.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(d)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
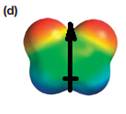
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is upward, as shown below.
Explanation of Solution
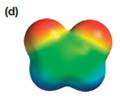
The electrostatic potential map shows a molecule with a negative charge distributed on two atoms at the top and the positive charge distributed on two atoms at the bottom. This shows that the individual bond dipoles both point approximately upward and slightly away from the center line. The vector addition of these dipoles will be nonzero. Therefore, the molecule is polar.
The direction of the net dipole moment will be upward as shown below.
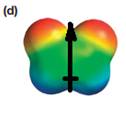
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(e)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
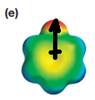
The electrostatic potential map shows that the molecule is polar.
The direction of the net molecular dipole is upward as shown below.
Explanation of Solution
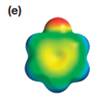
The electrostatic potential map shows a molecule with a negative charge concentrated on the atom at the top center, with positive charge distributed over atoms on the side and at the bottom. This shows that the individual bond dipoles are not symmetrically distributed and will not cancel out completely. Therefore, the molecule is polar.
The direction of the net molecular dipole will be upward because of the concentration of negative charge at the top and a symmetrical distribution of the positive charge in the rest of the molecule.
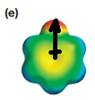
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
(f)
Interpretation:
The given uncharged molecule is to be identified as polar or nonpolar, using its electrostatic potential map. If the molecule is polar, the direction of its net molecular dipole moment it to be determined.
Concept introduction:
A polar bond is one in which the bond pair is unequally shared by the two atoms.
A partial positive charge is developed on the less electronegative atom while and an equal but negative partial charge is developed on the more electronegative atom.
Molecules that contain more than one polar bond may or may not have a net dipole moment. The dipole moment is a vector quantity. The net molecular dipole moment is the result of the vector addition of all the individual dipole moments. Depending on the symmetry of the molecule, the individual bond dipoles can partly or completely cancel or reinforce each other.
A bond dipole or a molecular dipole is represented by an arrow pointing from the atom or region with a partial positive charge toward an atom or region with a partial negative charge.
Electrostatic potential maps of molecules show the distribution of electron density in different parts of the molecule. The electron density is represented by different colors, ranging from blue to red. Blue color indicates a low electron density, an atom or region with a partial positive charge. Red color indicates high electron density, an atom or region with a partial negative charge.
Answer to Problem 2.40P
The electrostatic potential map shows that the molecule is nonpolar.
Explanation of Solution
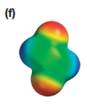
The electrostatic potential map shows a charge distribution that is symmetric about the center of the molecule with two negative regions opposite each other across the center as well as two positive regions across the center. The individual bond dipoles will therefore cancel out completely. Therefore, the net dipole moment will be zero, and the molecule will be nonpolar.
The net dipole moment of a molecule is the vector sum of the individual bond dipoles.
Want to see more full solutions like this?
Chapter 2 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Please help me solve these problems. Thank you in advance.arrow_forwardPredict the products of this organic reaction: O N IN A N + H2O + HCI ? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. 田 C + Explanation Check Click and drag to start drawing a structure. C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward6. For each of the following, fill in the synthesis arrows with reagents and show the intermediates. You DO NOT need to use the same number of arrows that are shown (you may use more or less), but the product must be formed from the reactant. Then write the mechanism of one step in the synthesis (you can choose which step to write the mechanism for), including all reagents required, clearly labeling the nucleophile and electrophile for each step, and using curved arrows to show the steps in the mechanism. a. b. OHarrow_forward
- Draw the productsarrow_forwardDraw the correct productsarrow_forwardE Organic Chemistry Maxwell Draw the correct products, in either order, for the ozonolysis reaction: 1) O3, CH2Cl2, -78 °C Product 1 + Product 2 2) Zn, HOAc Draw product 1. Select Draw Templates More C H O presented by M Draw product 2. Erase Select Draw Templates M / # # carrow_forward
- ✓ edict the products of this organic reaction: ---- ။ A CH3–C−NH–CH2–C−CH3 + KOH ? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. C 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibiliarrow_forwardPredict the product of this organic reaction: A HO-C-CH3 + CH3NH2 P+ H2O Specifically, in the drawing area below draw the condensed structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. marrow_forwardH 1) OsO4, pyridine 2) Na2SO3 or NaHSO3 in H₂O 2 productsarrow_forward
- ● Biological Macromolecules Naming and drawing cyclic monosaccharides Your answer is incorrect. • Row 1: Your answer is incorrect. Row 3: Your answer is incorrect. • Row 4: Your answer is incorrect. Try again... 0/5 Give the complete common name, including anomer and stereochemistry labels, of the following molecules. You will find helpful information in the ALEKS resource. CH2OH OH OH H H I H OH OH H] H CH2OH H OH ẞ-L-sorbose HOCH2 OH OH H HOCH2 H OH OH H OH H H CH2OH OH H H OH H I- H OH H OH Explanation Recheck W E R % 25 α B Y X & 5 D F G H McGraw Hill LLC. All Rights Reserved. Terms of Use | Pr Parrow_forwardWhat is the missing reactant in this organic reaction? + R -A HO IN + H₂O Specifically, in the drawing area below draw the skeletal ("line") structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. Explanation Check Click and drag to start drawing a structure. © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forwardStuc X ctclix ALE X A ALE אן A ALEX Lab (195 X Nut x M Inb x NU X NUT X Unt x + → C www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-lgNslkr7j8P3jH-IQ1g8NUi-mObKa_ZLx2twjEhK7mVG6PulJI006NcKTV37JxMpZuyrVCdQolLAKqp_7U3r1GUD3... New Chrome available: Naomi Question 26 of 39 (4 points) | Question Attempt: 1 of Unlimited Give the IUPAC name. 2,3-dimethylhexane Part: 1/2 Part 2 of 2 Draw the skeletal structure of a constitutional isomer of the alkane above that contains a different number of carbons in its longest chain. Skip Part Check Click and drag to start drawing a structure. 3 Finance headline Q Search mwa Harvard Intensifi... X Save For Later 00 dlo HB Submit Assignment 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility a 9:11 PM 4/22/2025arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
