
General Chemistry: Atoms First
2nd Edition
ISBN: 9780321809261
Author: John E. McMurry, Robert C. Fay
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 2.27CP
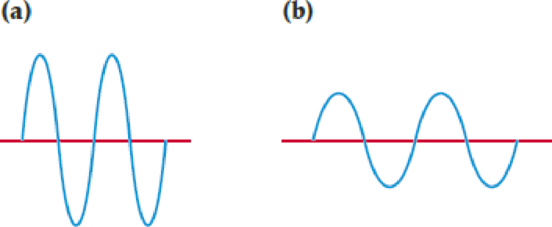
Two
- (a) Which wave has the greater intensity?
- (b) Which wave corresponds to higher-energy radiation?
- (c) Which wave represents yellow light, and which represents infrared radiation?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In the electrode Pt, H2(1 atm) | H+(a=1), if the electrode balance potential is -0.118 V and the interface potential difference is +5 mV. The current voltage will be 0.005 - (-0.118) = 0.123 V ¿Correcto?
In the electrode Pt, H2(1 atm) | H+(a=1) at 298K is 0.79 mA cm-2. If the balance potential of the electrode is -0.118 V and the potential difference of the interface is +5 mV. Determine its potential.
In one electrode: Pt, H2(1 atm) | H+(a=1), the interchange current density at 298K is 0.79 mA·cm-2. If the voltage difference of the interface is +5 mV. What will be the correct intensity at pH = 2?. Maximum transfer voltage and beta = 0.5.
Chapter 2 Solutions
General Chemistry: Atoms First
Ch. 2.1 - What is the frequency of a gamma ray with = 3.56 ...Ch. 2.1 - What is the wavelength in meters of an FM radio...Ch. 2.1 - Two electromagnetic waves are represented below....Ch. 2.2 - The Balmer equation can be extended beyond the...Ch. 2.2 - What is the longest-wavelength line in nanometers...Ch. 2.2 - What is the shortest-wavelength line in nanometers...Ch. 2.3 - What is the energy in kilojoules per mole of...Ch. 2.3 - The biological effects of a given dose of radiant...Ch. 2.3 - The work function of zinc metal is 350 kJ/mol....Ch. 2.3 - What is the work function of nickel metal if light...
Ch. 2.4 - What is the de Broglie wavelength in meters of a...Ch. 2.6 - Extend Table 2.2 to show allowed combinations of...Ch. 2.6 - Give orbital notations for electrons in orbitals...Ch. 2.6 - Give the allowed combinations of three quantum...Ch. 2.7 - Prob. 2.15PCh. 2.7 - Give a possible combination of n and l quantum...Ch. 2.9 - Calculate in kilojoules per mole the energy...Ch. 2.12 - Look at the electron configurations in Figure...Ch. 2.13 - Give expected ground-state electron configurations...Ch. 2.13 - Take a guess. What do you think is a likely...Ch. 2.13 - Identify the atom with the following ground-state...Ch. 2.14 - Which atom in each of the following pairs would...Ch. 2.14 - Prob. 2.23PCh. 2 - Where on the blank outline of the periodic table...Ch. 2 - Where on the periodic table do elements that meet...Ch. 2 - One of the elements shown on the following...Ch. 2 - Two electromagnetic waves are represented below....Ch. 2 - Prob. 2.28CPCh. 2 - The following orbital-filling diagram represents...Ch. 2 - Prob. 2.30CPCh. 2 - Identify each of the following orbitals, and give...Ch. 2 - Which has the higher frequency, red light or...Ch. 2 - Prob. 2.33SPCh. 2 - The Hubble Space Telescope detects radiant energy...Ch. 2 - Prob. 2.35SPCh. 2 - What is the wavelength in meters of ultraviolet...Ch. 2 - Prob. 2.37SPCh. 2 - Calculate the energies of the following waves in...Ch. 2 - The MRI (magnetic resonance imaging) body scanners...Ch. 2 - A certain cellular telephone transmits at a...Ch. 2 - Prob. 2.41SPCh. 2 - What is the wavelength in meters of photons with...Ch. 2 - What is the energy of each of the following...Ch. 2 - The data encoded on CDs, DVDs, and Blu-ray discs...Ch. 2 - The semimetal germanium is used as a component in...Ch. 2 - Prob. 2.46SPCh. 2 - According to the values cited in Problem 2.46, the...Ch. 2 - The work function of silver metal is 436 kJ/mol....Ch. 2 - What is the work function of gold metal in kJ/mol...Ch. 2 - Protons and electrons can be given very high...Ch. 2 - If a proton (mass = 1.673 1024 g) is accelerated...Ch. 2 - What is the de Broglie wavelength in meters of a...Ch. 2 - What is the de Broglie wavelength in meters of a...Ch. 2 - At what speed in meters per second must a 145 g...Ch. 2 - What velocity would an electron (mass = 9.11 1031...Ch. 2 - Use the Heisenberg uncertainty principle to...Ch. 2 - The mass of a helium-4 atom is 4.0026 u, and its...Ch. 2 - What are the four quantum numbers, and what does...Ch. 2 - What is the Heisenberg uncertainty principle, and...Ch. 2 - Why do we have to use an arbitrary value such as...Ch. 2 - How many nodal surfaces does a 4s orbital have?...Ch. 2 - Prob. 2.62SPCh. 2 - How does electron shielding in multielectron atoms...Ch. 2 - Give the allowable combinations of quantum numbers...Ch. 2 - Give the orbital designations of electrons with...Ch. 2 - Tell which of the following combinations of...Ch. 2 - Which of the following combinations of quantum...Ch. 2 - What is the maximum number of electrons in an atom...Ch. 2 - What is the maximum number of electrons in an atom...Ch. 2 - The wavelength of light at which the Balmer series...Ch. 2 - One series of lines of the hydrogen spectrum is...Ch. 2 - Prob. 2.72SPCh. 2 - Excited rubidium atoms emit red light with = 795...Ch. 2 - Why does the number of elements in successive...Ch. 2 - Prob. 2.75SPCh. 2 - Prob. 2.76SPCh. 2 - Prob. 2.77SPCh. 2 - According to the aufbau principle, which orbital...Ch. 2 - Prob. 2.79SPCh. 2 - Prob. 2.80SPCh. 2 - Give the expected ground-state electron...Ch. 2 - Prob. 2.82SPCh. 2 - Draw orbital-filling diagrams for atoms with the...Ch. 2 - Prob. 2.84SPCh. 2 - Prob. 2.85SPCh. 2 - Prob. 2.86SPCh. 2 - Prob. 2.87SPCh. 2 - Prob. 2.88SPCh. 2 - Prob. 2.89SPCh. 2 - Prob. 2.90SPCh. 2 - Prob. 2.91SPCh. 2 - Prob. 2.92SPCh. 2 - Which atom in each of the following pairs has a...Ch. 2 - Prob. 2.94SPCh. 2 - Prob. 2.95SPCh. 2 - What is the expected ground-state electron...Ch. 2 - Prob. 2.97SPCh. 2 - Orbital energies in single-electron atoms or ions,...Ch. 2 - Prob. 2.99CHPCh. 2 - Prob. 2.100CHPCh. 2 - Prob. 2.101CHPCh. 2 - Prob. 2.102CHPCh. 2 - What is the wavelength in meters of photons with...Ch. 2 - Prob. 2.104CHPCh. 2 - The second in the SI system is defined as the...Ch. 2 - Prob. 2.106CHPCh. 2 - Prob. 2.107CHPCh. 2 - Cesium metal is often used in photoelectric...Ch. 2 - Prob. 2.109CHPCh. 2 - Prob. 2.110CHPCh. 2 - Prob. 2.111CHPCh. 2 - Youre probably familiar with using Scotch Tape for...Ch. 2 - Hard wintergreen-flavored candies are...Ch. 2 - Prob. 2.114CHPCh. 2 - Prob. 2.115CHPCh. 2 - Prob. 2.116CHPCh. 2 - Prob. 2.117CHPCh. 2 - Prob. 2.118CHPCh. 2 - X rays with a wavelength of 1.54 1010 m are...Ch. 2 - Prob. 2.120CHPCh. 2 - Assume that the rules for quantum numbers are...Ch. 2 - Given the subshells 1s, 2s, 2p, 3s, 3p, and 3d,...Ch. 2 - Prob. 2.123CHPCh. 2 - A minimum energy of 7.21 1019 J is required to...Ch. 2 - A photon produced by an X-ray machine has an...Ch. 2 - An energetically excited hydrogen atom has its...Ch. 2 - Prob. 2.127MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In a Pt electrode, H2(1 atm) | H+(a=1), the interchange current density of an electrode is 0.79 mA cm-2. ¿Qué corriente flow across the electrode of área 5 cm2 when the difference in potential of the interface is +5 mV?.arrow_forwardIf the current voltage is n = 0.14 V, indicate which of the 2 voltage formulas of the ley of Tafel must be applied i a a) == exp (1-B). xp[(1 - ß³): Fn Fn a b) == exp B RT RTarrow_forwardIf the current voltage is n = 0.14 V. Indicate which of the 2 formulas must be applied a) = a T = i exp[(1 - p) F Fn Fn b) i==exp B RTarrow_forward
- Topic: Photochemistry and Photophysics of Supramoleculesarrow_forwardTwo cations that exchange an electron in an interface, the exchange density is worth 1.39 mA/cm2 and the current density is worth 15 mA/cm2 at 25°C. If the overvoltage is 0.14 V, calculate the reaction rate and symmetry factor. Data: R = 8,314 J mol-1 k-1: F = 96500 Carrow_forwardWith the help of the Tafel line, it is estimated that the interchange density of the VO2+/VO2+ system on the carbon paper has a value of 3 mA cm-2. Calculate a) the current density if the voltage has a value of 1.6 mV and the temperature is 25°C. b) the beta value of the anódico process if the Tafel pendulum is 0.6 V at 25°C. Data: R = 8.314 JK-1mol-1, y F = 96485 C mol-1.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY