
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
10th Edition
ISBN: 9781305957510
Author: ZUMDAHL, Steven S.; Zumdahl, Susan A.; DeCoste, Donald J.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 125CP
Each of the following statements is true, but Dalton might have had trouble explaining some of them with his atomic theory. Give explanations for the following statements.
- a. The space-filling models for ethyl alcohol and dimethyl ether are shown below.
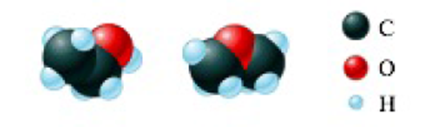
These two compounds have die same composition by mass (52% carbon, 13% hydrogen, and 35% oxygen), yet the two have different melting points, boiling points, and solubilities in water.
- b. Burning wood leaves an ash that is only a small fraction of the mass of the original wood.
- c. Atoms can be broken down into smaller particles.
- d. One sample of lithium hydride is 87.4% lithium by mass, while another sample of lithium hydride Ls 74.9% lithium by mass. However, the two samples have the same chemical properties.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
pressure (atm)
3
The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the
temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes.
0
0
200
temperature (K)
400
а
er your payment details | bar xb Home | bartleby
x +
aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1
O States of Matter
Sketching a described thermodynamic change on a phase diagram
0/5
The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the
temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes.
pressure (atm)
1
3-
0-
0
200
Explanation
Check
temperature (K)
400
X
Q Search
L
G
2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Ce
5.
Chapter 2 Solutions
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
Ch. 2 - Use Daltons atomic theory to account for each of...Ch. 2 - What evidence led to the conclusion that cathode...Ch. 2 - What discoveries were made by J. J. Thomson, Henri...Ch. 2 - Consider Ernest Rutherfords -particle bombardment...Ch. 2 - Do the proton and the neutron have exactly the...Ch. 2 - What is the distinction between atomic number and...Ch. 2 - Distinguish between the terms family and period in...Ch. 2 - The compounds AlCl3, CrCl3, and ICl3 have similar...Ch. 2 - Prob. 9RQCh. 2 - How would you name HBrO4, KIO3, NaBrO2, and HIO?...
Ch. 2 - Which of the following is true about an individual...Ch. 2 - How would you go about finding the number of chalk...Ch. 2 - These questions concern the work of J. J. Thomson....Ch. 2 - Prob. 4ALQCh. 2 - You have a chemical in a sealed glass container...Ch. 2 - The formula of water is If-O. Which of the...Ch. 2 - You may have noticed that when water boils, you...Ch. 2 - One of the best indications of a useful theory is...Ch. 2 - Prob. 9ALQCh. 2 - Label each of the following as an atomic element,...Ch. 2 - Why is the term sodium chloride molecule incorrect...Ch. 2 - Prob. 12ALQCh. 2 - Label each of the following as an atomic element,...Ch. 2 - Prob. 14ALQCh. 2 - Prob. 15ALQCh. 2 - Prob. 16ALQCh. 2 - Which of tire following explain how an ion is...Ch. 2 - What refinements had to be made in Daltons atomic...Ch. 2 - When hydrogen is burned in oxygen to form water,...Ch. 2 - The two most reactive families of elements are the...Ch. 2 - Explain the law of conservation of mass, the law...Ch. 2 - Section 2.3 describes the postulates of Daltons...Ch. 2 - The contributions of J. J. Thomson and Ernest...Ch. 2 - Prob. 24QCh. 2 - The number of protons in an atom determines the...Ch. 2 - If the volume of a proton were similar to the...Ch. 2 - Prob. 27QCh. 2 - List some characteristic properties that...Ch. 2 - Consider the elements of Group 4A (the carbon...Ch. 2 - Chlorine has two natural isotopes: 1737Cl and...Ch. 2 - Before an electrocardiogram (ECG) is recorded for...Ch. 2 - Distinguish between the following terms. a....Ch. 2 - Label the type of bonding for each of the...Ch. 2 - The vitamin niacin (nicotinic acid. C6H5NO2) can...Ch. 2 - Prob. 35QCh. 2 - Prob. 36QCh. 2 - When mixtures of gaseous H2 and gaseous Cl2 react,...Ch. 2 - Observations of the reaction between nitrogen gas...Ch. 2 - A sample of chloroform is found to contain 12.0 g...Ch. 2 - A sample of H2SO4 contains 2.02 g of hydrogen,...Ch. 2 - Consider 80.0-g samples of two different compounds...Ch. 2 - Several compounds containing sulfur and fluorine...Ch. 2 - The three most stable oxides of carbon ire carbon...Ch. 2 - Two elements. R and Q, combine to form two binary...Ch. 2 - In Section 1.1 of the text, the concept of a...Ch. 2 - In a combustion reaction, 46.0 g of ethanol reacts...Ch. 2 - Early tables of atomic weights (masses) were...Ch. 2 - Indium oxide contains 4.784 g of indium for every...Ch. 2 - Prob. 49ECh. 2 - If you wanted to make an accurate scale model of...Ch. 2 - In an experiment it was found that the total...Ch. 2 - A chemist in a galaxy tar, far away performed the...Ch. 2 - What are the symbols of the following nonmetals:...Ch. 2 - Prob. 55ECh. 2 - In the periodic table, how many elements are found...Ch. 2 - a. Classify the following elements as metals or...Ch. 2 - a. List the noble gas elements. Which of the noble...Ch. 2 - For each of the following sets of elements, label...Ch. 2 - Prob. 60ECh. 2 - Prob. 61ECh. 2 - Write the atomic symbol (ZAX) for each of the...Ch. 2 - Write the symbol of each atom using the ZAX...Ch. 2 - For carbon-14 and carbon-12, how many protons and...Ch. 2 - How many protons and neutrons are in the nucleus...Ch. 2 - Prob. 66ECh. 2 - For each of the following ions, indicate the...Ch. 2 - How many protons, neutrons, and electrons are in...Ch. 2 - Prob. 69ECh. 2 - What is the symbol of an ion with 16 protons, 18...Ch. 2 - Complete the following table: Symbol Number of...Ch. 2 - Complete the following table: Symbol Number of...Ch. 2 - Would you expect each of the following atoms to...Ch. 2 - Prob. 74ECh. 2 - Name the compounds in parts ad and write the...Ch. 2 - Name the compounds in parts a-d and write the...Ch. 2 - Prob. 77ECh. 2 - Prob. 78ECh. 2 - Prob. 79ECh. 2 - Prob. 80ECh. 2 - Prob. 81ECh. 2 - Prob. 82ECh. 2 - Prob. 83ECh. 2 - Prob. 84ECh. 2 - Prob. 85ECh. 2 - Prob. 86ECh. 2 - Prob. 87ECh. 2 - Write the formula for each of the following...Ch. 2 - Prob. 89ECh. 2 - Write the formula for each of the following...Ch. 2 - Prob. 91ECh. 2 - Each of the following compounds is incorrectly...Ch. 2 - Insulin is a complex protein molecule produced by...Ch. 2 - Carbohydrates, a class of compounds containing the...Ch. 2 - Prob. 95AECh. 2 - What are the symbols for the following nonmetal...Ch. 2 - Four Fe2+ ions are key components of hemoglobin,...Ch. 2 - Which of the following statements is(are) true?...Ch. 2 - The isotope of an unknown element, X, has a mass...Ch. 2 - Prob. 100AECh. 2 - Prob. 101AECh. 2 - Identify each of the following elements: a. a...Ch. 2 - Prob. 103AECh. 2 - Prob. 104AECh. 2 - Prob. 105AECh. 2 - Prob. 106AECh. 2 - Prob. 107AECh. 2 - Prob. 108AECh. 2 - Consider 100.0-g samples of two different...Ch. 2 - Give the systematic name for the following...Ch. 2 - Prob. 111CWPCh. 2 - Prob. 112CWPCh. 2 - Complete the following table. Atom/Ion Protons...Ch. 2 - Which of the following is{are) correct? a. 40Ca2...Ch. 2 - Prob. 115CWPCh. 2 - Prob. 116CWPCh. 2 - Prob. 117CWPCh. 2 - Prob. 118CWPCh. 2 - Reaction of 2.0 L of hydrogen gas with 1.0 L of...Ch. 2 - A combustion reaction involves the reaction of a...Ch. 2 - A chemistry instructor makes the following claim:...Ch. 2 - The early alchemists used to do an experiment in...Ch. 2 - Consider the chemical reaction as depicted below....Ch. 2 - Each of the following statements is true, but...Ch. 2 - You have two distinct gaseous compounds made from...Ch. 2 - A single molecule has a mass of 7.31 1023 g....Ch. 2 - You take three compounds, each consisting of two...Ch. 2 - Prob. 129IPCh. 2 - Prob. 130IPCh. 2 - Using the information in Table 2.1, answer the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6.arrow_forward0/5 alekscgi/x/sl.exe/1o_u-IgNglkr7j8P3jH-IQs_pBaHhvlTCeeBZbufuBYTi0Hz7m7D3ZcSLEFovsXaorzoFtUs | AbtAURtkqzol 1HRAS286, O States of Matter Sketching a described thermodynamic change on a phase diagram The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 3 pressure (atm) + 0- 0 5+ 200 temperature (K) 400 Explanation Check X 0+ F3 F4 F5 F6 F7 S 2025 McGraw Hill LLC All Rights Reserved. Terms of Use Privacy Center Accessibility Q Search LUCR + F8 F9 F10 F11 F12 * % & ( 5 6 7 8 9 Y'S Dele Insert PrtSc + Backsarrow_forward5.arrow_forward
- 9arrow_forwardalekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZS18w-nDB10538ZsAtmorZoFusYj2Xu9b78gZo- O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 3- 200 temperature (K) Explanation Chick Q Sowncharrow_forward0+ aleksog/x/lsl.exe/1ou-lgNgkr7j8P3H-IQs pBaHhviTCeeBZbufuBYTOHz7m7D3ZStEPTBSB3u9bsp3Da pl19qomOXLhvWbH9wmXW5zm O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 Gab The temperature on a sample of pure X held at 0.75 atm and -229. °C is increased until the sample sublimes. The temperature is then held constant and the pressure is decreased by 0.50 atm. On the phase diagram below draw a path that shows this set of changes. F3 pressure (atm) 0- 0 200 Explanation temperature (K) Check F4 F5 ☀+ Q Search Chill Will an 9 ENG F6 F7 F8 F9 8 Delete F10 F11 F12 Insert PrtSc 114 d Ararrow_forward
- x + LEKS: Using a phase diagram a X n/alekscgi/x/lsl.exe/10_u-IgNsikr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpw ○ States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 1.1 atm. pressure (atm) 16 08- solid liquid- 0 200 400 gas 600 temperature (K) Note: your answer must be within 25 °C of the exact answer to be graded correct. × 5arrow_forwardS: Using a phase diagram leksogi/x/sl.exe/1ou-IgNs kr 7j8P3jH-IQs_pBan HhvTCeeBZbufuBYTI0Hz7m7D3ZdHYU+80XL-5alyVp O States of Matter Using a phase diagram to find a phase transition temperature or pressure se the phase diagram of Substance X below to find the boiling point of X when the pressure on the liquid is 1.6 atm. pressure (atm) 32- 16- solid liquid 0. gas 100 200 temperature (K) 300 Note: your answer must be within 12.5 °C of the exact answer to be graded correct. 10 Explanation Check § Q Search J 2025 McGraw Hill LLC. All Rights Researrow_forward151.2 254.8 85.9 199.6 241.4 87.6 242.5 186.4 155.8 257.1 242.9 253.3 256.0 216.6 108.7 239.0 149.7 236.4 152.1 222.7 148.7 278.2 268.7 234.4 262.7 283.2 143.6 QUESTION: Using this group of data on salt reduced tomato sauce concentration readings answer the following questions: 1. 95% Cl Confidence Interval (mmol/L) 2. [Na+] (mg/100 mL) 3. 95% Na+ Confidence Interval (mg/100 mL)arrow_forward
- Results Search Results Best Free Coursehero Unloc xb Success Confirmation of Q x O Google Pas alekscgi/x/lsl.exe/1o_u-IgNslkr 7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCavJ17dZtpxbFD0Qggd1J O States of Matter Using a phase diagram to find a phase transition temperature or pressure Gabr 3/5 he pressure above a pure sample of solid Substance X at 101. °C is lowered. At what pressure will the sample sublime? Use the phase diagram of X below to nd your answer. pressure (atm) 24- 12 solid liquid gas 200 400 temperature (K) 600 ote: your answer must be within 0.15 atm of the exact answer to be graded correct. atm Thanation Check © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center I Q Search L³ ملةarrow_forward301.7 348.9 193.7 308.6 339.5 160.6 337.7 464.7 223.5 370.5 326.6 327.5 336.1 317.9 203.8 329.8 221.9 331.7 211.7 309.6 223.4 353.7 334.6 305.6 340.0 304.3 244.7 QUESTION: Using this group of data on regular tomato sauce concentration readings answer the following questions: 1. 95% Cl Confidence Interval (mmol/L) 2. [Na+] (mg/100 mL) 3. 95% Na+ Confidence Interval (mg/100 mL)arrow_forwardSearch Results Search Results Best Free Coursehero Unlo x b Success Confirmation of Q aleks.com/alekscgi/x/sl.exe/10_u-lgNslkr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTIOHz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCav States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the temperature at which X turns to a gas, if the pressure above the solid is 3.7 atm. pressure (atm) 0. 32- 16 solid liquid gas 200 temperature (K) Note: your answer must be within 20 °C of the exact answer to be graded correct. Дос Xarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Atomic Number, Atomic Mass, and the Atomic Structure | How to Pass ChemistryThe Nucleus: Crash Course Chemistry #1; Author: Crash Course;https://www.youtube.com/watch?v=FSyAehMdpyI;License: Standard YouTube License, CC-BY