
Conceptual Physical Science (6th Edition)
6th Edition
ISBN: 9780134060491
Author: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 54E
One of the skin-irritating components of poison oak is tetrahvdrourushiol:
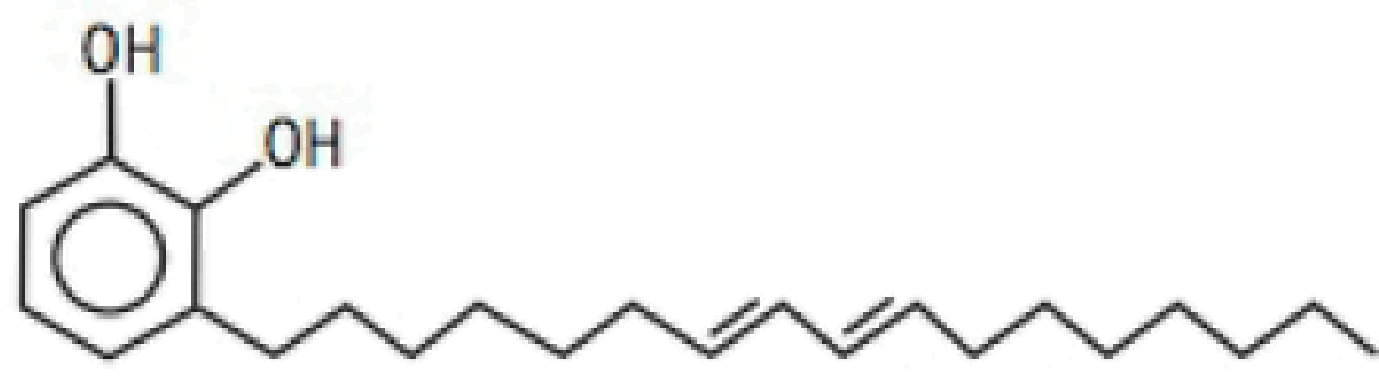
The long, nonpolar hydrocarbon tail embeds itself in a person's oily skin, where the molecule initiates an allergic response. Scratching the itch spreads tetrahvdrourushiol molecules over a greater surface area, causing the zone of irritation to grow. Is this compound an alcohol or a phenol? Defend your answer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Three point-like charges in the attached image are placed at the corners of an equilateral triangle as shown in the figure. Each side of the triangle has a length of 38.0 cm, and the point (C) is located half way between q1 and q3 along the side. Find the magnitude of the electric field at point (C). Let q1 = −2.80 µC, q2 = −3.40 µC, and q3 = −4.50 µC. Thank you.
Three point-like charges are placed as shown in the attach image, where r1 = r2 = 44.0 cm. Find the magnitude of the electric force exerted on the charge q3. Let q1 = -1.90 uC, q2 = -2.60 uC, and q3 = +3.60 uC. Thank you.
The drawing attached shows an edge-on view of two planar surfaces that intersect and are mutually perpendicular. Surface (1) has an area of 1.90 m², while Surface (2) has an area of 3.90 m². The electric field in magnitude of 215 N/C. Find the magnitude of the electric flux through surface (1 and 2 combined) if the angle theta made between the electric field with surface (2) is 30.0 degrees. Thank you.
Chapter 19 Solutions
Conceptual Physical Science (6th Edition)
Ch. 19 - How do two structural isomers differ from each...Ch. 19 - How are two structural isomers similar to each...Ch. 19 - What physical property of hydrocarbons is used in...Ch. 19 - What types of hydrocarbons are more abundant in...Ch. 19 - To how many atoms is a saturated carbon atom...Ch. 19 - What is the difference between a saturated...Ch. 19 - How many multiple bonds must a hydrocarbon have in...Ch. 19 - What kind of ring do aromatic compounds contain?Ch. 19 - What is a heteroatom?Ch. 19 - Why do heteroatoms make such a difference in the...
Ch. 19 - Why are low-formula-mass alcohols soluble in...Ch. 19 - What distinguishes an alcohol from a phenol?Ch. 19 - What distinguishes an alcohol from an ether?Ch. 19 - Which heteroatom is characteristic of an amine?Ch. 19 - Do amines tend to be acidic, neutral, or basic?Ch. 19 - Are alkaloids found in nature?Ch. 19 - What are some examples of alkaloids?Ch. 19 - Which elements make up the carbonyl group?Ch. 19 - How are ketones and aldehydes related to each...Ch. 19 - How are amides and carboxylic acids related to...Ch. 19 - From what naturally occurring compound is aspirin...Ch. 19 - What happens to the double bond of a monomer...Ch. 19 - What is released in the formation of a...Ch. 19 - Why is plastic wrap made of polyvinylidene...Ch. 19 - Prob. 25RCQCh. 19 - Rank the following molecules in order of the phase...Ch. 19 - Rank the following hydrocarbons in order of...Ch. 19 - Rank the following hydrocarbons in order of...Ch. 19 - Rank the following organic molecules in order of...Ch. 19 - Rank the following organic molecules in order of...Ch. 19 - What property of carbon allows for the formation...Ch. 19 - Why does the melting point of hydrocarbons...Ch. 19 - Draw all the structural isomers for hydrocarbons...Ch. 19 - How many structural isomers are shown here?Ch. 19 - According to Figure 19.3, which has the higher...Ch. 19 - The temperatures in a fractionating tower at an...Ch. 19 - Prob. 40ECh. 19 - Do heavier hydrocarbons tend to produce more or...Ch. 19 - What do these two structures have in common?Ch. 19 - What do the compounds cyclopropane and propene...Ch. 19 - What are the chemical formulas for the following...Ch. 19 - Prob. 45ECh. 19 - Prob. 46ECh. 19 - Identify the following functional groups in this...Ch. 19 - What must be added to a double bond to transform...Ch. 19 - What do phenols and carboxylic acids have in...Ch. 19 - What is the difference between a ketone and an...Ch. 19 - Prob. 51ECh. 19 - Prob. 52ECh. 19 - What is the percent volume of water in 80-proof...Ch. 19 - One of the skin-irritating components of poison...Ch. 19 - Cetyl alcohol, C16H34O, is a common ingredient of...Ch. 19 - A common inactive ingredient in products such as...Ch. 19 - A common inactive ingredient in products such as...Ch. 19 - The phosphoric acid salt of caffeine has the...Ch. 19 - Prob. 59ECh. 19 - In water, does the following molecule act as an...Ch. 19 - If you saw the label phenylephrine-HCl on a...Ch. 19 - The amino acid lysine is shown below. What...Ch. 19 - Prob. 63ECh. 19 - Suggest an explanation why aspirin has a sour...Ch. 19 - Benzaldehyde is a fragrant oil. If stored in an...Ch. 19 - What products are formed upon the reaction of...Ch. 19 - The disodium salt of ethylenediaminetetraacetic...Ch. 19 - Would you expect polypropylene to be more dense or...Ch. 19 - Hydrocarbons release a lot of energy when ignited....Ch. 19 - The polymer styrene-butadiene rubber (SBR), shown...Ch. 19 - Citral and camphor are both 10-carbon odoriferous...Ch. 19 - Many of the natural product molecules synthesized...Ch. 19 - The solvent diethyl ether can be mixed with water...Ch. 19 - Alkaloid salts are not very soluble in the organic...Ch. 19 - Why does the melting point of hydrocarbons...Ch. 19 - How many structural isomers are there for...Ch. 19 - Which contains more hydrogen atoms: a five-carbon...Ch. 19 - Prob. 4RATCh. 19 - Why might a high-formula-mass alcohol be insoluble...Ch. 19 - Alkaloid salts are not very soluble in the organic...Ch. 19 - Explain why caprylic acid, CH3(CH2)6 COOH,...Ch. 19 - How many oxygen atoms are bonded to the carbon of...Ch. 19 - One solution to the problem of our overflowing...Ch. 19 - Which would you expect to be more viscous: a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Compare and contrast the carbon, sulfur, and nitrogen cycles in terms of the physiologies of the organisms that...
Brock Biology of Microorganisms (15th Edition)
Two culture media were inoculated with four different bacteria. After incubation, the following results were ob...
Microbiology: An Introduction
All of the following processes are involved in the carbon cycle except: a. photosynthesis b. cell respiration c...
Human Biology: Concepts and Current Issues (8th Edition)
What is the mass of 225 mL of a liquid that has a density of 0.880 g/mL? a. 198 g b. 0.0039 g c. 0.198 g d. 0.2...
Introductory Chemistry (6th Edition)
What percentage of Earths land surface do glaciers presently cover? ____________
Applications and Investigations in Earth Science (9th Edition)
Which compound is more easily decarboxylated?
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A car driving at 27m/s veers to the left to avoid a deer in the road. The maneuver takes 2.0s and the direction of travel is altered by 20 degrees. What is the average acceleration during the constant speed maneuver? Do this in accordance with the example in the chapter.arrow_forwardNo No No Chatgpt pls will upvotearrow_forward2 C01: Physical Quantities, Units and Measurementscobris alinu zotinUD TRO Bendemeer Secondary School Secondary Three Express Physics Chpt 1: Physical Quantities, Unit and Measurements Assignment Name: Chen ShiMan loov neowled soria 25 ( 03 ) Class: 3 Respect 6 Date: 2025.01.22 1 Which group consists only of scalar quantities? ABCD A acceleration, moment and energy store distance, temperature and time length, velocity and current mass, force and speed B D. B Which diagram represents the resultant vector of P and Q? lehtele 시 bas siqpeq olarist of beau eldeo qirie-of-qi P A C -B qadmis rle mengaib priwollot erT S Quilons of qira ono mont aboog eed indicator yh from West eril to Inioqbim srij enisinoo MA (6) 08 bas 8A aldao ni nolent or animaleb.gniweb slepe eld 260 km/h D 1 D. e 51arrow_forward
- The figure gives the acceleration a versus time t for a particle moving along an x axis. The a-axis scale is set by as = 12.0 m/s². At t = -2.0 s, the particle's velocity is 11.0 m/s. What is its velocity at t = 6.0 s? a (m/s²) as -2 0 2 t(s) 4arrow_forwardTwo solid cylindrical rods AB and BC are welded together at B and loaded as shown. Knowing that the average normal stress must not exceed 150 MPa in either rod, determine the smallest allowable values of the diameters d₁ and d2. Take P= 85 kN. P 125 kN B 125 kN C 0.9 m 1.2 m The smallest allowable value of the diameter d₁ is The smallest allowable value of the diameter d₂ is mm. mm.arrow_forwardWestros, from Game of Thrones, has an area of approximately 6.73⋅106 miles26.73⋅106miles2. Convert the area of Westros to km2 where 1.00 mile = 1.609 km.arrow_forward
- a) What is the lenght of x? b) Findθ c) Find ϕarrow_forwardA surveyor measures the distance across a straight river by the following method: Starting directly across from a tree on the opposite bank, he walks x = 97.7 m along the riverbank to establish a baseline. Then he sights across to the tree. The angle from his baseline to the tree is θ = 33.0 °. How wide is the river?arrow_forwardA small turtle moves at a speed of 697. furlong/fortnight. Find the speed of the turtle in centimeters per second. Note that 1.00 furlong = 220. yards, 1.00 yard = 3.00 feet, 1.00 foot = 12.0 inches, 1.00 inch = 2.54 cm, and 1.00 fortnight = 14.0 days.arrow_forward
- The landmass of Sokovia lifted in the air in Avengers: Age of Ultron had a volume of about 1.98 km3. What volume is that in m3?arrow_forwardA fathom is a unit of length, usually reserved for measuring the depth of water. A fathom is exactly 6.00 ft in length. Take the distance from Earth to the Moon to be 252,000 miles, and use the given approximation to find the distance in fathoms. 1 mile = 5280 ft. (Answer in sig fig.)arrow_forwardNo chatgpt pls will upvotearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning Stars and GalaxiesPhysicsISBN:9781305120785Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Stars and GalaxiesPhysicsISBN:9781305120785Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning



Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Foundations of Astronomy (MindTap Course List)
Physics
ISBN:9781337399920
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning

Stars and Galaxies
Physics
ISBN:9781305120785
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY