
(a)
Interpretation:
Synthesis of the given target molecule from phenylmethanol (benzyl alcohol) is to be determined.
Concept introduction:
Acid is converted to ester group by esterification reaction with the help of a strong base like
Answer to Problem 19.75P
Synthesis of the target molecule from phenylmethanol (benzyl alcohol) is
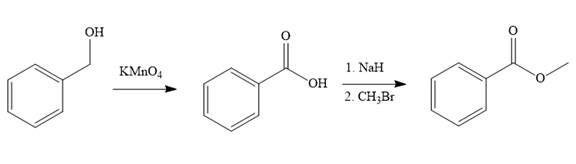
Explanation of Solution
Structure of the target molecule is
In the first step of the reaction, phenylmethanol (benzyl alcohol) is treated with oxidizing agent
In the second step of the reaction, benzoic acid undergoes esterification reaction. In this step, benzoic acid is first treated with a strong base like
The complete synthesis of the target molecule is as shown below.
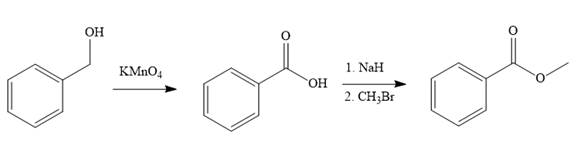
By using the
(b)
Interpretation:
Synthesis of the given target molecule from phenylmethanol (benzyl alcohol) is to be determined.
Concept introduction:
For the conversion of alcohol group to Grignard reagent, alcohol (bad leaving group) is treated with
When Grignard reagent is reacted with
For the removal of water, tertiary alcohol is treated with conc.
The bromination reaction on alkene takes place in presence of
Answer to Problem 19.75P
Synthesis of the target molecule from phenylmethanol (benzyl alcohol) is
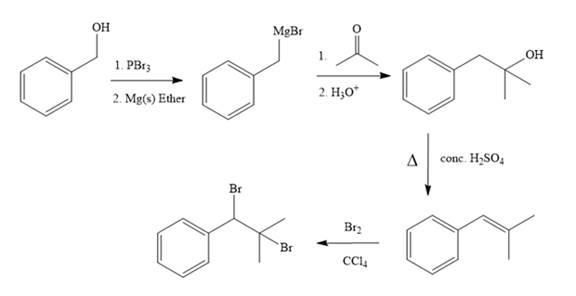
Explanation of Solution
Structure of the target molecule is
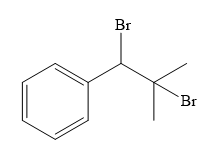
In the first step, the formation of Grignard reagent takes place. In this step, benzyl alcohol is treated with
The complete synthesis of the target molecule is as shown below.
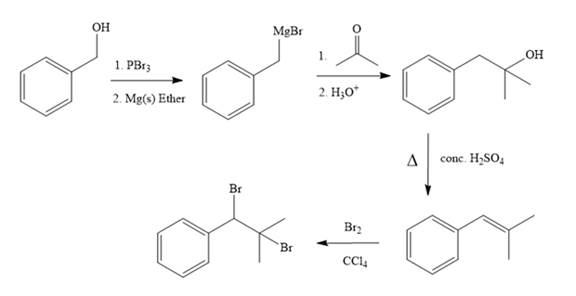
By using the functional group conversion reactions, the synthesis of the target molecule is determined.
(c)
Interpretation:
Synthesis of the given target molecule from phenylmethanol (benzyl alcohol) is to be determined.
Concept introduction:
Grignard reagent, in presence of acidic condition, is used for the conversion of aldehyde or ketone to alcohol.
For the removal of water, tertiary alcohol is treated with conc.
Reaction of alkene with diazomethane in presence of heat results in the formation of cyclopropane ring.
Answer to Problem 19.75P
Synthesis of the target molecule from phenylmethanol (benzyl alcohol) is
Explanation of Solution
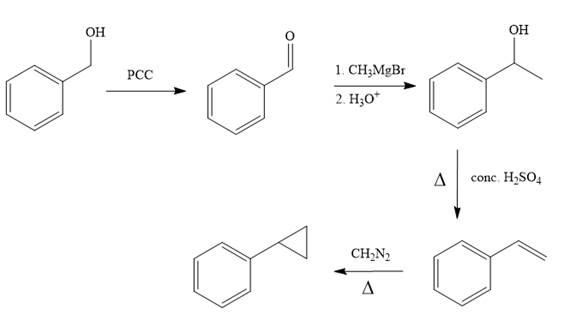
Structure of the target molecule is
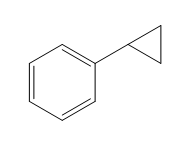
In the first step, benzyl alcohol is treated with the oxidizing agent
In the second step, benzaldehyde is treated with Grignard reagent
In the third step, the presence of conc.
In the last step, alkene is treated with diazomethane in presence of heat that results in the formation of the cyclopropane ring.
The complete synthesis of the target molecule is as shown below.
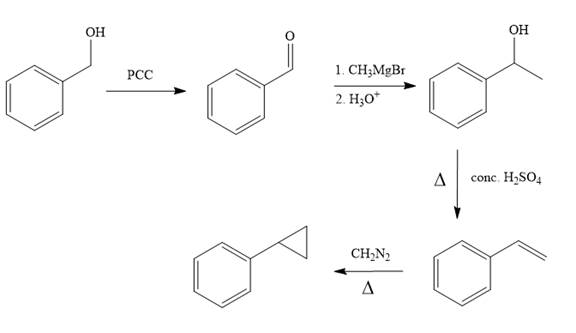
By using the functional group conversion reactions, the synthesis of the target molecule is determined.
(d)
Interpretation:
Synthesis of the given target molecule from phenylmethanol (benzyl alcohol) is to be determined.
Concept introduction:
Grignard reagent, in presence of acidic condition, is used for the conversion of aldehyde or ketone to alcohol.
Answer to Problem 19.75P
Synthesis of the target molecule from phenylmethanol (benzyl alcohol) is
Explanation of Solution
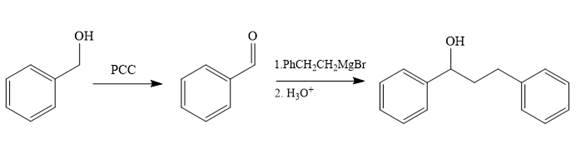
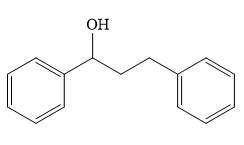
Structure of the target molecule is
In the first step, benzyl alcohol is treated with the oxidizing agent
In the second step, benzaldehyde is treated with Grignard reagent
The complete synthesis of the target molecule is as shown below.

By using the functional group, conversion reactions synthesis of the target molecule is determined.
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIf possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forward
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
- Question 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are mixed with sodium ethoxide in ethanol.arrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





