
CHEM 212:STUDENT SOLUTION MANUAL
8th Edition
ISBN: 9781260304473
Author: SILBERBERG
Publisher: MCGRAW-HILL CUSTOM PUBLISHING
expand_more
expand_more
format_list_bulleted
Question
Chapter 19, Problem 19.45P
Interpretation Introduction
Interpretation:
The color change of an indicator takes place over a range of 2
Concept Introduction:
Basis of an indicator detection:
An indicator is a substance (mostly in liquid form) that it changes its colour based on the
For an example, phenolphthalein is an acid-base indicator, and its color changes when the
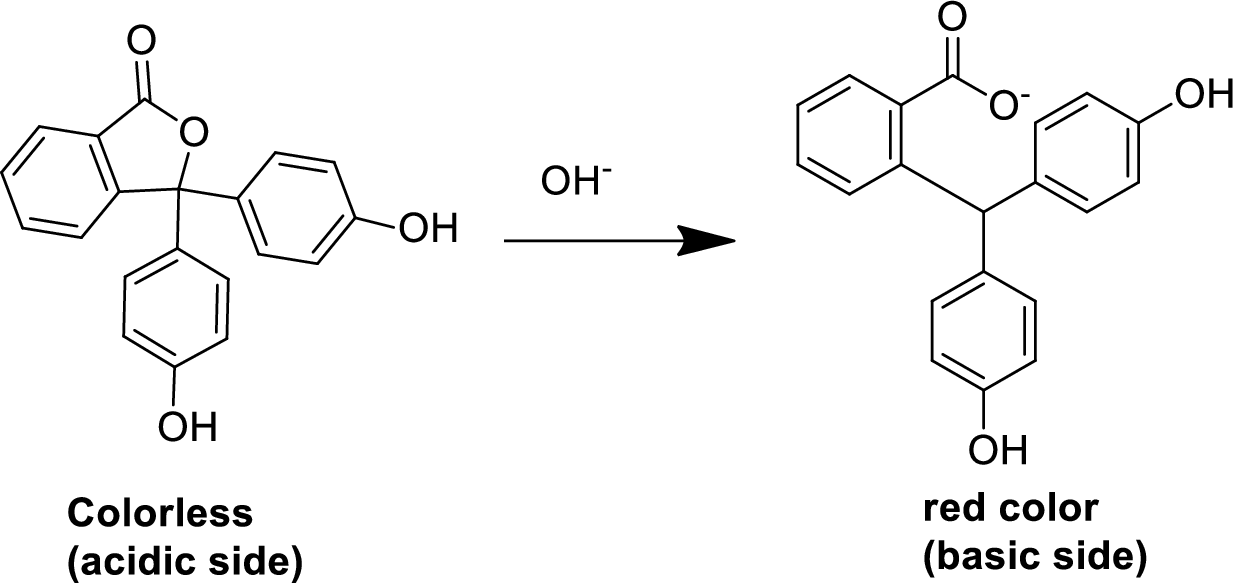
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the 'cartoon' drawing of the acceptor orbital in the first mechanistic step of an electrophilic addition reaction of butadiene with HBr. Please
H-
H
H
H
H
H
H
Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above.
H-
H
H
H
A.
H
H
H
H-C
CI
H
H
D.
H
H
H
H
H
H
C
C
-H
H
C
C
H
H
H
H
B.
H
CI
H
H-
C
C
H
H
H
H
E.
H
CI
H
C.
Why doesn't this carry on to form a ring by deprotonating the alpha carbon and the negatively-charged carbon attacking the C=O?
Chapter 19 Solutions
CHEM 212:STUDENT SOLUTION MANUAL
Ch. 19.1 - Calculate the pH of a buffer consisting of 0.50 M...Ch. 19.1 - Prob. 19.1BFPCh. 19.1 - Prob. 19.2AFPCh. 19.1 - The molecular scenes below show three samples of a...Ch. 19.1 - Prob. 19.3AFPCh. 19.1 - What is the component concentration ratio, [NH3]/...Ch. 19.2 - A chemist titrates 20.00 mL of 0.2000 M HBrO (Ka =...Ch. 19.2 - For the titration of 30.00 mL of 0.1000 M benzoic...Ch. 19.3 - Write the ion-product expression at equilibrium...Ch. 19.3 - Prob. 19.5BFP
Ch. 19.3 - Prob. 19.6AFPCh. 19.3 - Prob. 19.6BFPCh. 19.3 - Prob. 19.7AFPCh. 19.3 - Prob. 19.7BFPCh. 19.3 - Prob. 19.8AFPCh. 19.3 - Prob. 19.8BFPCh. 19.3 - Prob. 19.9AFPCh. 19.3 - Prob. 19.9BFPCh. 19.3 - Prob. 19.10AFPCh. 19.3 - Prob. 19.10BFPCh. 19.3 - Prob. 19.11AFPCh. 19.3 - Prob. 19.11BFPCh. 19.3 - Prob. 19.12AFPCh. 19.3 - Prob. 19.12BFPCh. 19.3 - An environmental technician collects a sample of...Ch. 19.3 - A lake that has a surface area of 10.0 acres (1...Ch. 19.4 - Cyanide ion is toxic because it forms stable...Ch. 19.4 - Prob. 19.13BFPCh. 19.4 - Prob. 19.14AFPCh. 19.4 - Calculate the solubility of PbCl2 in 0.75 M NaOH....Ch. 19 - Prob. 19.1PCh. 19 - Prob. 19.2PCh. 19 - Prob. 19.3PCh. 19 - Prob. 19.4PCh. 19 - Prob. 19.5PCh. 19 - Prob. 19.6PCh. 19 - Prob. 19.7PCh. 19 - Prob. 19.8PCh. 19 - Does the pH increase or decrease with each of the...Ch. 19 - The scenes below depict solutions of the same...Ch. 19 - The scenes below show three samples of a buffer...Ch. 19 - What are the [H3O+] and the pH of a propanoic...Ch. 19 - What are the [H3O+] and the pH of a benzoic...Ch. 19 - Prob. 19.14PCh. 19 - Prob. 19.15PCh. 19 - Prob. 19.16PCh. 19 - Find the pH of a buffer that consists of 0.95 M...Ch. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - Prob. 19.20PCh. 19 - Find the pH of a buffer that consists of 0.50 M...Ch. 19 - A buffer consists of 0.22 M KHCO3 and 0.37 M...Ch. 19 - A buffer consists of 0.50 M NaH2PO4 and 0.40 M...Ch. 19 - What is the component concentration ratio,...Ch. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - Prob. 19.28PCh. 19 - A buffer that contains 0.40 M of a base, B, and...Ch. 19 - A buffer that contains 0.110 M HY and 0.220 M Y−...Ch. 19 - A buffer that contains 1.05 M B and 0.750 M BH+...Ch. 19 - A buffer is prepared by mixing 204 mL of 0.452 M...Ch. 19 - A buffer is prepared by mixing 50.0 mL of 0.050 M...Ch. 19 - Prob. 19.34PCh. 19 - Prob. 19.35PCh. 19 - Prob. 19.36PCh. 19 - Choose specific acid-base conjugate pairs to make...Ch. 19 - An industrial chemist studying bleaching and...Ch. 19 - Oxoanions of phosphorus are buffer components in...Ch. 19 - The scenes below depict the relative...Ch. 19 - Prob. 19.41PCh. 19 - What species are in the buffer region of a weak...Ch. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49PCh. 19 - Prob. 19.50PCh. 19 - Prob. 19.51PCh. 19 - Prob. 19.52PCh. 19 - Prob. 19.53PCh. 19 - Prob. 19.54PCh. 19 - Prob. 19.55PCh. 19 - Prob. 19.56PCh. 19 - Prob. 19.57PCh. 19 - Prob. 19.58PCh. 19 - Prob. 19.59PCh. 19 - Prob. 19.60PCh. 19 - Prob. 19.61PCh. 19 - Use figure 19.9 to find an indicator for these...Ch. 19 - Prob. 19.63PCh. 19 - Prob. 19.64PCh. 19 - Prob. 19.65PCh. 19 - Prob. 19.66PCh. 19 - Write the ion-product expressions for (a) silver...Ch. 19 - Write the ion-product expressions for (a)...Ch. 19 - Write the ion-product expressions for (a) calcium...Ch. 19 - Prob. 19.70PCh. 19 - The solubility of silver carbonate is 0.032 M at...Ch. 19 - Prob. 19.72PCh. 19 - Prob. 19.73PCh. 19 - The solubility of calcium sulfate at 30°C is 0.209...Ch. 19 - Prob. 19.75PCh. 19 - Prob. 19.76PCh. 19 - Prob. 19.77PCh. 19 - Calculate the molar solubility of Ag2SO4 in (a)...Ch. 19 - Prob. 19.79PCh. 19 - Prob. 19.80PCh. 19 - Prob. 19.81PCh. 19 - Prob. 19.82PCh. 19 - Prob. 19.83PCh. 19 - Write equations to show whether the solubility of...Ch. 19 - Prob. 19.85PCh. 19 - Prob. 19.86PCh. 19 - Prob. 19.87PCh. 19 - Does any solid PbCl2 form when 3.5 mg of NaCl is...Ch. 19 - Prob. 19.89PCh. 19 - Prob. 19.90PCh. 19 - Prob. 19.91PCh. 19 - A 50.0-mL volume of 0.50 M Fe(NO3)3 is mixed with...Ch. 19 - Prob. 19.93PCh. 19 - Prob. 19.94PCh. 19 - Prob. 19.95PCh. 19 - Write a balanced equation for the reaction of in...Ch. 19 - Prob. 19.97PCh. 19 - Prob. 19.98PCh. 19 - Prob. 19.99PCh. 19 - What is [Ag+] when 25.0 mL each of 0.044 M AgNO3...Ch. 19 - Prob. 19.101PCh. 19 - Prob. 19.102PCh. 19 - Prob. 19.103PCh. 19 - When 0.84 g of ZnCl2 is dissolved in 245 mL of...Ch. 19 - When 2.4 g of Co(NO3)2 is dissolved in 0.350 L of...Ch. 19 - Prob. 19.106PCh. 19 - A microbiologist is preparing a medium on which to...Ch. 19 - As an FDA physiologist, you need 0.700 L of formic...Ch. 19 - Tris(hydroxymethyl)aminomethane [(HOCH2)3CNH2],...Ch. 19 - Water flowing through pipes of carbon steel must...Ch. 19 - Gout is caused by an error in metabolism that...Ch. 19 - In the process of cave formation (Section 19.3),...Ch. 19 - Phosphate systems form essential buffers in...Ch. 19 - The solubility of KCl is 3.7 M at 20°C. Two...Ch. 19 - It is possible to detect NH3 gas over 10−2 M NH3....Ch. 19 - Manganese(II) sulfide is one of the compounds...Ch. 19 - The normal pH of blood is 7.40 ± 0.05 and is...Ch. 19 - A bioengineer preparing cells for cloning bathes a...Ch. 19 - Sketch a qualitative curve for the titration of...Ch. 19 - Prob. 19.120PCh. 19 - The scene at right depicts a saturated solution of...Ch. 19 - Prob. 19.122PCh. 19 - The acid-base indicator ethyl orange turns from...Ch. 19 - Prob. 19.124PCh. 19 - Prob. 19.125PCh. 19 - Prob. 19.126PCh. 19 - Prob. 19.127PCh. 19 - Prob. 19.128PCh. 19 - Prob. 19.129PCh. 19 - Calcium ion present in water supplies is easily...Ch. 19 - Calculate the molar solubility of Hg2C2O4 (Ksp =...Ch. 19 - Environmental engineers use alkalinity as a...Ch. 19 - Human blood contains one buffer system based on...Ch. 19 - Quantitative analysis of Cl− ion is often...Ch. 19 - An ecobotanist separates the components of a...Ch. 19 - Some kidney stones form by the precipitation of...Ch. 19 - Prob. 19.137PCh. 19 - Prob. 19.138PCh. 19 - Because of the toxicity of mercury compounds,...Ch. 19 - A 35.0-mL solution of 0.075 M CaCl2 is mixed with...Ch. 19 - Rainwater is slightly acidic due to dissolved CO2....Ch. 19 - Prob. 19.142PCh. 19 - Ethylenediaminetetraacetic acid (abbreviated...Ch. 19 - Buffers that are based on...Ch. 19 - NaCl is purified by adding HCl to a saturated...Ch. 19 - Scenes A to D represent tiny portions of 0.10 M...Ch. 19 - Prob. 19.147PCh. 19 - Prob. 19.148PCh. 19 - Prob. 19.149P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6. A solution (0.0004 M) of Fe(S2CNEt2)3 (see the structural drawing below) in chloroform has absorption bands at: 350 nm (absorbance A = 2.34); 514 nm(absorbance A = 0.0532); Calculate the molar absorptivity values for these bands. Comment on their possible nature (charge transfer transitions or d-d S N- transitions?). (4 points)arrow_forwardWhat is the mechanism for this?arrow_forwardFor questions 1-4, consider the following complexes: [Co(CN)6], [COC14]², [Cr(H2O)6]²+ 4. Room temperature (20°C) measurement of molar magnetic susceptibility (Xm) for Fe(NH4)2(SO4)2×6H2O is 1.1888 x 102 cgs (Gaussian units). Calculate effective magnetic moment and provide a number of unpaired electrons for the iron ion. Use this number to rationalize the coordination geometry around iron center. (4 points)arrow_forward
- 7. Describe the expected 31P and 19F (where applicable) NMR spectral patterns for the following compounds (indicate number of signals and their splitting patterns). a) tetraphenyldiphosphine Ph Ph P-P Ph Ph Ph Ph ' b) tetraphenyldiphosphine monoxide P-P-Ph Ph (2 points) (2 points c) tetrafluorophosphonium hexafluorophosphate [PF4]*[PF6]¯ (4 points)arrow_forward3. For questions 1-4, consider the following complexes: [Co(CN)6]4, [COC14]², [Cr(H2O)6]²+ Which (if any) of these complexes would be expected to display Jahn-Teller distortion? (2 points)arrow_forwardWhat is Instrumental Neutron Activation and what are the advantages and disadvantages in using its applications? (I'm doing an in class assignment and need better understanding of what the instrument can be used for) Please include references so that I can better understand the application of how the instrument works!arrow_forward
- What is Isotope Analysis and what are the advantages and disadvantages in using its applications and instrumentalization? Please include references so that I can better understand how the instrument works!arrow_forward5. Count the electrons on the following complexes and state whether they follow the 18- electron rule: (3 points) Fe(CO)5 Ni(PMe3)4 PMe3 is trimethylphosphine Mn(CO)5Brarrow_forwardFor questions 1-4, consider the following complexes: [Co(CN)6]+, [CoCl4]², [Cr(H2O)6]²+ 2. Draw the corresponding d-orbital splitting for each of the complexes; predict the spin- state (low-spin/high spin) for each of the complexes (if applicable); explain your arguments. Calculate the crystal field stabilization energy for each complex (in Ao or At). (6 points)arrow_forward
- For questions 1-4, consider the following complexes: [Co(CN)6]4, [COC14]², [Cr(H2O)6]²+ 1. Assign oxidation number to the metal, then indicate d-electron count. (3 points)arrow_forwardUsing iodometry I want to titrate a sodium thiosulfate solution and I use 15 mL. If I have 50 mL of a 0.90 M copper solution and KI, what will be the molarity of sodium thiosulfate?arrow_forwardDraw the product formed when the following pair of compounds is treated with NaOEt in ethanol. + i CNarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Acid-Base Equilibrium; Author: Bozeman Science;https://www.youtube.com/watch?v=l5fk7HPmo5g;License: Standard YouTube License, CC-BY
Introduction to Titrimetric analysis; Author: Vidya-mitra;https://www.youtube.com/watch?v=uykGVfn9q24;License: Standard Youtube License