
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18.5D, Problem 18.2P
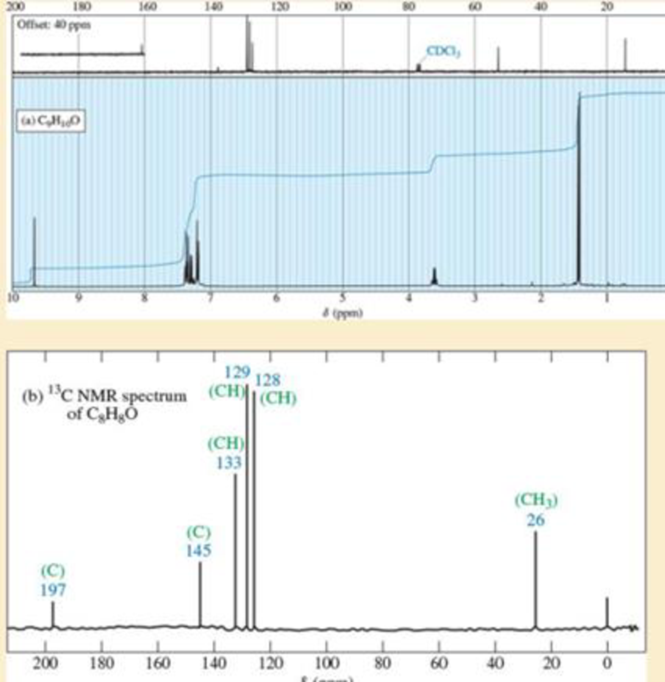
NMR spectra for two compounds are given here, together with the molecular formulas. Each compound is a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose a synthesis of 1-butanamine from the following:
(a) a chloroalkane of three carbons
(b) a chloroalkane of four carbons
Select the stronger base from each pair of compounds.
(a) H₂CNH₂ or EtzN
(b)
CI
or
NH2
NH2
(c)
.Q
or EtzN
(d)
or
(e)
N
or
(f)
H
or
H
4. Provide a clear arrow-pushing mechanism for each of the following reactions. Do not skip proton
transfers, do not combine steps, and make sure your arrows are clear enough to be interpreted
without ambiguity.
a.
2.
1. LDA
3. H3O+
HO
Chapter 18 Solutions
Organic Chemistry (9th Edition)
Ch. 18.3 - Give the IUPAC name and (if possible) a common...Ch. 18.5D - NMR spectra for two compounds are given here,...Ch. 18.5D - Why were no products from the McLafferty...Ch. 18.5D - Use equations to show the fragmentation leading to...Ch. 18.5E - Prob. 18.5PCh. 18.7D - Show how you would synthesize each compound from...Ch. 18.8 - Prob. 18.7PCh. 18.9 - Predict the products of the following reactions....Ch. 18.9 - Show how the following transformations may be...Ch. 18.10 - Prob. 18.10P
Ch. 18.11 - Show how you would accomplish the following...Ch. 18.11 - Prob. 18.12PCh. 18.12 - Propose mechanisms for a. the acid-catalyzed...Ch. 18.12 - Rank the following compounds in order of...Ch. 18.13 - Prob. 18.15PCh. 18.13 - Show how you would accomplish the following...Ch. 18.14 - Prob. 18.17PCh. 18.14 - Prob. 18.18PCh. 18.14 - Prob. 18.19PCh. 18.14 - Prob. 18.20PCh. 18.15 - 2,4-Dinitrophenylhydrazine is frequently used for...Ch. 18.15 - Prob. 18.22PCh. 18.15 - Prob. 18.23PCh. 18.16 - Prob. 18.24PCh. 18.16 - Prob. 18.25PCh. 18.16 - Show what alcohols and carbonyl compounds give the...Ch. 18.16 - In the mechanism for acetal hydrolysis shown, the...Ch. 18.16 - Prob. 18.28PCh. 18.17 - Show how you would accomplish the following...Ch. 18.18 - Prob. 18.30PCh. 18.18 - Prob. 18.31PCh. 18.18 - Prob. 18.32PCh. 18.18 - Show how Wittig reactions might be used to...Ch. 18.19 - Predict the major products of the following...Ch. 18.20C - Prob. 18.35PCh. 18.20C - Predict the major products of the following...Ch. 18 - Draw structures of the following derivatives. a....Ch. 18 - Prob. 18.38SPCh. 18 - Predict the major products of the following...Ch. 18 - Rank the following carbonyl compounds in order of...Ch. 18 - Acetals can serve as protecting groups for...Ch. 18 - Sketch the expected proton NMR spectrum of...Ch. 18 - A compound of formula C6H10O2 shows only two...Ch. 18 - The proton NMR spectrum of a compound of formula...Ch. 18 - The following compounds undergo McLafferty...Ch. 18 - An unknown compound gives a molecular ion of m/z...Ch. 18 - Show how you would accomplish the following...Ch. 18 - Prob. 18.48SPCh. 18 - Prob. 18.49SPCh. 18 - Propose mechanisms for the following reactions.Ch. 18 - Show how you would accomplish the following...Ch. 18 - Show how you would synthesize the following...Ch. 18 - Predict the products formed when cyclohexanone...Ch. 18 - Predict the products formed when...Ch. 18 - Show how you would synthesize octan-2-one from...Ch. 18 - Prob. 18.56SPCh. 18 - Both NaBH4 and NaBD4 are commercially available,...Ch. 18 - When LiAIH4 reduces 3-methylcyclopentanone, the...Ch. 18 - Prob. 18.59SPCh. 18 - Show how you would accomplish the following...Ch. 18 - There are three dioxane isomers 1,2-dioxane,...Ch. 18 - Two structures for the sugar glucose are shown on...Ch. 18 - Prob. 18.63SPCh. 18 - Prob. 18.64SPCh. 18 - Prob. 18.65SPCh. 18 - Prob. 18.66SPCh. 18 - Within each set of structures, indicate which will...Ch. 18 - Prob. 18.68SPCh. 18 - Prob. 18.69SPCh. 18 - Prob. 18.70SPCh. 18 - The UV spectrum of an unknown compound shows...Ch. 18 - a. Simple aminoacetals hydrolyze quickly and...Ch. 18 - The mass spectrum of unknown compound A shows a...Ch. 18 - Prob. 18.74SPCh. 18 - Prob. 18.75SPCh. 18 - Prob. 18.76SPCh. 18 - Prob. 18.77SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- b. H3C CH3 H3O+ ✓ H OHarrow_forward2. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases. a. CH3arrow_forwardIdentify and provide an explanation that distinguishes a qualitative and quantitative chemical analysis. Provide examples.arrow_forward
- Identify and provide an explanation of the operational principles behind a Atomic Absorption Spectrometer (AAS). List the steps involved.arrow_forwardInstructions: Complete the questions in the space provided. Show all your work 1. You are trying to determine the rate law expression for a reaction that you are completing at 25°C. You measure the initial reaction rate and the starting concentrations of the reactions for 4 trials. BrO³¯ (aq) + 5Br¯ (aq) + 6H* (aq) → 3Br₂ (l) + 3H2O (l) Initial rate Trial [BrO3] [H*] [Br] (mol/L) (mol/L) | (mol/L) (mol/L.s) 1 0.10 0.10 0.10 8.0 2 0.20 0.10 0.10 16 3 0.10 0.20 0.10 16 4 0.10 0.10 0.20 32 a. Based on the above data what is the rate law expression? b. Solve for the value of k (make sure to include proper units) 2. The proposed reaction mechanism is as follows: i. ii. BrО¸¯ (aq) + H+ (aq) → HBrO3 (aq) HBrO³ (aq) + H* (aq) → H₂BrO3* (aq) iii. H₂BrO³* (aq) + Br¯ (aq) → Br₂O₂ (aq) + H2O (l) [Fast] [Medium] [Slow] iv. Br₂O₂ (aq) + 4H*(aq) + 4Br(aq) → 3Br₂ (l) + H2O (l) [Fast] Evaluate the validity of this proposed reaction. Justify your answer.arrow_forwardе. Д CH3 D*, D20arrow_forward
- H3C. H3C CH 3 CH 3 CH3 1. LDA 2. PhSeCl 3. H2O2arrow_forwardPlease predict the products for each of the following reactions: 1.03 2. H₂O NaNH, 1. n-BuLi 2. Mel A H₂ 10 9 0 H2SO4, H₂O HgSO4 Pd or Pt (catalyst) B 9 2 n-BuLi ♡ D2 (deuterium) Lindlar's Catalyst 1. NaNH2 2. EtBr Na, ND3 (deuterium) 2. H₂O2, NaOH 1. (Sia)2BH с Darrow_forwardin the scope of ontario SCH4U grade 12 course, please show ALL workarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY