
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18.19, Problem 18.34P
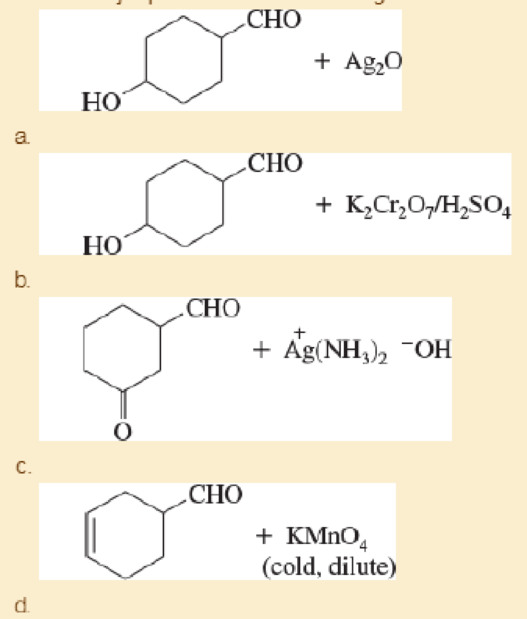
Predict the major products of the following reactions.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the mechanism for this?
21.50 Determine the combinations of haloalkane(s) and alkoxide(s) that could be used to
synthesize the following ethers through Williamson ether synthesis.
(a)
(c)
(d)
(e)
(f)
H₂CO
1. Arrange the following in order of increasing bond energy (lowest bond energy first, highest bond
energy last). Provide your rationale. C=C, C-F, C=C, C-N, C-C
List the bond order for each example.
Chapter 18 Solutions
Organic Chemistry (9th Edition)
Ch. 18.3 - Give the IUPAC name and (if possible) a common...Ch. 18.5D - NMR spectra for two compounds are given here,...Ch. 18.5D - Why were no products from the McLafferty...Ch. 18.5D - Use equations to show the fragmentation leading to...Ch. 18.5E - Prob. 18.5PCh. 18.7D - Show how you would synthesize each compound from...Ch. 18.8 - Prob. 18.7PCh. 18.9 - Predict the products of the following reactions....Ch. 18.9 - Show how the following transformations may be...Ch. 18.10 - Prob. 18.10P
Ch. 18.11 - Show how you would accomplish the following...Ch. 18.11 - Prob. 18.12PCh. 18.12 - Propose mechanisms for a. the acid-catalyzed...Ch. 18.12 - Rank the following compounds in order of...Ch. 18.13 - Prob. 18.15PCh. 18.13 - Show how you would accomplish the following...Ch. 18.14 - Prob. 18.17PCh. 18.14 - Prob. 18.18PCh. 18.14 - Prob. 18.19PCh. 18.14 - Prob. 18.20PCh. 18.15 - 2,4-Dinitrophenylhydrazine is frequently used for...Ch. 18.15 - Prob. 18.22PCh. 18.15 - Prob. 18.23PCh. 18.16 - Prob. 18.24PCh. 18.16 - Prob. 18.25PCh. 18.16 - Show what alcohols and carbonyl compounds give the...Ch. 18.16 - In the mechanism for acetal hydrolysis shown, the...Ch. 18.16 - Prob. 18.28PCh. 18.17 - Show how you would accomplish the following...Ch. 18.18 - Prob. 18.30PCh. 18.18 - Prob. 18.31PCh. 18.18 - Prob. 18.32PCh. 18.18 - Show how Wittig reactions might be used to...Ch. 18.19 - Predict the major products of the following...Ch. 18.20C - Prob. 18.35PCh. 18.20C - Predict the major products of the following...Ch. 18 - Draw structures of the following derivatives. a....Ch. 18 - Prob. 18.38SPCh. 18 - Predict the major products of the following...Ch. 18 - Rank the following carbonyl compounds in order of...Ch. 18 - Acetals can serve as protecting groups for...Ch. 18 - Sketch the expected proton NMR spectrum of...Ch. 18 - A compound of formula C6H10O2 shows only two...Ch. 18 - The proton NMR spectrum of a compound of formula...Ch. 18 - The following compounds undergo McLafferty...Ch. 18 - An unknown compound gives a molecular ion of m/z...Ch. 18 - Show how you would accomplish the following...Ch. 18 - Prob. 18.48SPCh. 18 - Prob. 18.49SPCh. 18 - Propose mechanisms for the following reactions.Ch. 18 - Show how you would accomplish the following...Ch. 18 - Show how you would synthesize the following...Ch. 18 - Predict the products formed when cyclohexanone...Ch. 18 - Predict the products formed when...Ch. 18 - Show how you would synthesize octan-2-one from...Ch. 18 - Prob. 18.56SPCh. 18 - Both NaBH4 and NaBD4 are commercially available,...Ch. 18 - When LiAIH4 reduces 3-methylcyclopentanone, the...Ch. 18 - Prob. 18.59SPCh. 18 - Show how you would accomplish the following...Ch. 18 - There are three dioxane isomers 1,2-dioxane,...Ch. 18 - Two structures for the sugar glucose are shown on...Ch. 18 - Prob. 18.63SPCh. 18 - Prob. 18.64SPCh. 18 - Prob. 18.65SPCh. 18 - Prob. 18.66SPCh. 18 - Within each set of structures, indicate which will...Ch. 18 - Prob. 18.68SPCh. 18 - Prob. 18.69SPCh. 18 - Prob. 18.70SPCh. 18 - The UV spectrum of an unknown compound shows...Ch. 18 - a. Simple aminoacetals hydrolyze quickly and...Ch. 18 - The mass spectrum of unknown compound A shows a...Ch. 18 - Prob. 18.74SPCh. 18 - Prob. 18.75SPCh. 18 - Prob. 18.76SPCh. 18 - Prob. 18.77SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the major enolate formed when treated with LDA? And why that one?arrow_forward4. Calculate the total number of sigma bonds and total number of pi bonds in each of the following compounds. a. HH :D: +1 I H-N-C-C-O-H I H b. HH H Н :N=C-C-C=C-CEC-H :0: total o H-C-H H-C = `C-H I H. 11 H-C = C= CH H total o total π total π 1 Harrow_forwardIn the following reaction, what quantity in moles of CH₃OH are required to give off 4111 kJ of heat? 2 CH₃OH (l) + 3 O₂ (g) → 2 CO₂ (g) + 4 H₂O(g) ∆H° = -1280. kJarrow_forward
- Indicate the processes in the dismutation of Cu2O.arrow_forward1. Consider these three reactions as the elementary steps in the mechanism for a chemical reaction. 2600 2400 2200 2000 1800 1600 1400 1200 1000 800 Potential Energy (kJ) 600 400 200 0 -200- -400 -600- -800 (i) Cl₂ (g) + Pt(s) → 2Cl (g) + Pt(s) (ii) Cl (g)+ CO (g) + Pt (s) → CICO (g) + Pt (s) Ea = 1550 kJ Ea = 2240 kJ (iii) Cl (g) + CICO (g) → Cl₂CO (g) Ea = 2350 kJ AH=-950 kJ ΔΗ = 575 ΚΙ AH=-825 kJ a. Draw the potential energy diagram for the reaction. Label the data points for clarity. The potential energy of the reactants is 600 kJ Reaction Progress b. What is the overall chemical equation? c. What is the overall change in enthalpy for the above chemical reaction? d. What is the overall amount of activation energy for the above chemical reaction? e. Which reaction intermediate would be considered a catalyst (if any) and why? f. If you were to add 2700kJ of energy to the reaction (e.g. 2700 kl of heat or electricity), would you be able to make the reaction reverse itself (i.e. have…arrow_forwarddraw the enolate anion and the carbonyl that would be needed to make this product through an aldol addition reaction.arrow_forward
- Draw the Michael Adduct and the final product of the Robinson annulation reaction. Ignore inorganic byproducts.arrow_forwardDraw the Michael adduct and final product of the Robinson annulation reaction. Ignore inorganic byproductsarrow_forwardPost Lab Questions. 1) Draw the mechanism of your Diels-Alder cycloaddition. 2) Only one isomer of product is formed in the Diels-Alder cycloaddition. Why? 3) Imagine that you used isoprene as diene - in that case you don't have to worry about assigning endo vs exo. Draw the "endo" and "exo" products of the Diels-Alder reaction between isoprene and maleic anhydride, and explain why the distinction is irrelevant here. 4) This does not hold for other dienes. Draw the exo and endo products of the reaction of cyclohexadiene with maleic anhydride. Make sure you label your answers properly as endo or exo. 100 °C Xylenes ??? 5) Calculate the process mass intensity for your specific reaction (make sure to use your actual amounts of reagent).arrow_forward
- Indicate the product(s) A, B C and D that are formed in the reaction: H + NH-NH-CH [A+B] [C+D] hydrazonesarrow_forwardHow can you prepare a 6 mL solution of 6% H2O2, if we have a bottle of 30% H2O2?arrow_forwardHow many mL of H2O2 from the 30% bottle must be collected to prepare 6 mL of 6% H2O2.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License