
Physics (5th Edition)
5th Edition
ISBN: 9780321976444
Author: James S. Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18.2, Problem 2EYU
Enhance Your Understanding
(Answers given at the end of the chapter)
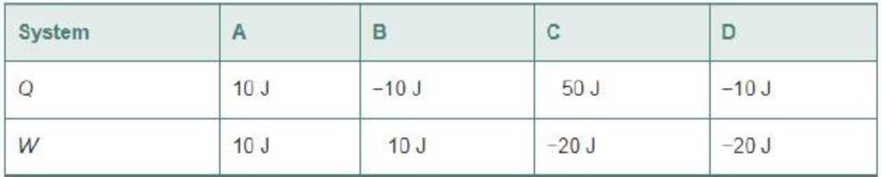
2. The following systems exchange energy with the external world in the form of heat Q and work W. Rank the systems in order of increasing change in internal energy, from most negative to most positive. Indicate ties where appropriate.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can someone help me answer this physics 2 questions. Thank you.
Four capacitors are connected as shown in the figure below. (Let C = 12.0 μF.)
a
C
3.00 με
Hh.
6.00 με
20.0 με
HE
(a) Find the equivalent capacitance between points a and b.
5.92
HF
(b) Calculate the charge on each capacitor, taking AV ab = 16.0 V.
20.0 uF capacitor 94.7
6.00 uF capacitor 67.6
32.14
3.00 µF capacitor
capacitor C
☑
με
με
The 3 µF and 12.0 uF capacitors are in series and that combination is in parallel with the 6 μF capacitor. What quantity is the same for capacitors in parallel? μC
32.14
☑
You are correct that the charge on this capacitor will be the same as the charge on the 3 μF capacitor. μC
In the pivot assignment, we observed waves moving on a string stretched by hanging
weights. We noticed that certain frequencies produced standing waves. One such
situation is shown below:
0 ст
Direct Measurement
©2015 Peter Bohacek I.
20
0 cm 10
20
30
40
50
60
70
80
90
100
Which Harmonic is this?
Do NOT include units!
What is the wavelength of this wave in cm with only no decimal places?
If the speed of this wave is 2500 cm/s, what is the frequency of this harmonic (in Hz, with
NO decimal places)?
Chapter 18 Solutions
Physics (5th Edition)
Ch. 18.1 - System 1 is at 0 C and system 2 is at 0 F. If...Ch. 18.2 - Enhance Your Understanding (Answers given at the...Ch. 18.3 - Enhance Your Understanding (Answers given at the...Ch. 18.4 - Enhance Your Understanding (Answers given at the...Ch. 18.5 - Enhance Your Understanding (Answers given at the...Ch. 18.6 - Enhance Your Understanding (Answers given at the...Ch. 18.7 - Enhance Your Understanding (Answers given at the...Ch. 18.8 - Enhance Your Understanding (Answers given at the...Ch. 18.9 - Enhance Your Understanding (Answers given at the...Ch. 18.10 - Enhance Your Understanding (Answer given at the...
Ch. 18 - Prob. 1CQCh. 18 - Heat is added to a substance. Is it safe to...Ch. 18 - Are there thermodynamic processes in which all the...Ch. 18 - An ideal gas is held in an insulated container at...Ch. 18 - Prob. 5CQCh. 18 - Which law of thermodynamics would be violated if...Ch. 18 - Heat engines always give off a certain amount of...Ch. 18 - Prob. 8CQCh. 18 - Which law of thermodynamics is most pertinent to...Ch. 18 - Which has more entropy: (a) popcorn kernels, or...Ch. 18 - Prob. 1PCECh. 18 - A gas expands, doing 100 J of work. How much heat...Ch. 18 - A swimmer does 7.7 105 J of work and gives off...Ch. 18 - When 1310 J of heat are added to one mole of an...Ch. 18 - Three different processes act on a system. (a) In...Ch. 18 - A container holds a gas consisting of 2.85 moles...Ch. 18 - The Charge on Adhesive Tape When adhesive tape is...Ch. 18 - Predict/Calculate One mole of an ideal monatomic...Ch. 18 - Prob. 9PCECh. 18 - A cylinder contains 4.0 moles of a monatomic gas...Ch. 18 - An ideal gas is taken through the three processes...Ch. 18 - Figure 18-26 shows three different multistep...Ch. 18 - Prob. 13PCECh. 18 - An ideal gas is compressed at constant pressure to...Ch. 18 - As an ideal gas expands at constant pressure from...Ch. 18 - A system consisting of an ideal gas at the...Ch. 18 - Prob. 17PCECh. 18 - (a) Find the work done by a monatomic ideal gas as...Ch. 18 - Prob. 19PCECh. 18 - Predict/Calculate If 9.50 moles of a monatomic...Ch. 18 - Suppose 118 moles of a monatomic ideal gas undergo...Ch. 18 - A weather balloon contains an ideal gas and has a...Ch. 18 - Prob. 23PCECh. 18 - During an adiabatic process, the temperature of...Ch. 18 - An ideal gas follows the three-part process shown...Ch. 18 - With the pressure held constant at 260 kPa, 43 mol...Ch. 18 - Prob. 27PCECh. 18 - A system expands by 0.75 m3 at a constant pressure...Ch. 18 - Prob. 29PCECh. 18 - A certain amount of a monatomic ideal gas...Ch. 18 - An ideal gas doubles its volume in one of three...Ch. 18 - Predict/Explain You plan to add a certain amount...Ch. 18 - Find the amount of heat needed to increase the...Ch. 18 - (a) If 585 J of heat are added to 49 moles of a...Ch. 18 - A system consists of 3.5 mol of an ideal monatomic...Ch. 18 - Find the change in temperature if 170 J of heat...Ch. 18 - Gasoline Ignition Consider a short time span just...Ch. 18 - Prob. 38PCECh. 18 - Prob. 39PCECh. 18 - A monatomic ideal gas is held in a thermally...Ch. 18 - Consider the expansion of 60.0 moles of a...Ch. 18 - A Carnot engine can be operated with one of the...Ch. 18 - What is the efficiency of an engine that exhausts...Ch. 18 - An engine receives 660 J of heat from a hot...Ch. 18 - A Carnot engine operates between the temperatures...Ch. 18 - A nuclear power plant has a reactor that produces...Ch. 18 - At a coal-burning power plant a steam turbine is...Ch. 18 - Predict/Calculate A portable generator produces...Ch. 18 - Predict/Calculate The efficiency of a particular...Ch. 18 - During each cycle a reversible engine absorbs 3100...Ch. 18 - Prob. 51PCECh. 18 - The operating temperatures for a Carnot engine are...Ch. 18 - A certain Carnot engine takes in the heat Qh and...Ch. 18 - Predict/Explain (a) If the temperature in the...Ch. 18 - The refrigerator in your kitchen does 490 J of...Ch. 18 - A refrigerator with a coefficient of performance...Ch. 18 - Prob. 57PCECh. 18 - Prob. 58PCECh. 18 - An air conditioner is used to keep the interior of...Ch. 18 - A reversible refrigerator has a coefficient of...Ch. 18 - A freezer has a coefficient of performance equal...Ch. 18 - Predict/Explain (a) If you rub your hands...Ch. 18 - Predict/Explain (a) An ideal gas is expanded...Ch. 18 - Predict/Explain (a) A gas is expanded reversibly...Ch. 18 - Find the change in entropy when 1.85 kg of water...Ch. 18 - Determine the change in entropy that occurs when...Ch. 18 - Prob. 67PCECh. 18 - On a cold winters day heat leaks slowly out of a...Ch. 18 - An 88-kg parachutist descends through a vertical...Ch. 18 - Predict/Calculate Consider the air-conditioning...Ch. 18 - A heat engine operates between a high-temperature...Ch. 18 - It can be shown that as a mass m with specific...Ch. 18 - Prob. 73GPCh. 18 - Figure 18-34 Problem 74 74 CE An ideal gas has...Ch. 18 - The heat that goes into a particular Carnot engine...Ch. 18 - Predict/Calculate Consider 132 moles of a...Ch. 18 - Prob. 77GPCh. 18 - Prob. 78GPCh. 18 - Predict/Calculate Engine A has an efficiency of...Ch. 18 - Nuclear Versus Natural Gas Energy Because of...Ch. 18 - A freezer with a coefficient of performance of...Ch. 18 - Entropy and the Sun The surface of the Sun has a...Ch. 18 - Prob. 83GPCh. 18 - A cylinder with a movable piston holds 2.95 mol of...Ch. 18 - Making Ice You place 0.410 kg of cold water inside...Ch. 18 - An inventor claims a new cyclic engine that uses...Ch. 18 - Predict/Calculate A small dish containing 530 g of...Ch. 18 - Predict/Calculate An ideal gas is taken through...Ch. 18 - One mole of an ideal monatomic gas follows the...Ch. 18 - When a heat Q is added to a monatomic ideal gas at...Ch. 18 - The Carnot Cycle Figure 18-36 shows an example of...Ch. 18 - A Carnot engine and a Carnot refrigerator operate...Ch. 18 - Prob. 93PPCh. 18 - Energy from the Ocean Whenever two objects are at...Ch. 18 - Prob. 95PPCh. 18 - Energy from me Ocean Whenever two objects are at...Ch. 18 - Predict/Calculate Referring to Example 18-21...Ch. 18 - Predict/Calculate Referring to Example 18-21...
Additional Science Textbook Solutions
Find more solutions based on key concepts
2. List the subdivisions of the thoracic and abdominopelvic cavities.
Human Anatomy & Physiology (2nd Edition)
Choose the best answer to each of the following. Explain your reasoning. If you observe two Cepheid variable st...
Cosmic Perspective Fundamentals
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
When a hydrochloric acid solution is combined with a potassium hydroxide solution, an acid-base reaction occurs...
Introductory Chemistry (6th Edition)
18. SCIENTIFIC THINKING By measuring the fossil remains of Homo floresiensis, scientists have estimated its wei...
Campbell Biology: Concepts & Connections (9th Edition)
a. Draw the mechanism for the following reaction if it a involves specific-base catalysis. b. Draw the mechanis...
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Four capacitors are connected as shown in the figure below. (Let C = 12.0 µF.) A circuit consists of four capacitors. It begins at point a before the wire splits in two directions. On the upper split, there is a capacitor C followed by a 3.00 µF capacitor. On the lower split, there is a 6.00 µF capacitor. The two splits reconnect and are followed by a 20.0 µF capacitor, which is then followed by point b. (a) Find the equivalent capacitance between points a and b. µF(b) Calculate the charge on each capacitor, taking ΔVab = 16.0 V. 20.0 µF capacitor µC 6.00 µF capacitor µC 3.00 µF capacitor µC capacitor C µCarrow_forwardTwo conductors having net charges of +14.0 µC and -14.0 µC have a potential difference of 14.0 V between them. (a) Determine the capacitance of the system. F (b) What is the potential difference between the two conductors if the charges on each are increased to +196.0 µC and -196.0 µC? Varrow_forwardPlease see the attached image and answer the set of questions with proof.arrow_forward
- How, Please type the whole transcript correctly using comma and periods as needed. I have uploaded the picture of a video on YouTube. Thanks,arrow_forwardA spectra is a graph that has amplitude on the Y-axis and frequency on the X-axis. A harmonic spectra simply draws a vertical line at each frequency that a harmonic would be produced. The height of the line indicates the amplitude at which that harmonic would be produced. If the Fo of a sound is 125 Hz, please sketch a spectra (amplitude on the Y axis, frequency on the X axis) of the harmonic series up to the 4th harmonic. Include actual values on Y and X axis.arrow_forwardSketch a sign wave depicting 3 seconds of wave activity for a 5 Hz tone.arrow_forward
- Sketch a sine wave depicting 3 seconds of wave activity for a 5 Hz tone.arrow_forwardThe drawing shows two long, straight wires that are suspended from the ceiling. The mass per unit length of each wire is 0.050 kg/m. Each of the four strings suspending the wires has a length of 1.2 m. When the wires carry identical currents in opposite directions, the angle between the strings holding the two wires is 20°. (a) Draw the free-body diagram showing the forces that act on the right wire with respect to the x axis. Account for each of the strings separately. (b) What is the current in each wire? 1.2 m 20° I -20° 1.2 marrow_forwardplease solve thisarrow_forward
- please solve everything in detailarrow_forward6). What is the magnitude of the potential difference across the 20-02 resistor? 10 Ω 11 V - -Imm 20 Ω 10 Ω 5.00 10 Ω a. 3.2 V b. 7.8 V C. 11 V d. 5.0 V e. 8.6 Varrow_forward2). How much energy is stored in the 50-μF capacitor when Va - V₁ = 22V? 25 µF b 25 µF 50 µFarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY