
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
14th Edition
ISBN: 9780136873891
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 8E
The first stage of treatment at the reverse osmosis plant in Carlsbad, California, is to flow the water through rock, sand, and gravel as shown here. Would this step remove particulate matter? Would this step remove dissolved salts? [Section 18.4]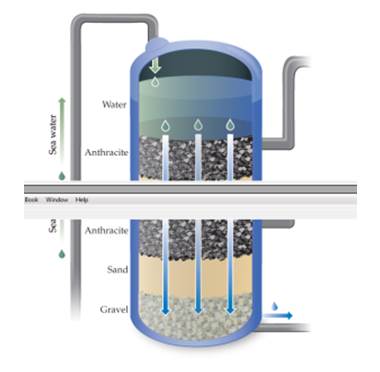
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule02:10
Students have asked these similar questions
Fill in the blanks by selecting the appropriate term from below:
For a process that is non-spontaneous and that favors products at equilibrium, we know that a) ΔrG∘ΔrG∘ _________, b) ΔunivSΔunivS _________, c) ΔsysSΔsysS _________, and d) ΔrH∘ΔrH∘ _________.
Highest occupied molecular orbital
Lowest unoccupied molecular orbital
Label all nodes and regions of highest and lowest electron density for both orbitals.
Relative Intensity
Part VI. consider the multi-step reaction below for compounds A, B, and C.
These compounds were subjected to mass spectrometric analysis and
the following spectra for A, B, and C was obtained.
Draw the structure of B and C and match all three compounds
to the correct spectra.
Relative Intensity
Relative Intensity
20
NaоH
0103
Br
(B)
H2504
→ (c)
(A)
100-
MS-NU-0547
80
40
20
31
10
20
100-
MS2016-05353CM
80
60
100
MS-NJ-09-3
80
60
40
20
45
J.L
80
S1
84
M+
absent
राग
135 137
S2
62
164 166
11
S3
25
50
75
100
125
150
175
m/z
Chapter 18 Solutions
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
Ch. 18.1 - Prob. 18.1.1PECh. 18.1 - Prob. 18.1.2PECh. 18.1 - Prob. 18.2.1PECh. 18.1 - Practice Exercise 2 The bond energy in N2 is 941...Ch. 18.2 - Prob. 18.3.1PECh. 18.2 - Prob. 18.3.2PECh. 18 - Prob. 1DECh. 18 - Prob. 1ECh. 18 - Prob. 2ECh. 18 - The figure shows the three lowest regions of...
Ch. 18 - Prob. 4ECh. 18 - Where does the energy come from to evaporate the...Ch. 18 - Prob. 6ECh. 18 - Prob. 7ECh. 18 - The first stage of treatment at the reverse...Ch. 18 - Prob. 9ECh. 18 - Prob. 10ECh. 18 - Prob. 11ECh. 18 - How are the boundaries between the regions of the...Ch. 18 - Air pollution in the Mexico City metropolitan area...Ch. 18 - Prob. 14ECh. 18 - Prob. 15ECh. 18 - Prob. 16ECh. 18 - Prob. 17ECh. 18 - Prob. 18ECh. 18 - Distinguish between photodissociation and...Ch. 18 - Prob. 20ECh. 18 - Prob. 21ECh. 18 - Prob. 22ECh. 18 - Do the reactions involved in ozone depletion...Ch. 18 - Prob. 24ECh. 18 - Prob. 25ECh. 18 - Prob. 26ECh. 18 - Prob. 27ECh. 18 - Prob. 28ECh. 18 - Prob. 29ECh. 18 - Prob. 30ECh. 18 - Prob. 31ECh. 18 - Prob. 32ECh. 18 - Alcohol-based fuels for automobiles lead to the...Ch. 18 - Prob. 34ECh. 18 - Prob. 35ECh. 18 - Prob. 36ECh. 18 - Prob. 37ECh. 18 - Prob. 38ECh. 18 - Prob. 39ECh. 18 - Prob. 40ECh. 18 - Prob. 41ECh. 18 - Prob. 42ECh. 18 - Although there are many ions in seawater, the...Ch. 18 - The Ogallala aquifer described in the Close Look...Ch. 18 - Prob. 45ECh. 18 - Prob. 46ECh. 18 - List the common products formed when an organic...Ch. 18 - Prob. 48ECh. 18 - Prob. 49ECh. 18 - Prob. 50ECh. 18 - Prob. 51ECh. 18 - Prob. 52ECh. 18 - Prob. 53ECh. 18 - Prob. 54ECh. 18 - Prob. 55ECh. 18 - Prob. 56ECh. 18 - Prob. 57ECh. 18 - Prob. 58ECh. 18 - Prob. 59ECh. 18 - Prob. 60ECh. 18 - Prob. 61AECh. 18 - Prob. 62AECh. 18 - Prob. 63AECh. 18 - Prob. 64AECh. 18 - Prob. 65AECh. 18 - Prob. 66AECh. 18 - Prob. 67AECh. 18 - Explain, using Le Châtelier’s principle, why the...Ch. 18 - Prob. 69AECh. 18 - Prob. 70AECh. 18 - Prob. 71AECh. 18 - Prob. 72AECh. 18 - Prob. 73AECh. 18 - Prob. 74AECh. 18 - Prob. 75AECh. 18 - Prob. 76AECh. 18 - Prob. 77AECh. 18 - Prob. 78IECh. 18 - Prob. 79IECh. 18 - Prob. 80IECh. 18 - Prob. 81IECh. 18 - Prob. 82IECh. 18 - Prob. 83IECh. 18 - Prob. 84IECh. 18 - 18.85 The main reason that distillation is a...Ch. 18 - Prob. 86IECh. 18 - Prob. 87IECh. 18 - Prob. 88IECh. 18 - Prob. 89IECh. 18 - Prob. 90IECh. 18 - Prob. 91IECh. 18 - Prob. 92IE
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why is petroleum jelly used in the hanging-drop procedure?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Another cross in Drosophila involved the recessive, X-linked genes yellow (y), white (w), and cut (ct). A yello...
Concepts of Genetics (12th Edition)
10.1 Indicate whether each of the following statements is characteristic of an acid, a base, or
both:
has a so...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Identify each of the following reproductive barriers as prezygotic or postzygotic. a. One lilac species lives o...
Campbell Essential Biology with Physiology (5th Edition)
57. Which buffer system is the best choice to create a buffer with pH = 7.20? For the best system, calculate th...
Chemistry: A Molecular Approach (4th Edition)
Foods packed in plastic for microwaving are a. dehydrated. b. freeze-dried. c. packaged aseptically. d. commerc...
Microbiology: An Introduction
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A composite material reinforced with aligned fibers, consisting of 20% by volume of silicon carbide (SiC) fibers and 80% by volume of polycarbonate (PC) matrix. The mechanical characteristics of the 2 materials are in the table. The stress of the matrix when the fiber breaks is 45 MPa. Calculate the longitudinal strength? SiC PC Elastic modulus (GPa) Tensile strength (GPa) 400 2,4 3,9 0,065arrow_forwardQuestion 2 What starting materials or reagents are best used to carry out the following reaction? 2Fe, 3Br2 ○ FeCl3 2Fe, 4Br2 O Heat and Br2 Heat and HBr Brarrow_forwardWhat is/are the major product(s) of the following reaction? O AICI -Chts +arrow_forward
- Shown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H. C H H C H :Ö: Click and drag to start drawing a structure.arrow_forwardShown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H. C H H C. H H H H Click and drag to start drawing a structure. Xarrow_forwardA new brand of lotion is causing skin rush unlike the old brand of the same lotion. With the aid of well labelled diagram describe an experiment that could be done to isolate the pigment that cause the skin rusharrow_forward
- Don't used hand raitingarrow_forwardDon't used hand raitingarrow_forwardRelative Intensity Part VI. consider the multi-step reaction below for compounds A, B, and C. These compounds were subjected to mass spectrometric analysis and the following spectra for A, B, and C was obtained. Draw the structure of B and C and match all three compounds to the correct spectra. Relative Intensity Relative Intensity 100 HS-NJ-0547 80 60 31 20 S1 84 M+ absent 10 30 40 50 60 70 80 90 100 100- MS2016-05353CM 80- 60 40 20 135 137 S2 164 166 0-m 25 50 75 100 125 150 m/z 60 100 MS-NJ-09-43 40 20 20 80 45 S3 25 50 75 100 125 150 175 m/zarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER
ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY