
EBK STUDENT SOLUTIONS MANUAL TO ACCOMPA
7th Edition
ISBN: 9781119360902
Author: HYSLOP
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 70RQ
Which system has a higher entropy?
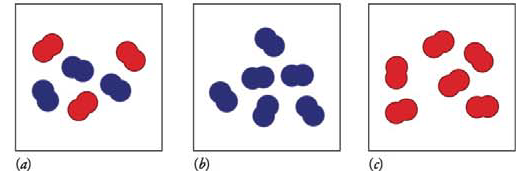
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
None
Part II. two unbranched ketone have molecular formulla (C8H100). El-ms showed that
both of them
have a molecular ion peak at m/2 =128. However ketone
(A) has a fragment peak at m/2 = 99 and 72
while ketone (B) snowed a
fragment peak at m/2 = 113 and 58.
9) Propose the most plausible structures for both ketones
b) Explain how you arrived at your conclusion by drawing the
Structures of the distinguishing fragments for each ketone,
including their fragmentation mechanisms.
Part V. Draw the structure of compound tecla using the IR spectrum Cobtained from
the compound in KBr pellet) and the mass spectrum as shown below.
The mass spectrum of compound Tesla showed strong mt peak at 71.
TRANSMITTANCE
LOD
Relative Intensity
100
MS-NW-1539
40
20
80
T
44
55
10
15
20
25
30
35
40
45
50
55
60
65
70
75
m/z
D
4000
3000
2000
1500
1000
500
HAVENUMBERI-11
Chapter 18 Solutions
EBK STUDENT SOLUTIONS MANUAL TO ACCOMPA
Ch. 18 - Molecules of an ideal gas have no intermolecular...Ch. 18 - If a gas is compressed under adiabatic conditions...Ch. 18 - Calculate the difference, in kilojoules, between...Ch. 18 - The reaction CaO(s)+2HCl(g)CaCl2(s)+H2O(g) has...Ch. 18 - Are the following processes spontaneous? (a) Ice...Ch. 18 - The following processes are nor spontaneous as...Ch. 18 - Would you expect the to be positive or negative...Ch. 18 - Predict the sign of the entropy change for (a) the...Ch. 18 - Predict the sing of S for the following reactions:...Ch. 18 - Would you expect the following reactions to be...
Ch. 18 - What change in temperature would make the process...Ch. 18 - Calculate SfforNH3(g). (Hint: Write the equation...Ch. 18 - Calculate the standard entropy change, S,inJK-1...Ch. 18 - Prob. 14PECh. 18 - Use the data in Table 6.2 and Table 18.1 to...Ch. 18 - Calculate for the reaction of iron(III) oxide...Ch. 18 - Calculate Greaction in kilojoules for the...Ch. 18 - Calculate the maximum work that could be obtained...Ch. 18 - Calculate the maximum work that could be obtained...Ch. 18 - The heat of vaporization of ammonia is and the...Ch. 18 - The heat of vaporization of mercury is 60.7...Ch. 18 - Use the data in Table 18.2 to determine whether...Ch. 18 - Use the data in Table 18.2 to determine whether we...Ch. 18 - In Examples 18.3 and 18.4 we computed SandH for...Ch. 18 - Use the data in Table 18.2 to determine for the...Ch. 18 - Calculate for the reaction described in the...Ch. 18 - In which direction will the reaction described in...Ch. 18 - The reaction...Ch. 18 - The reaction...Ch. 18 - Determine the heat of formation of gaseous...Ch. 18 - Determine the heat of formation of gaseous...Ch. 18 - First Law of Thermodynamics
18.1 What is the...Ch. 18 - First Law of Thermodynamics
18.2 State the first...Ch. 18 - First Law of Thermodynamics
18.3 How is a change...Ch. 18 - First Law of Thermodynamics
18.4 What is the...Ch. 18 - First Law of Thermodynamics Which quantities in...Ch. 18 - First Law of Thermodynamics Which thermodynamic...Ch. 18 - Second Law of Thermodynamics
18.7 What are the...Ch. 18 - First Law of Thermodynamics If there is a decrease...Ch. 18 - First Law of Thermodynamics Which of the following...Ch. 18 - Spontaneous Change What is a spontaneous change?...Ch. 18 - Spontaneous Change List five changes that you have...Ch. 18 - Spontaneous Change
18.12 Which of the items that...Ch. 18 - Spontaneous Change At constant pressure, what role...Ch. 18 - Spontaneous Change How do the probabilities of the...Ch. 18 - Entropy An instant cold pack purchased in a...Ch. 18 - Entropy What is entropy?Ch. 18 - Entropy How is the entropy of a substance affected...Ch. 18 - Entropy
18.18 Will the entropy change for each of...Ch. 18 - Entropy On the basis of our definition of entropy,...Ch. 18 - Second Law of Thermodynamics State the second law...Ch. 18 - Second Law of Thermodynamics How can a process...Ch. 18 - Second Law of Thermodynamics Explain the terms...Ch. 18 - Second Law of Thermodynamics Explain how the...Ch. 18 - Second Law of Thermodynamics
18.24 What is the...Ch. 18 - Second Law of Thermodynamics Define Gibbs free...Ch. 18 - Second Law of Thermodynamics
18.26 In terms of the...Ch. 18 - Second Law of Thermodynamics Under what...Ch. 18 - Third Law of Thermodynamics State the third law of...Ch. 18 - Third Law of Thermodynamics Explain why the units...Ch. 18 - Third Law of Thermodynamics Explain why the values...Ch. 18 - Third Law of Thermodynamics Would you expect the...Ch. 18 - Third Law of Thermodynamics Why does entropy...Ch. 18 - Third Law of Thermodynamics Does glass have S = 0...Ch. 18 - Standard Free Energy Change, G What is the...Ch. 18 - Standard Free Energy Change, G Why can G be...Ch. 18 - Maximum Work and G How is free energy related to...Ch. 18 - Maximum Work and
18.37 What is a...Ch. 18 - Maximum Work and G How is the rate at which energy...Ch. 18 - Maximum Work and
18.39 When glucose is oxidized...Ch. 18 - Maximum Work and G Why are real, observable...Ch. 18 - Free Energy and Equilibrium
18.41 In what way is...Ch. 18 - Free Energy and Equilibrium How can boiling points...Ch. 18 - Free Energy and Equilibrium Considering the fact...Ch. 18 - Free Energy and Equilibrium When a warm object is...Ch. 18 - Free Energy and Equilibrium Sketch the shape of...Ch. 18 - Free Energy and Equilibrium Many reactions that...Ch. 18 - Equilibrium Constants and
18.47 Suppose a...Ch. 18 - Equilibrium Constants and G Write the equation...Ch. 18 - Equilibrium Constants and G How is the equilibrium...Ch. 18 - Equilibrium Constants and
18.50 What is the value...Ch. 18 - Equilibrium Constants and
18.51 How does the...Ch. 18 - Bond Energies Define the term atomization enerey.Ch. 18 - Bond Energies Why are the heats of formation of...Ch. 18 - Bond Energies The gaseous C2 molecule has a bond...Ch. 18 - First Law of Thermodynamics
18.55 A certain system...Ch. 18 - First Law of Thermodynamics The value of E for a...Ch. 18 - 18.57 Suppose that you were pumping an automobile...Ch. 18 - 18.58 Consider the reaction between aqueous...Ch. 18 - Calculate H and E for the following reactions at...Ch. 18 - Calculate H and E for the following reactions at...Ch. 18 - The reaction 2N2O(g)2N2(g)+O2(g) has H=-163.14kJ....Ch. 18 - 18.62 A 10.0 L vessel at contains butane, , at a...Ch. 18 - Spontaneous Change Predict the sign of S for the...Ch. 18 - Spontaneous Change
18.64 Predict the sign of for...Ch. 18 - 18.65 Use the data from Table 6.2 to calculate ...Ch. 18 - Use the data from Table 6.2 to calculate H for the...Ch. 18 - Entropy There are two chemical systems, A and B,...Ch. 18 - Entropy
18.68 A chemical system has three panicles...Ch. 18 - Which system has a higher entropy? Explain your...Ch. 18 - Which system has a higher entropy?Ch. 18 - What factors must you consider to determine the...Ch. 18 - 18.72 What factors must you consider to determine...Ch. 18 - 18.73 Predict the algebraic sign of the entropy...Ch. 18 - 18.74 Predict the algebraic sign of the entropy...Ch. 18 - Second Law of Thermodynamics Under what conditions...Ch. 18 - Second Law of Thermodynamics
18.76 Under what...Ch. 18 - Third Law of Thermodynamics Calculate S for the...Ch. 18 - Third Law of Thermodynamics
18.78 Calculate for...Ch. 18 - Calculate Sfo for these compounds in J mol-1K-1....Ch. 18 - Calculate Sfo for these compounds in J mol-1K-1....Ch. 18 - Nitrogen dioxide, NO2, an air pollutant, dissolves...Ch. 18 - Good wine will turn to vinegar if it is left...Ch. 18 - Standard Free Energy Change, G Phosgene, COCl2,...Ch. 18 - Standard Free Energy Change, G Aluminum oxidizes...Ch. 18 - 18.85 Compute in kJ for the following reactions,...Ch. 18 - Prob. 86RQCh. 18 - Given the following,...Ch. 18 - *18.88 Given the following reactions and their ...Ch. 18 - Maximum Work and G Gasohol is a mixture of...Ch. 18 - Maximum Work and
18.90 What is the maximum amount...Ch. 18 - Free Energy and Equilibrium
18.91 Chloroform,...Ch. 18 - For the melting of aluminum,...Ch. 18 - Isooctane, a minor constituent of gasoline, has a...Ch. 18 - Acetone (nail polish remover) has a boiling point...Ch. 18 - 18.95 Determine whether the following reaction...Ch. 18 - Which of the following reactions (equations...Ch. 18 - Equilibrium Constants and G Calculate the value of...Ch. 18 - Equilibrium Constants and
18.98 Calculate the...Ch. 18 - 18.99 The reaction . A 1.00 L reaction vessel at ...Ch. 18 - The reaction...Ch. 18 - A reaction that can convert coal to methane (the...Ch. 18 - 18.102 One of the important reactions in living...Ch. 18 - What is the value of the equilibrium constant for...Ch. 18 - Methanol, a potential replacement for gasoline as...Ch. 18 - Bond Energies
18.105 Use the data in Table 18.4 to...Ch. 18 - 18.106 Approximately how much energy would be...Ch. 18 - 18.107 The standard heat of formation of ethanol...Ch. 18 - The standard heat of formation of ethylene,...Ch. 18 - Carbon disulfide, CS2, has the Lewis structure ,...Ch. 18 - Gaseous hydrogen sulfide, H2S,hasHf=20.15kJmol-1....Ch. 18 - 18.111 For . Use the data in Table 18.3 to...Ch. 18 - 18.112 Use the results of the preceding problem...Ch. 18 - Use the data in Tables 18.3 and 18.4 to estimate...Ch. 18 - What would be the approximate heat of formation of...Ch. 18 - *18.115 Which substance should have the more...Ch. 18 - Would you expect the value of Hf for benzene,...Ch. 18 - 18.117 Look at Table C.2 in Appendix C. Some of...Ch. 18 - Calculate the G for the dissolution of calcium...Ch. 18 - 18.119 If pressure is expressed in atmospheres and...Ch. 18 - 18.120 Calculate the work, in joules, done by a...Ch. 18 - When an ideal gas expands at a constant...Ch. 18 - When a real gas expands at a constant temperature,...Ch. 18 - 18.123 An ideal gas in a cylinder fitted with a...Ch. 18 - A cylinder fitted with a piston contains 5.00 L of...Ch. 18 - The experiment described in Exercise 18.124 is...Ch. 18 - When potassium iodide dissolves in water, the...Ch. 18 - The enthalpy of combustion Hcombustiono, of oxalic...Ch. 18 - Many biochemical reactions have positive values...Ch. 18 - *18.129 The reaction
has . Determine the value of...Ch. 18 - At 1500C,Kc=5.67 for the reaction...Ch. 18 - 18.131 Given the following reactions and their...Ch. 18 - *18.132 At room temperature , the gas ClNO is...Ch. 18 - *18.133 The reaction
has are placed in a 2.00 L...Ch. 18 - Use the data in Table 18.3 to calculate the bond...Ch. 18 - 18.135 The heat of vaporization of carbon...Ch. 18 - At 25C, 0.0560molO2and0.020molN2O were placed in a...Ch. 18 - For the substance SO2F2(g),Hfo=-858kJmol-1. The...Ch. 18 - *18.138 Ethyl alcohol, , has been suggested as an...Ch. 18 - When solutions of sodium hydroxide are used to...Ch. 18 - Prob. 140RQCh. 18 - A certain weak acid has a pKa of 5.83. When 100.0...Ch. 18 - The average CH bond energy calculated using the...Ch. 18 - 18.144 If a catalyst were able to affect the...Ch. 18 - At the beginning of this chapter we noted that the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
How do food chains and food webs differ? Which is the more accurate representation of feeding relationships in ...
Biology: Life on Earth with Physiology (11th Edition)
Analyzing ecological footprints reveals that (A) Earth's carrying capacity would increase if per capita meat co...
Campbell Biology (11th Edition)
63. A proton follows the path shown in Figure P21.63. Its initial speed is vo = 1.9 × 106 m/s. What is the prot...
College Physics: A Strategic Approach (3rd Edition)
3. What is free-fall, and why does it make you weightless? Briefly describe why astronauts are weightless in th...
The Cosmic Perspective (8th Edition)
12.1 Give the IUPAC name for each of the following:
a. CH3-CH2-OH
b.
c.
d.
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Did all the organisms living in or on the environments sampled grow on your nutrient agar? Briefly explain.
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Technetium is the first element in the periodic chart that does not have any stable isotopes. Technetium-99m is an especially interesting and valuable isotope as it emits a gamma ray with a half life ideally suited for medical tests. It would seem that the decay of technetium should fit the treatment above with the result In(c/c) = -kt. The table below includes data from the two sites: http://dailymed.nlm.nih.gov/dailymed/druginfo.cfm?id=7130 http://wiki.medpedia.com/Clinical: Neutrospec_(Technetium_(99m Tc)_fanolesomab). a. b. C. Graph the fraction (c/c.) on the vertical axis versus the time on the horizontal axis. Also graph In(c/c.) on the vertical axis versus time on the horizontal axis. When half of the original amount of starting material has hours fraction remaining disappeared, c/c = ½ and the equation In(c/c.) = -kt becomes In(0.5) = -kt1/2 where t₁₂ is the half life (the time for half of the material to decay away). Determine the slope of your In(c/c.) vs t graph and…arrow_forwardPlease correct answer and don't use hand ratingarrow_forward1. a) Assuming that an atom of arsenic has hydrogen-like atomic orbitals, sketch the radial probability plots for 4p and 4d orbitals of S atom. Indicate angular and radial nodes in these orbitals. (4 points) b) Calculate Zeff experienced by and electron in 4p AO's in a arsenic atom. Use Slater rules that were discussed in lecture. (3 points)arrow_forward
- What is the pH of the Tris buffer after the addition of 10 mL of 0.01M NaOH? How would I calculate this?arrow_forwardWhy do isopolianions form polymeric species with a defined molecular weight? What does it depend on?arrow_forwardWhat are isopolianions? Describe the structural unit of isopolianions.arrow_forward
- Justify the polymerization of vanadates VO43-, as a function of concentration and pH.arrow_forwardWhat is the preparation of 500 mL of 100mM MOPS buffer (pH=7.5) starting with 1 M MOPS and 1 M NaOH? How would I calculate the math?arrow_forwardIndicate the correct option.a) Isopolianions are formed around metallic atoms in a low oxidation state.b) Non-metals such as N, S, C, Cl, ... give rise to polyacids (oxygenated).c) Both are incorrect.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning  Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY