
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 13ALQ
Consider the following galvanic cell:
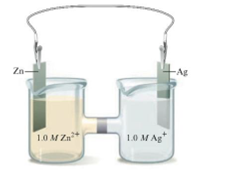
What happens to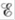 as the concentration of Zn2+ is increased? As the concentration of Ag+ is increased? What happens to
as the concentration of Zn2+ is increased? As the concentration of Ag+ is increased? What happens to 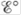 in these cases?
in these cases?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
help
The temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the
pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes.
pressure (atm)
2
0
0
200
400
temperature (K)
X
QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes
*The values are all provided in the photo attached*
Chapter 18 Solutions
Chemistry
Ch. 18 - What is a half-reaction? Why must the number of...Ch. 18 - Galvanic cells harness spontaneous...Ch. 18 - Table 17-1 lists common half-reactions along with...Ch. 18 - Consider the equation G = -nF. What are the four...Ch. 18 - The Nernst equation allows determination of the...Ch. 18 - What are concentration cells? What is in a...Ch. 18 - Prob. 7RQCh. 18 - Prob. 8RQCh. 18 - What characterizes an electrolytic cell? What is...Ch. 18 - Prob. 1ALQ
Ch. 18 - When balancing reactions in Chapter 3, we did not...Ch. 18 - Sketch a galvanic cell, and explain how it works....Ch. 18 - In making a specific galvanic cell, explain how...Ch. 18 - Prob. 5ALQCh. 18 - Prob. 6ALQCh. 18 - Sketch a cell that forms iron metal from iron(II)...Ch. 18 - Which of the following is the best reducing agent:...Ch. 18 - You are told that metal A is a better reducing...Ch. 18 - Explain the following relationships: G and w, cell...Ch. 18 - Explain why cell potentials are not multiplied by...Ch. 18 - What is the difference between and ? When is equal...Ch. 18 - Consider the following galvanic cell: What happens...Ch. 18 - Look up the reduction potential for Fe3+ to Fe2+....Ch. 18 - If the cell potential is proportional to work and...Ch. 18 - Is the following statement true or false?...Ch. 18 - Define oxidation and reduction in terms of both...Ch. 18 - Assign oxidation numbers to all the atoms in each...Ch. 18 - Specify which of the following equations represent...Ch. 18 - The Ostwald process for the commercial production...Ch. 18 - Balance the following oxidation-reduction...Ch. 18 - Balance the following oxidation-reduction...Ch. 18 - What is electrochemistry? What are redox...Ch. 18 - Prob. 24QCh. 18 - When magnesium metal is added to a beaker of...Ch. 18 - How can one construct a galvanic cell from two...Ch. 18 - The free energy change for a reaction, G, is an...Ch. 18 - What is wrong with the following statement: The...Ch. 18 - When jump-starting a car with a dead battery, the...Ch. 18 - In theory, most metals should easily corrode in...Ch. 18 - Consider the electrolysis of a molten salt of some...Ch. 18 - Consider the following electrochemical cell: a. If...Ch. 18 - Prob. 33QCh. 18 - Prob. 34QCh. 18 - Consider the following galvanic cell: Label the...Ch. 18 - Consider the following galvanic cell: a. Label the...Ch. 18 - Sketch the galvanic cells based on the following...Ch. 18 - Sketch the galvanic cells based on the following...Ch. 18 - Calculate values for the galvanic cells in...Ch. 18 - Calculate values for the galvanic cells in...Ch. 18 - Sketch the galvanic cells based on the following...Ch. 18 - Sketch the galvanic cells based on the following...Ch. 18 - Give the standard line notation for each cell in...Ch. 18 - Give the standard line notation for each cell in...Ch. 18 - Consider the following galvanic cells: For each...Ch. 18 - Give the balanced cell equation and determine for...Ch. 18 - Calculate values for the following g cells. Which...Ch. 18 - Calculate values for the following cells. Which...Ch. 18 - Chlorine dioxide (C1O2), which is produced by the...Ch. 18 - The amount of manganese in steel is determined by...Ch. 18 - Calculate the maximum amount of work that can be...Ch. 18 - Calculate the maximum amount of work that can be...Ch. 18 - Estimate for the half-reaction 2H2O+2eH2+2OH given...Ch. 18 - The equation G = nF also can be applied to...Ch. 18 - Glucose is the major fuel for most living cells....Ch. 18 - Direct methanol fuel cells (DMFCs) have shown some...Ch. 18 - Using data from Table 17-1, place the following in...Ch. 18 - Using data from Table 17-1, place the following in...Ch. 18 - Answer the following questions using data from...Ch. 18 - Answer the following questions using data from...Ch. 18 - Consider only the species (at standard conditions)...Ch. 18 - Prob. 62ECh. 18 - Use the table of standard reduction potentials...Ch. 18 - Consider the concentration cell in Fig. 17-10. If...Ch. 18 - Consider the concentration cell shown below....Ch. 18 - Consider a concentration cell similar to the one...Ch. 18 - The overall reaction in the lead storage battery...Ch. 18 - Calculate the pH of the cathode compartment for...Ch. 18 - Consider the cell described below:...Ch. 18 - Consider the cell described below:...Ch. 18 - Calculate G and K at 25C for the reactions in...Ch. 18 - Calculate G and K at 25C for the reactions in...Ch. 18 - Consider the galvanic cell based on the following...Ch. 18 - Consider the galvanic cell based on the following...Ch. 18 - An electrochemical cell consists of a standard...Ch. 18 - Prob. 78ECh. 18 - An electrochemical cell consists of a standard...Ch. 18 - An electrochemical cell consists of a nickel metal...Ch. 18 - Consider a concentration cell that has both...Ch. 18 - You have a concentration cell in which the cathode...Ch. 18 - Under standard conditions, what reaction occurs,...Ch. 18 - A disproportionation reaction involves a substance...Ch. 18 - Consider the following galvanic cell at 25C:...Ch. 18 - An electrochemical cell consists of a silver metal...Ch. 18 - Cadmium sulfide is used in some semiconductor...Ch. 18 - For the following half-reaction, = 2.07 V:...Ch. 18 - Calculate for the following half-reaction:...Ch. 18 - The solubility product for CuI(s) is 1.1 102...Ch. 18 - How long will it take to plate out each of the...Ch. 18 - The electrolysis of BiO+ produces pure bismuth....Ch. 18 - What mass of each of the following substances can...Ch. 18 - Aluminum is produced commercially by the...Ch. 18 - Electrolysis of an alkaline earth metal chloride...Ch. 18 - What volume of F2 gas, at 25C and 1.00 atm, is...Ch. 18 - What volumes of H2(g) and O2(g) at STP are...Ch. 18 - A single HallHeroult cell (as shown in Fig. 17-22)...Ch. 18 - A factory wants to produce 1.00 103 kg barium...Ch. 18 - It took 2.30 min using a current of 2.00 A to...Ch. 18 - A solution containing Pt4+ is electrolyzed with a...Ch. 18 - A solution at 25C contains 1.0 M Cd2+, 1.0 M Ag+,...Ch. 18 - A solution at 25C contains 1.0 M Cu2 and 1.0 104...Ch. 18 - In the electrolysis of an aqueous solution of...Ch. 18 - Copper can be plated onto a spoon by placing the...Ch. 18 - Prob. 107ECh. 18 - Prob. 108ECh. 18 - What reactions take place at the cathode and the...Ch. 18 - What reaction will take place at the Cathode and...Ch. 18 - The saturated calomel electrode. abbreviated SCE....Ch. 18 - Consider the following half-reactions: Explain why...Ch. 18 - Consider the standard galvanic cell based on the...Ch. 18 - A standard galvanic cell is constructed so that...Ch. 18 - The black silver sulfide discoloration of...Ch. 18 - Prob. 116AECh. 18 - When aluminum foil is placed in hydrochloric acid,...Ch. 18 - Prob. 118AECh. 18 - Prob. 119AECh. 18 - Prob. 120AECh. 18 - A fuel cell designed to react grain alcohol with...Ch. 18 - The overall reaction and equilibrium constant...Ch. 18 - Prob. 123AECh. 18 - The overall reaction and standard cell potential...Ch. 18 - Prob. 125AECh. 18 - The ultimate electron acceptor in the respiration...Ch. 18 - One of the few industrial-scale processes that...Ch. 18 - It took 150. s for a current of 1.25 A to plate...Ch. 18 - Prob. 129AECh. 18 - In the electrolysis of a sodium chloride solution,...Ch. 18 - An aqueous solution of an unknown salt of...Ch. 18 - Which of the following statement(s) is/are true?...Ch. 18 - Consider a galvanic cell based on the following...Ch. 18 - Prob. 134CWPCh. 18 - Consider a galvanic cell based on the following...Ch. 18 - An electrochemical cell consists of a silver metal...Ch. 18 - An aqueous solution of PdCl2 is electrolyzed for...Ch. 18 - Consider the following half-reactions:...Ch. 18 - Consider the following reduction potentials: Co3++...Ch. 18 - Calculate and G for the reaction 2H2O(l) 2H2(g)...Ch. 18 - Prob. 141CPCh. 18 - The overall reaction in the lead storage battery...Ch. 18 - Consider the following galvanic cell: Calculate...Ch. 18 - A zinc-copper battery is constructed at follows at...Ch. 18 - A galvanic cell is based on the following...Ch. 18 - Consider a cell based on the following...Ch. 18 - Prob. 147CPCh. 18 - You have a concentration cell with Cu electrodes...Ch. 18 - A galvanic cell is based on the following...Ch. 18 - Given the following two standard reduction...Ch. 18 - Consider the following galvanic cell: Calculate...Ch. 18 - Prob. 152CPCh. 18 - Consider the following galvanic cell: A 15 0-mole...Ch. 18 - When copper reacts with nitric acid, a mixture of...Ch. 18 - The following standard reduction potentials have...Ch. 18 - An electrochemical cell is set up using the...Ch. 18 - Three electrochemical cells were connected in...Ch. 18 - A silver concentration cell is set up at 25C as...Ch. 18 - A galvanic cell is based on the following...Ch. 18 - The table below lists the cell potentials for the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
An aluminum calorimeter with a mass of 100 g contains 250 g of water. The calorimeter and water are in thermal ...
Physics for Scientists and Engineers
2. Why is it that the range of resting blood pressures of humans is best represented by a bell-shaped curve co...
Human Biology: Concepts and Current Issues (8th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
What process causes the Mediterranean intermediate Water MIW to become more dense than water in the adjacent At...
Applications and Investigations in Earth Science (9th Edition)
60. The solar system is 25,000 light years from the center of our Milky Way galaxy. One light year is the dista...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- pressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward5.arrow_forward
- 6.arrow_forward0/5 alekscgi/x/sl.exe/1o_u-IgNglkr7j8P3jH-IQs_pBaHhvlTCeeBZbufuBYTi0Hz7m7D3ZcSLEFovsXaorzoFtUs | AbtAURtkqzol 1HRAS286, O States of Matter Sketching a described thermodynamic change on a phase diagram The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 3 pressure (atm) + 0- 0 5+ 200 temperature (K) 400 Explanation Check X 0+ F3 F4 F5 F6 F7 S 2025 McGraw Hill LLC All Rights Reserved. Terms of Use Privacy Center Accessibility Q Search LUCR + F8 F9 F10 F11 F12 * % & ( 5 6 7 8 9 Y'S Dele Insert PrtSc + Backsarrow_forward5.arrow_forward
- 9arrow_forwardalekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZS18w-nDB10538ZsAtmorZoFusYj2Xu9b78gZo- O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 3- 200 temperature (K) Explanation Chick Q Sowncharrow_forward0+ aleksog/x/lsl.exe/1ou-lgNgkr7j8P3H-IQs pBaHhviTCeeBZbufuBYTOHz7m7D3ZStEPTBSB3u9bsp3Da pl19qomOXLhvWbH9wmXW5zm O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 Gab The temperature on a sample of pure X held at 0.75 atm and -229. °C is increased until the sample sublimes. The temperature is then held constant and the pressure is decreased by 0.50 atm. On the phase diagram below draw a path that shows this set of changes. F3 pressure (atm) 0- 0 200 Explanation temperature (K) Check F4 F5 ☀+ Q Search Chill Will an 9 ENG F6 F7 F8 F9 8 Delete F10 F11 F12 Insert PrtSc 114 d Ararrow_forward
- x + LEKS: Using a phase diagram a X n/alekscgi/x/lsl.exe/10_u-IgNsikr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpw ○ States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 1.1 atm. pressure (atm) 16 08- solid liquid- 0 200 400 gas 600 temperature (K) Note: your answer must be within 25 °C of the exact answer to be graded correct. × 5arrow_forwardS: Using a phase diagram leksogi/x/sl.exe/1ou-IgNs kr 7j8P3jH-IQs_pBan HhvTCeeBZbufuBYTI0Hz7m7D3ZdHYU+80XL-5alyVp O States of Matter Using a phase diagram to find a phase transition temperature or pressure se the phase diagram of Substance X below to find the boiling point of X when the pressure on the liquid is 1.6 atm. pressure (atm) 32- 16- solid liquid 0. gas 100 200 temperature (K) 300 Note: your answer must be within 12.5 °C of the exact answer to be graded correct. 10 Explanation Check § Q Search J 2025 McGraw Hill LLC. All Rights Researrow_forward151.2 254.8 85.9 199.6 241.4 87.6 242.5 186.4 155.8 257.1 242.9 253.3 256.0 216.6 108.7 239.0 149.7 236.4 152.1 222.7 148.7 278.2 268.7 234.4 262.7 283.2 143.6 QUESTION: Using this group of data on salt reduced tomato sauce concentration readings answer the following questions: 1. 95% Cl Confidence Interval (mmol/L) 2. [Na+] (mg/100 mL) 3. 95% Na+ Confidence Interval (mg/100 mL)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Introduction to Electrochemistry; Author: Tyler DeWitt;https://www.youtube.com/watch?v=teTkvUtW4SA;License: Standard YouTube License, CC-BY