
Concept explainers
(a)
Interpretation: The IUPAC name of the following compound should be determined:
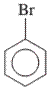
Concept Introduction: The ring structures of the compound having uncommon stability due to delocalized pi electron density shared in between all the carbon atoms of the ring is said to be an aromatic compound.
In order to give the name to the multiple substituted
- For single substituted aromatic compound (when the substituent contains six or fewer carbons), the name of the substituted group is written first followed by the name of the aromatic compound.
- For single substituted aromatic compound (when the substituent contains more than six carbons), the name of the aromatic compound is written first followed by the name of the substituted group.
- For single substituted aromatic compound, the numbering on the ring is done in such a way that the multiple substituents get the lowest number.
(b)
Interpretation: The IUPAC name of the following compound should be determined:
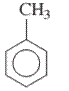
Concept Introduction: The ring structures of the compound having uncommon stability due to delocalized pi electron density shared in between all the carbon atoms of the ring is said to be an aromatic compound.
In order to give the name to the multiple substituted aromatic compounds, the following steps are followed:
- For single substituted aromatic compound (when the substituent contains six or fewer carbons), the name of the substituted group is written first followed by the name of the aromatic compound.
- For single substituted aromatic compound (when the substituent contains more than six carbons), the name of the aromatic compound is written first followed by the name of the substituted group.
- For single substituted aromatic compound, the numbering on the ring is done in such a way that the multiple substituents get the lowest number.
(c)
Interpretation: The IUPAC name of the following compound should be determined:
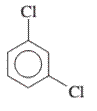
Concept Introduction: The ring structures of the compound having uncommon stability due to delocalized pi electron density shared in between all the carbon atoms of the ring is said to be an aromatic compound.
In order to give the name to the multiple substituted aromatic compounds, the following steps are followed:
- For single substituted aromatic compound (when the substituent contains six or fewer carbons), the name of the substituted group is written first followed by the name of the aromatic compound.
- For single substituted aromatic compound (when the substituent contains more than six carbons), the name of the aromatic compound is written first followed by the name of the substituted group.
- For single substituted aromatic compound, the numbering on the ring is done in such a way that the multiple substituents get the lowest number.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EP BASIC CHEMISTRY-STANDALONE ACCESS
- Hello, I am doing a court case analysis in my Analytical Chemistry course. The case is about a dog napping and my role is prosecution of the defendant. I am tasked in the Area of Expertise in Neutron Activation and Isotopic Analysis. Attached is the following case study reading of my area of expertise! The landscaping stone was not particularly distinctive in its decoration but matched both the color and pattern of the Fluential’s landscaping stone as well as the stone in the back of the recovered vehicle. Further analysis of the stone was done using a technique called instrumental neutron activation analysis. (Proceed to Neutron Activation data) Photo Notes: Landscaping stone recovered in vehicle. Stone at Fluential’s home is similar inappearance. Finally, the white paint on the brick was analyzed using stable isotope analysis. The brick recovered at the scene had smeared white paint on it. A couple of pieces of brick in the back of the car had white paint on them. They…arrow_forwardCite the stability criteria of an enamine..arrow_forwardCalculate the pH of a 0.01m solution of acetic acid use pka of 4.75arrow_forward
- What is the product of the reaction? F3C. CF3 OMe NaOH / H₂Oarrow_forwardWhat is the product of the reaction? F3C. CF3 OMe NaOH / H₂Oarrow_forwardWhat would you expect to be the major product obtained from the following reaction? Please explain what is happening here. Provide a detailed explanation and a drawing showing how the reaction occurs. The correct answer to this question is V.arrow_forward
- Please answer the question for the reactions, thank youarrow_forwardWhat is the product of the following reaction? Please include a detailed explanation of what is happening in this question. Include a drawing showing how the reagent is reacting with the catalyst to produce the correct product. The correct answer is IV.arrow_forwardPlease complete the reactions, thank youarrow_forward
- Consider the synthesis. What is compound Y? Please explain what is happening in this question. Provide a detailed explanation and a drawing to show how the compound Y creates the product. The correct answer is D.arrow_forwardWhat would be the major product of the following reaction? Please include a detailed explanation of what is happening in this question. Include steps and a drawing to show this reaction proceeds and how the final product is formed. The correct answer is B. I put answer D and I don't really understand what is going on in the question.arrow_forwardWhat is the product of the following reaction? Please explain what is happening in this question. Provide a detailed explanation and a drawing showing how the reagent is reacting with the catalysts to product the correct product. The correct answer is B.arrow_forward

 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




