
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
8th Edition
ISBN: 9780135213711
Author: Paula Bruice
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 91P
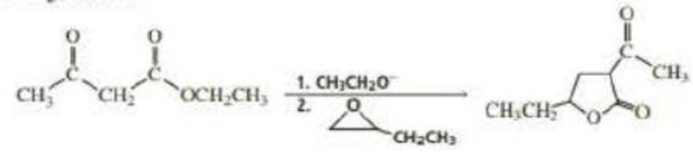
Propose a mechanism for the following reaction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a tetramer of this alternating copolymer.
H
I
T
H
HH
H
-H
C.
H-
Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above.
H-
H
CIH
H
H
H
HHHH
H
H
0
·H
H–
冊
CH
CHI
HH C-
H-
H
H-
H
H
A.
H
H C
H
H-
-H
HH
H
B.
H-
-H
D.
H
H H
H
•
H
-H
E.
-H
H
H
HICH
T
HHH
F.
H-
Polylactic acid (shown below) is a biodegradable polymer used for food packaging. Identify the monomer(s) used in the production of this polymer using a condensation process.
Chapter 17 Solutions
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
Ch. 17.1 - Prob. 1PCh. 17.1 - Prob. 2PCh. 17.1 - Prob. 3PCh. 17.1 - Prob. 4PCh. 17.1 - Rank the compounds in each of the following groups...Ch. 17.2 - Explain why 92% of 2,4-pemtanedione exists as the...Ch. 17.3 - Draw the enol tautomers for each of the following...Ch. 17.3 - Prob. 8PCh. 17.4 - Prob. 9PCh. 17.4 - Prob. 10P
Ch. 17.5 - Show how the following compounds can be prepared...Ch. 17.6 - What compound is formed when a dilute solution of...Ch. 17.7 - Prob. 13PCh. 17.7 - Prob. 14PCh. 17.7 - How many stereoisomers are obtained from each of...Ch. 17.7 - Prob. 16PCh. 17.8 - Prob. 17PCh. 17.9 - Prob. 18PCh. 17.9 - What reagents should be used to prepare the...Ch. 17.10 - Prob. 20PCh. 17.10 - What aldehyde or ketone would be obtained when...Ch. 17.11 - Prob. 22PCh. 17.11 - How could you prepare the following compound using...Ch. 17.12 - Prob. 25PCh. 17.12 - What two carbonyl compound are required for the...Ch. 17.12 - Propose a mechanism for the following reaction:Ch. 17.13 - Draw the products of the following reactions:Ch. 17.13 - Prob. 29PCh. 17.13 - Prob. 30PCh. 17.14 - Prob. 31PCh. 17.15 - Write the mechanism for the reaction of a...Ch. 17.15 - Prob. 33PCh. 17.15 - Prob. 34PCh. 17.15 - Draw the product of the reaction of each of the...Ch. 17.16 - Draw the product obtained by heating each pair of...Ch. 17.16 - What two carbonyl compounds are needed to...Ch. 17.17 - Prob. 38PCh. 17.18 - Prob. 39PCh. 17.18 - Prob. 40PCh. 17.19 - Prob. 41PCh. 17.20 - Prob. 43PCh. 17.21 - Propose a mechanism for the formation of...Ch. 17.21 - Prob. 45PCh. 17.21 - a. If the biosynthesis of palmitic acid were...Ch. 17.21 - Prob. 47PCh. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Number the following compounds in order of...Ch. 17 - The 1H NMR chemical shifts of nitromethane,...Ch. 17 - Prob. 52PCh. 17 - Draw the products of the following reactions: a....Ch. 17 - A racemic mixture of 2-methyl-1-phenyl-1-butanone...Ch. 17 - Draw the products of the following reaction:Ch. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - In the presence of excess base and excess halogen,...Ch. 17 - Identify A-L. (Hint: A shows three singles in its...Ch. 17 - Using cyclopentanone as the reactant, show the...Ch. 17 - Show how 4-methyl-3-hexanol can be synthesized...Ch. 17 - Show how the following compound can be synthesized...Ch. 17 - Show how the following compounds can be prepared...Ch. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Indicate how each of the following compounds can...Ch. 17 - Prob. 67PCh. 17 - The ketone whose 1H NMR spectrum is shown here was...Ch. 17 - Indicate how the following compounds can be...Ch. 17 - Compound A with molecular formula C6H10 has two...Ch. 17 - Prob. 71PCh. 17 - Draw the products of the following reactions:Ch. 17 - Prob. 73PCh. 17 - a. Show how the amino acid alanine can be...Ch. 17 - Show how the following compounds can be...Ch. 17 - Prob. 76PCh. 17 - Explain why the following bromoketone forms...Ch. 17 - Prob. 78PCh. 17 - A carboxylic arid is formed when an -haloketone...Ch. 17 - An , -unsaturated carbonyl compound can be...Ch. 17 - What carbonyl compounds are required to prepare a...Ch. 17 - Prob. 82PCh. 17 - A Cannizzaro reaction is the reaction of an...Ch. 17 - Propose a mechanism for each of the following...Ch. 17 - The following reaction is known as the benzoni...Ch. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Prob. 89PCh. 17 - Prob. 90PCh. 17 - Propose a mechanism for the following reaction:Ch. 17 - What reagents are required to convert the reactant...Ch. 17 - Starting with bromocyclohexane, how can each of...Ch. 17 - Describe how the following compounds can be...Ch. 17 - Prob. 4PCh. 17 - Describe three ways to synthesize the following...Ch. 17 - Explain why 92% of 2.4-pentanedione exists as the...Ch. 17 - Describe how the following compound can be...Ch. 17 - Prob. 8PCh. 17 - Prob. 9PCh. 17 - Prob. 10PCh. 17 - Show how the following compounds can be...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the product of the reaction shown below. Ignore small byproducts that would evaporate pleasearrow_forwardPoly(ethylene adipate) is a biodegradable polyester (shown below). Identify the type of polymerization process used in the production of this polymer.arrow_forwardPolymers may be composed of thousands of monomers. draw two repeat units(dimer) of the polymer formed in this reaction. assume there are hydrogen atoms on the two ends of the dimer. ignore inorganic byproducts pleasearrow_forward
- Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore inorganic byproductsarrow_forwardDraw the product of this reaction please. Ignore inorganic byproductsarrow_forwardOne of the pi molecular orbitals of 1,3-butadiene (CH2=CHCH=CH2) is shown below. Please identify the number of nodal planes perpendicular to the bonding axisarrow_forward
- Draw the monomers required to synthesize this condensation polymer please.arrow_forwardProvide the correct systematic name for the compound shown here. Please take into account the keyboard options belowarrow_forwardcurved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s)arrow_forward
- Identify the 'cartoon' drawing of the acceptor orbital in the first mechanistic step of an electrophilic addition reaction of butadiene with HBr. Pleasearrow_forwardH- H H H H H H Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H H H A. H H H H-C CI H H D. H H H H H H C C -H H C C H H H H B. H CI H H- C C H H H H E. H CI H C.arrow_forwardWhy doesn't this carry on to form a ring by deprotonating the alpha carbon and the negatively-charged carbon attacking the C=O?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License