
Conceptual Physical Science (6th Edition)
6th Edition
ISBN: 9780134060491
Author: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 50E
Use the following illustration to answer Exercises 49-51.
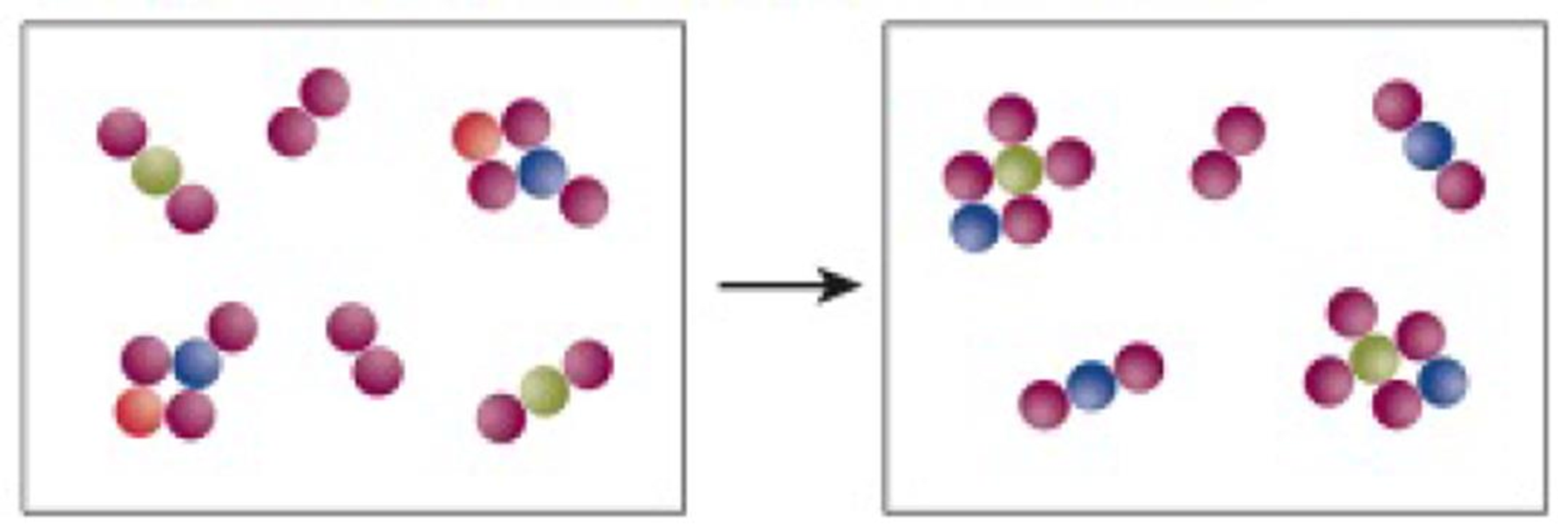
50. There is an excess of at least one of the reactant molecules. Which one?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A fluid with density 263 kg/m3 flows through a pipe of varying diameter and height. At location 1 the flow speed is 13.5 m/s and the diameter of the pipe is 7.4 cm down to location 2 the pipe diameter is 16.9 cm. Location 1 is 6.3 meters higher than location 2.
What is the difference in pressure P2 - P1?
Using units in Pascals and use g = 9.81 m/s2.
The kitchen had a temperature 46 degrees Fahrenheit and was converted it to Kelvin. What is the correct number for this temperature (46 F) on the Kelvin scale?
Water is traveling at a speed of 0.65 m/s through a pipe with a cross-section radius of 0.23 meters. The water enters a section of pipe that has a smaller radius, only 0.11 meters. What is the speed of the water traveling in this narrower section of pipe?
Chapter 17 Solutions
Conceptual Physical Science (6th Edition)
Ch. 17 - Prob. 1RCQCh. 17 - Prob. 2RCQCh. 17 - Prob. 3RCQCh. 17 - Prob. 4RCQCh. 17 - Why dont equal masses of golf balls and Ping-Pong...Ch. 17 - Why dont equal masses of carbon atoms and oxygen...Ch. 17 - Prob. 7RCQCh. 17 - If you had 1 mole of marbles, how many marbles...Ch. 17 - Prob. 9RCQCh. 17 - How many molecules of water are there in 18g of...
Ch. 17 - Why is saying you have 1 mole of water molecules...Ch. 17 - What is the mass of an oxygen atom in atomic mass...Ch. 17 - Prob. 13RCQCh. 17 - What generally happens to the rate of a chemical...Ch. 17 - Which reactant molecules are the first to pass...Ch. 17 - What term is used to describe the minimum amount...Ch. 17 - What catalyst is effective in the destruction of...Ch. 17 - Prob. 18RCQCh. 17 - What net effect does a chemical reaction have on a...Ch. 17 - Why are catalysts so important to our economy?Ch. 17 - Prob. 21RCQCh. 17 - What is released by an exothermic reaction?Ch. 17 - What is absorbed by an endothermic reaction?Ch. 17 - As energy disperses, where does it go?Ch. 17 - What is always increasing?Ch. 17 - Prob. 26RCQCh. 17 - Show that there are 1.0 1022 carbon atoms in a...Ch. 17 - How many gold atoms are there in a 5.00-g sample...Ch. 17 - Show that 1 mole of KClO3 contains 122.55 g.Ch. 17 - Prob. 35TASCh. 17 - Prob. 36TASCh. 17 - Show that the formula mass of 2-propanol, C3H8O,...Ch. 17 - Prob. 38TASCh. 17 - Prob. 39TASCh. 17 - Prob. 40TASCh. 17 - Prob. 41TASCh. 17 - Prob. 43TARCh. 17 - Rank the following in order of increasing entropy:...Ch. 17 - Prob. 46ECh. 17 - Prob. 47ECh. 17 - Prob. 48ECh. 17 - Prob. 49ECh. 17 - Use the following illustration to answer Exercises...Ch. 17 - Use the following illustration to answer Exercises...Ch. 17 - The reactants shown schematically on the left...Ch. 17 - Prob. 53ECh. 17 - Which has more atoms: 17.031 grams of ammonia,...Ch. 17 - How many moles of molecules are there in each of...Ch. 17 - How many moles of atoms are there in each of the...Ch. 17 - Prob. 58ECh. 17 - Prob. 59ECh. 17 - Which has the greater mass, 1.204 1024 molecules...Ch. 17 - Prob. 61ECh. 17 - How many atoms of arsenic are there in a 145-gram...Ch. 17 - How is it possible for a jet airplane carrying 110...Ch. 17 - In the laboratory, endothermic reactions are...Ch. 17 - Prob. 65ECh. 17 - Why does a glowing splint of wood burn only slowly...Ch. 17 - Prob. 67ECh. 17 - Explain the connection between photosynthetic life...Ch. 17 - Does the ozone pollution from automobiles help...Ch. 17 - Chlorine is put into the atmosphere by volcanoes...Ch. 17 - Prob. 71ECh. 17 - Prob. 72ECh. 17 - Exothermic reactions are favored because they...Ch. 17 - Prob. 74ECh. 17 - Prob. 75ECh. 17 - What role does entropy play in chemical reactions?Ch. 17 - Prob. 77ECh. 17 - Under what conditions will a hot pie not lose heat...Ch. 17 - As the Sun shines on a snow-capped mountain, much...Ch. 17 - Wild plants readily grow all by themselves, yet...Ch. 17 - Prob. 81DQCh. 17 - Prob. 1RATCh. 17 - Which has the greatest number of atoms? (a) 28 g...Ch. 17 - Prob. 4RATCh. 17 - The yeast in bread dough feeds on sugar to produce...Ch. 17 - What can you deduce about the activation energy of...Ch. 17 - What role do CFCs play in the catalytic...Ch. 17 - Prob. 8RATCh. 17 - How much energy, in kilojoules, is released or...Ch. 17 - Prob. 10RAT
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why is petroleum jelly used in the hanging-drop procedure?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
What type of cut would separate the brain into anterior and posterior parts?
Anatomy & Physiology (6th Edition)
SCIENTIFIC INQUIRY You are handed a mystery pea plant with tall stems and axial flowers and asked to determine ...
Campbell Biology (11th Edition)
Police Captain Jeffers has suffered a myocardial infarction. a. Explain to his (nonmedically oriented) family w...
Human Physiology: An Integrated Approach (8th Edition)
What are the minimum and maximum ages of the island of Kauai? Minimum age: ______million yr Maximum age: ______...
Applications and Investigations in Earth Science (9th Edition)
Explain all answer clearly, with complete sentences and proper essay structure if needed. An asterisk (*) desig...
Cosmic Perspective Fundamentals
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- A particular water pipe has a radius of 0.28 meters. If the pipe is completely filled with water, moving with average velocity 0.45 m/s, what is the flow rate of water through the pipe with units of cubic meters of water per second?arrow_forwardWater is flowing through a horizontal pipe with two segments. In one segment, the water flows at a speed v1 = 4.52 m/s. In the second segment the speed of the water is v2 = 2.38 m/s. Based on Bernoulli's Principle, what is the difference in pressure (P2 - P1) between the two segments? Assume that the density of the water is 997 kg/m3 and give your answer as the number of Pascals (i.e. N/m2).arrow_forwardWater from the faucet is supplied to the hose at a rate of 0.00057 m3/s. At what speed (number of meters per second) does the water exit the nozzle if the cross sectional area of the narrow nozzle is 2.1 x 10-6 m2?arrow_forward
- Jason Fruits/Indiana University Research Communications Silver/ silver oxide Zinc zinc/oxidearrow_forwardCar P moves to the west with constant speed v0 along a straight road. Car Q starts from rest at instant 1, and moves to the west with increasing speed. At instant 5, car Q has speed w0 relative to the road (w0 < v0). Instants 1-5 are separated by equal time intervals. At instant 3, cars P and Q are adjacent to one another (i.e., they have the same position). In the reference frame o f the road, at instant 3 i s the speed o f car Q greater than, less than, or equal to the speed of car P? Explain.arrow_forwardCar P moves to the west with constant speed v0 along a straight road. Car Q starts from rest at instant 1, and moves to the west with increasing speed. At instant 5, car Q has speed w0 relative to the road (w0 < v0). Instants 1-5 are separated by equal time intervals.arrow_forward
- Car P moves to the west with constant speed v0 along a straight road. Car Q starts from rest at instant 1, and moves to the west with increasing speed. At instant 5, car Q has speed w0 relative to the road (w0 < v0). Instants 1-5 are separated by equal time intervals. Sketch and label a vector diagram illustrating the Galilean transformation of velocities that relates velocity of car P relative to the road, velocity of car Q relative to road, and velocity of car Q relative to car P at instant 3. In the frame of car P, at instant 3 is car Q moving to the west, moving to the east, or at rest? Explain.arrow_forwardJust 5 and 6 don't mind 7arrow_forwardIn an electron gun, electrons are accelerated through a region with an electric field of magnitude 1.5 × 104 N/C for a distance of 2.5 cm. If the electrons start from rest, how fast are they moving after traversing the gun?arrow_forward
- Please solve and answer this problem correctly please. Thank you!!arrow_forwardPlease solve and answer this problem correctly please. Thank you!!arrow_forwarda) Use the node-voltage method to find v1, v2, and v3 in the circuit in Fig. P4.14. b) How much power does the 40 V voltage source deliver to the circuit? Figure P4.14 302 202 w w + + + 40 V V1 80 Ω 02 ΣΑΩ 28 A V3 + w w 102 202arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning


University Physics Volume 3
Physics
ISBN:9781938168185
Author:William Moebs, Jeff Sanny
Publisher:OpenStax

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY