
Concept explainers
(a)
Interpretation:
The ratio of A- to [HA] with the help of pH and pKa values should be calculated.
Concept Introduction:
The pH of a solution indicates the number of H3 O+ ions in the solution. The mathematical relation between pH and H3 O+ is as given below;.
Acid dissociation constant Ka is related to pKa as;.
Answer to Problem 30P
Explanation of Solution
Given Information:
pKa = 5.0.
pH = 2.0.
Calculation:
From pH of solution, concentration of hydroxide ion can be calculated as follows:
Also,
Since,
Thus, by putting the values, the ratio
(b)
Interpretation:
The ratio of A- to [HA] with the help of pH and pKa values should be calculated.
Concept Introduction:
The pH of a solution indicates the number of H3 O+ ions in the solution. The mathematical relation between pH and H3 O+ is as given below;.
Acid dissociation constant Ka is related to pKa as;.
Answer to Problem 30P
Given:
pKa = 5.0.
pH = 5.0.
Hence [H3 O+ ] = 10-pH = 10-5.
And Ka = 10-pKa = 10-5.
Since 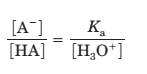
Plug the values of Ka and H3 O+ ions to calculate the ratio of [A- ] to [HA].
[A- ] / [HA] = 10-5 / 10-5 = 1.
Explanation of Solution
Given Information:
pKa = 5.0.
pH = 5.0.
Calculation:
From pH of solution, concentration of hydroxide ion can be calculated as follows:
Also,
Since,
Thus, by putting the values, the ratio
(c)
Interpretation:
The ratio of A- to [HA] with the help of pH and pKa values should be calculated.
Concept Introduction:
The pH of a solution indicates the number of H3 O+ ions in the solution. The mathematical relation between pH and H3 O+ is as given below;.
Acid dissociation constant Ka is related to pKa as;.
Answer to Problem 30P
Explanation of Solution
Given Information:
pKa = 5.0.
pH = 7.0.
Calculation:
From pH of solution, concentration of hydroxide ion can be calculated as follows:
Also,
Since,
Thus, by putting the values, the ratio
(d)
Interpretation:
The ratio of A- to [HA] with the help of pH and pKa values should be calculated.
Concept Introduction:
The pH of a solution indicates the number of H3 O+ ions in the solution. The mathematical relation between pH and H3 O+ is as given below;.
Acid dissociation constant Ka is related to pKa as;.
Answer to Problem 30P
Explanation of Solution
Given Information:
pKa = 5.0.
pH = 9.0.
Calculation:
From pH of solution, concentration of hydroxide ion can be calculated as follows:
Also,
Since,
Thus, by putting the values, the ratio
(e)
Interpretation:
The ratio of A- to [HA] with the help of pH and pKa values should be calculated.
Concept Introduction:
The pH of a solution indicates the number of H3 O+ ions in the solution. The mathematical relation between pH and H3 O+ is as given below;.
Acid dissociation constant Ka is related to pKa as;.
Answer to Problem 30P
Explanation of Solution
Given Information:
pKa = 5.0.
pH = 11.0.
Calculation:
From pH of solution, concentration of hydroxide ion can be calculated as follows:
Also,
Since,
Thus, by putting the values, the ratio
Want to see more full solutions like this?
Chapter 17 Solutions
Introduction to General, Organic and Biochemistry
- Draw the molecules.arrow_forwardDraw the mechanism for the acid-catalyzed dehydration of 2-methyl-hexan-2-ol with arrows please.arrow_forward. Draw the products for addition reactions (label as major or minor) of the reaction between 2-methyl-2-butene and with following reactants : Steps to follow : A. These are addition reactions you need to break a double bond and make two products if possible. B. As of Markovnikov rule the hydrogen should go to that double bond carbon which has more hydrogen to make stable products or major product. Here is the link for additional help : https://study.com/academy/answer/predict-the-major-and-minor-products-of-2-methyl- 2-butene-with-hbr-as-an-electrophilic-addition-reaction-include-the-intermediate- reactions.html H₂C CH3 H H3C CH3 2-methyl-2-butene CH3 Same structure CH3 IENCESarrow_forward
- Draw everything on a piece of paper including every single step and each name provided using carbons less than 3 please.arrow_forwardTopics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. H The IUPAC name isarrow_forward[Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is Submit Answer Retry Entire Group 9 more group attempts remainingarrow_forward
- Please draw.arrow_forwardA chromatogram with ideal Gaussian bands has tR = 9.0 minutes and w1/2 = 2.0 minutes. Find the number of theoretical plates that are present, and calculate the height of each theoretical plate if the column is 10 centimeters long.arrow_forwardAn open tubular column has an inner diameter of 207 micrometers, and the thickness of the stationary phase on the inner wall is 0.50 micrometers. Unretained solute passes through in 63 seconds and a particular solute emerges at 433 seconds. Find the distribution constant for this solute and find the fraction of time spent in the stationary phase.arrow_forward
- Consider a chromatography column in which Vs= Vm/5. Find the retention factor if Kd= 3 and Kd= 30.arrow_forwardTo improve chromatographic separation, you must: Increase the number of theoretical plates on the column. Increase the height of theoretical plates on the column. Increase both the number and height of theoretical plates on the column. Increasing the flow rate of the mobile phase would Increase longitudinal diffusion Increase broadening due to mass transfer Increase broadening due to multiple paths You can improve the separation of components in gas chromatography by: Rasing the temperature of the injection port Rasing the temperature of the column isothermally Rasing the temperature of the column using temperature programming In GC, separation between two different solutes occurs because the solutes have different solubilities in the mobile phase the solutes volatilize at different rates in the injector the solutes spend different amounts of time in the stationary phasearrow_forwardplease draw and example of the following: Show the base pair connection(hydrogen bond) in DNA and RNAarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


