
Physics (5th Edition)
5th Edition
ISBN: 9780321976444
Author: James S. Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.6, Problem 6EYU
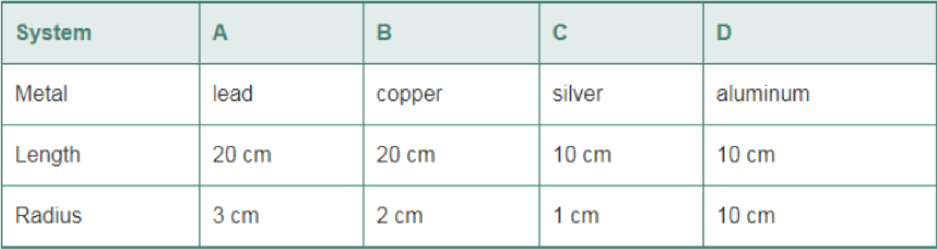
The following systems consist of a cylindrical metal rod with a given length and radius. Rank the systems in order of increasing heat
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Can someone help me answer this physics 2 questions. Thank you.
Four capacitors are connected as shown in the figure below. (Let C = 12.0 μF.)
a
C
3.00 με
Hh.
6.00 με
20.0 με
HE
(a) Find the equivalent capacitance between points a and b.
5.92
HF
(b) Calculate the charge on each capacitor, taking AV ab = 16.0 V.
20.0 uF capacitor 94.7
6.00 uF capacitor 67.6
32.14
3.00 µF capacitor
capacitor C
☑
με
με
The 3 µF and 12.0 uF capacitors are in series and that combination is in parallel with the 6 μF capacitor. What quantity is the same for capacitors in parallel? μC
32.14
☑
You are correct that the charge on this capacitor will be the same as the charge on the 3 μF capacitor. μC
In the pivot assignment, we observed waves moving on a string stretched by hanging
weights. We noticed that certain frequencies produced standing waves. One such
situation is shown below:
0 ст
Direct Measurement
©2015 Peter Bohacek I.
20
0 cm 10
20
30
40
50
60
70
80
90
100
Which Harmonic is this?
Do NOT include units!
What is the wavelength of this wave in cm with only no decimal places?
If the speed of this wave is 2500 cm/s, what is the frequency of this harmonic (in Hz, with
NO decimal places)?
Chapter 16 Solutions
Physics (5th Edition)
Ch. 16.1 - Prob. 1EYUCh. 16.2 - Is the size of a degree in the Fahrenheit scale...Ch. 16.3 - The following systems consist of a metal rod with...Ch. 16.4 - Prob. 4EYUCh. 16.5 - Prob. 5EYUCh. 16.6 - The following systems consist of a cylindrical...Ch. 16 - Prob. 1CQCh. 16 - Prob. 2CQCh. 16 - Prob. 3CQCh. 16 - If the glass in a glass thermometer had the same...
Ch. 16 - Prob. 5CQCh. 16 - Sometimes the metal lid on a glass jar has been...Ch. 16 - Prob. 7CQCh. 16 - The specific heat of concrete is greater than that...Ch. 16 - When you touch a piece of metal and a piece of...Ch. 16 - The rate of heat flow through a slab does not...Ch. 16 - Prob. 11CQCh. 16 - Updrafts of air allow hawks and eagles to glide...Ch. 16 - BIO The fur of polar bears consists of hollow...Ch. 16 - Object 2 has twice the emissivity of object 1,...Ch. 16 - Prob. 1PCECh. 16 - Prob. 2PCECh. 16 - Incandescent lightbulbs heat a tungsten filament...Ch. 16 - Normal body temperature for humans is 98.6 F. What...Ch. 16 - The temperature at the surface of the Sun is about...Ch. 16 - One day you notice that the outside temperature...Ch. 16 - The gas in a constant-volume gas thermometer has a...Ch. 16 - Prob. 8PCECh. 16 - Greatest Change in Temperature A world record for...Ch. 16 - Prob. 10PCECh. 16 - Prob. 11PCECh. 16 - When the bulb of a constant-volume gas thermometer...Ch. 16 - Bimetallic strip A is made of copper and steel;...Ch. 16 - Prob. 14PCECh. 16 - Predict/Explain A brass plate has a circular hole...Ch. 16 - Figure 16-25 shows five metal plates, all at the...Ch. 16 - Longest Suspension Bridge The worlds longest...Ch. 16 - A vinyl siding panel for a house is installed on a...Ch. 16 - A cylinder bore in an aluminum engine block has a...Ch. 16 - Prob. 20PCECh. 16 - At 18.75 C a brass sleeve has an inside diameter...Ch. 16 - Early in the morning, when the temperature is 5.5...Ch. 16 - Some cookware has a stainless steel interior ( =...Ch. 16 - Predict/Calculate You construct two wire-frame...Ch. 16 - A metal ball that is 1.2 m in diameter expands by...Ch. 16 - A copper ball with a radius of 1.7 cm is heated...Ch. 16 - Predict/Calculate An aluminum saucepan with a...Ch. 16 - Prob. 28PCECh. 16 - BIO An exercise machine indicates that you have...Ch. 16 - BIO A certain sandwich cookie contains 53 C of...Ch. 16 - BIO During a workout, a person repeatedly lifts a...Ch. 16 - Prob. 32PCECh. 16 - BIO It was shown in Example 16-18 that a typical...Ch. 16 - Predict/Explain Two objects are made of the same...Ch. 16 - Prob. 35PCECh. 16 - Prob. 36PCECh. 16 - Prob. 37PCECh. 16 - A 9.7-g lead bullet is fired into a fence post....Ch. 16 - Prob. 39PCECh. 16 - Prob. 40PCECh. 16 - A 225-g lead ball at a temperature of 81.2 C is...Ch. 16 - If 2200 J of heat are added to a 190-g object, its...Ch. 16 - Chips by the Ton Tortilla chips are manufactured...Ch. 16 - Prob. 44PCECh. 16 - To determine the specific heat of an object, a...Ch. 16 - Predict/Calculate A student drops a 0.33-kg piece...Ch. 16 - Prob. 47PCECh. 16 - Predict/Explain In a popular lecture...Ch. 16 - Figure 16-27 shows a composite slab of three...Ch. 16 - Figure 16-28 Problem 50 50. CE Heat is...Ch. 16 - Predict/Explain Two identical bowls of casserole...Ch. 16 - Two bowls of soup with identical temperatures are...Ch. 16 - A glass window 0.33 cm thick measures 81 cm by 39...Ch. 16 - BIO Assuming your skin temperature is 37.2 C and...Ch. 16 - Find the heat that flows in 1.0 s through a lead...Ch. 16 - Consider a double-paned window consisting of two...Ch. 16 - Predict/Calculate Two metal rods of equal...Ch. 16 - Two cylindrical metal rodsone copper, the other...Ch. 16 - Prob. 59PCECh. 16 - Predict/Calculate Consider two cylindrical metal...Ch. 16 - A copper rod 85 cm long is used to poke a fire....Ch. 16 - Two identical objects are placed in a room at 24...Ch. 16 - A block has the dimensions L, 2L, and 3L. When one...Ch. 16 - Prob. 64GPCh. 16 - CE A copper ring stands on edge with a metal rod...Ch. 16 - CE Referring to the copper ring in the previous...Ch. 16 - Prob. 67GPCh. 16 - Making Steel Sheets In the continuous-caster...Ch. 16 - The Coldest Place in the Universe The Boomerang...Ch. 16 - BIO The Hottest Living Things From the surreal...Ch. 16 - Thermal energy is added to 180 g of water at a...Ch. 16 - Prob. 72GPCh. 16 - BIO Brain Power As you read this problem, your...Ch. 16 - BIO Brain Food Your brain consumes about 22 W of...Ch. 16 - BIO The Cricket Thermometer The rate of chirping...Ch. 16 - Predict/Calculate A pendulum consists of a large...Ch. 16 - Prob. 77GPCh. 16 - A256-kg rock sits in full sunlight on the edge of...Ch. 16 - Prob. 79GPCh. 16 - Thermal Storage Solar heating of a house is much...Ch. 16 - Pave It Over Suppose city 1 leaves an entire block...Ch. 16 - Prob. 82GPCh. 16 - You turn a crank on a device similar to that shown...Ch. 16 - Prob. 84GPCh. 16 - The Solar Constant The surface of the Sun has a...Ch. 16 - Bars of two different metals are bolted together,...Ch. 16 - A grandfather clock has a simple brass pendulum of...Ch. 16 - Prob. 88GPCh. 16 - A layer of ice has formed on a small pond. The air...Ch. 16 - A Double-Paned Window An energy-efficient...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Cool Medicine In situations in which the brain is...Ch. 16 - Prob. 94PPCh. 16 - Referring to Example 16-12 Suppose the mass of the...Ch. 16 - Referring to Example 16-12 Suppose the initial...Ch. 16 - Prob. 97PPCh. 16 - Predict/Calculate Referring to Example 16-16...
Additional Science Textbook Solutions
Find more solutions based on key concepts
One isomer of methamphetamine is the addictive illegal drug known as crank. Another isomer is a medicine for si...
Campbell Essential Biology (7th Edition)
1. How many cervical, thoracic, lumbar, sacral, and coccygeal vertebrae are normally present in the vertebral ...
Human Anatomy & Physiology (2nd Edition)
The bioremediation process shown in the photograph is used to remove benzene and other hydrocarbons from soil c...
Microbiology: An Introduction
Based on your answers to Questions 2 and 3, which part of the Atlantic basin appears to have opened first?
Applications and Investigations in Earth Science (9th Edition)
23. How many significant figures are there in the following values?
a. 0.05 × 10-4 b. 0.00340
c. 7.2 × 104 ...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
In cats, tortoiseshell coat color appears in females. A tortoiseshell coat has patches of dark brown fur and pa...
Genetic Analysis: An Integrated Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Four capacitors are connected as shown in the figure below. (Let C = 12.0 µF.) A circuit consists of four capacitors. It begins at point a before the wire splits in two directions. On the upper split, there is a capacitor C followed by a 3.00 µF capacitor. On the lower split, there is a 6.00 µF capacitor. The two splits reconnect and are followed by a 20.0 µF capacitor, which is then followed by point b. (a) Find the equivalent capacitance between points a and b. µF(b) Calculate the charge on each capacitor, taking ΔVab = 16.0 V. 20.0 µF capacitor µC 6.00 µF capacitor µC 3.00 µF capacitor µC capacitor C µCarrow_forwardTwo conductors having net charges of +14.0 µC and -14.0 µC have a potential difference of 14.0 V between them. (a) Determine the capacitance of the system. F (b) What is the potential difference between the two conductors if the charges on each are increased to +196.0 µC and -196.0 µC? Varrow_forwardPlease see the attached image and answer the set of questions with proof.arrow_forward
- How, Please type the whole transcript correctly using comma and periods as needed. I have uploaded the picture of a video on YouTube. Thanks,arrow_forwardA spectra is a graph that has amplitude on the Y-axis and frequency on the X-axis. A harmonic spectra simply draws a vertical line at each frequency that a harmonic would be produced. The height of the line indicates the amplitude at which that harmonic would be produced. If the Fo of a sound is 125 Hz, please sketch a spectra (amplitude on the Y axis, frequency on the X axis) of the harmonic series up to the 4th harmonic. Include actual values on Y and X axis.arrow_forwardSketch a sign wave depicting 3 seconds of wave activity for a 5 Hz tone.arrow_forward
- Sketch a sine wave depicting 3 seconds of wave activity for a 5 Hz tone.arrow_forwardThe drawing shows two long, straight wires that are suspended from the ceiling. The mass per unit length of each wire is 0.050 kg/m. Each of the four strings suspending the wires has a length of 1.2 m. When the wires carry identical currents in opposite directions, the angle between the strings holding the two wires is 20°. (a) Draw the free-body diagram showing the forces that act on the right wire with respect to the x axis. Account for each of the strings separately. (b) What is the current in each wire? 1.2 m 20° I -20° 1.2 marrow_forwardplease solve thisarrow_forward
- please solve everything in detailarrow_forward6). What is the magnitude of the potential difference across the 20-02 resistor? 10 Ω 11 V - -Imm 20 Ω 10 Ω 5.00 10 Ω a. 3.2 V b. 7.8 V C. 11 V d. 5.0 V e. 8.6 Varrow_forward2). How much energy is stored in the 50-μF capacitor when Va - V₁ = 22V? 25 µF b 25 µF 50 µFarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY