
ORGANIC CHEMISTRY W/BIOLOGICAL TOPICS
6th Edition
ISBN: 9781260325294
Author: SMITH
Publisher: RENT MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16.6, Problem 15P
Draw all resonance structures for each compound and use the resonance structures to determine if the substituent has an electron-donating or electron-withdrawing resonance effect.
a. 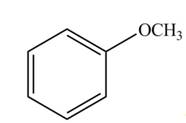 b.
b. 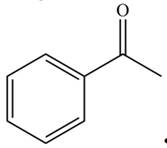
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote!
Please correct answer and don't used hand raiting
Please provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote!
(please correct answer and don't used hand raiting)
Please provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote!
Chapter 16 Solutions
ORGANIC CHEMISTRY W/BIOLOGICAL TOPICS
Ch. 16.1 - Prob. 1PCh. 16.2 - Prob. 2PCh. 16.3 - Prob. 3PCh. 16.4 - Prob. 4PCh. 16.5 - Prob. 5PCh. 16.5 - Prob. 6PCh. 16.5 - Prob. 9PCh. 16.5 - Problem 18.9 Draw the product of each reaction
a....Ch. 16.5 - Prob. 11PCh. 16.5 - Prob. 12P
Ch. 16.5 - Prob. 13PCh. 16.6 - Prob. 14PCh. 16.6 - Problem 18.14 Draw all resonance structures for...Ch. 16.6 - Problem 18.15 Classify each substituent as...Ch. 16.7 - Prob. 17PCh. 16.9 - Prob. 22PCh. 16.10 - Problem 18.20 Draw the products of each...Ch. 16.10 - Prob. 24PCh. 16.11 - Problem 18.22 Draw the products formed when each...Ch. 16.12 - Problem 18.23 Devise a synthesis of each compound...Ch. 16.13 - Problem 18.24 Draw the products of each...Ch. 16.13 - Problem 18.25 Draw a stepwise mechanism for the...Ch. 16.13 - Problem 18.26 Draw the products of each...Ch. 16.13 - Prob. 30PCh. 16 - Prob. 37PCh. 16 - 18.35 What is the major product formed by an...Ch. 16 - 18.36 Draw the products formed when phenol is...Ch. 16 - Problem 18.37 Draw the products formed when each...Ch. 16 - 18.38 Draw the products of each reaction.
a. d....Ch. 16 - 18.39 What products are formed when benzene is...Ch. 16 - Prob. 49PCh. 16 - 18.47 For each of the following substituted...Ch. 16 - 18.48 Consider the tetracyclic aromatic compound...Ch. 16 - 18.49 For each N-substituted benzene, predict...Ch. 16 - Prob. 54PCh. 16 - 18.51 Using resonance structures, explain why a...Ch. 16 - Prob. 56PCh. 16 - 18.53 Rank the aryl halides in each group in order...Ch. 16 - Prob. 64PCh. 16 - Prob. 65PCh. 16 - Prob. 66PCh. 16 - 18.63 Synthesize each compound from benzene and...Ch. 16 - Problem 18.64 Synthesize each compound from...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- CaTiO3 has a perovskite structure. Calculate the packing factor.Data: ionic radii Co+2 = 0.106 nm, Ti+4 = 0.064 nm, O-2 = 0.132 nm; lattice constant is a = 2(rTi4+ + rO-2).(a) 0.581(b) -0.581(c) 0.254(d) -0.254arrow_forwardIn the initial linear section of the stress-strain curve of a metal or alloy. Explain from the point of view of atomic structure?(a) No, the atomic level properties of the material can never be related to the linear section.(b) The elastic zone is influenced by the strength of the bonds between atoms.(c) The stronger the bond, the less rigid and the lower the Young's Modulus of the material tested.(d) The stronger the bond, the less stress is necessary to apply to the material to deform it elastically.arrow_forwardThe degree of polymerization of polytetrafluoroethylene (Teflon) is 7500 (mers/mol). If all polymer chains have equal length, state the molecular weight of the polymer and the total number of chains in 1000 g of the polymer(a) 50 000 g/mol; 0.03·1020 chains(b) 100 000 g/mol; 1.03·1020 chains(c) 750 000 g/mol; 8.03·1020 chainsarrow_forward
- In natural rubber or polyisoprene, the trans isomer leads to a higher degree of crystallinity and density than the cis isomer of the same polymer, because(a) it is more symmetrical and regular.(b) it is less symmetrical.(c) it is irregular.arrow_forwardMost ceramic materials have low thermal conductivities because:(a) Electron mobility is strongly restricted due to their strong ionic-covalent bonding.(b) False, in general they are excellent thermal conductors (they are used in ovens).(c) Electron mobility is dependent on T and therefore they are poor conductors at high temperatures.(d) Electron mobility is very restricted by secondary bonds.arrow_forwardResistivity and electrical conductivity.(a) In metals, resistivity decreases.(b) In metals, resistivity decreases and conductivity in semiconductors also decreases with increasing temperature.(c) With increasing temperature, resistivity in metals and conductivity in semiconductors also increases.(d) None of the above.arrow_forward
- State the difference between concrete and Portland cement.(a) There are no differences, in concrete the chemical composition is silicates and in cement aluminates.(b) The chemical composition of concrete is based on silicates and in cement aluminates.(c) Concrete is composed of aggregates bound by cement and cement "only" contains different minerals.(d) Cement is aggregates bound by concrete.arrow_forwardAmorphous polymers are usually transparent and semi-crystalline polymers are usually opaque. Correct?(a) No. They are all made up of polymer chains. True if they were monomers.(b) Yes. The arrangement of the chains determines the passage of light.(c) No. It is the other way around.(d) Crystallinity or amorphousness does not affect the transparency or opacity of the material.arrow_forwardThe name ferrites refers to a family of(a) ceramic materials that exhibit ferrimagnetic behavior due to their ionic composition.(b) polymeric materials that exhibit ferrimagnetic behavior due to their ionic composition.(c) concrete-based materials that exhibit ferrimagnetic behavior due to their ionic composition.(d) superconducting materials that exhibit ferrimagnetic behavior due to their ionic composition.arrow_forward
- State the two main factors affecting ion packing in the solid state.(a) Number of covalent bonds and their unsaturation.(b) Mechanical properties and degradation temperature.(c) Number of crystalline phases present and grain size.(d) Electroneutrality and ion size.arrow_forwardThe ceramic materials alumina (Al2O3) and chromium oxide (Cr2O3) form an isomorphic phase diagram. The solubility will be(a) unlimited of one ceramic in the other.(b) very limited depending on the weight % of Al2O3(c) very limited depending on the weight % of Cr2O3(d) partial of one ceramic in the other.arrow_forwardAmong the main characteristics of optical fibers, indicate which of the following is not included:(a) Opacity and Rigidity(b) Flexibility(c) Transparency(d) Low thicknessarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY