
Conceptual Physical Science (6th Edition)
6th Edition
ISBN: 9780134060491
Author: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 67E
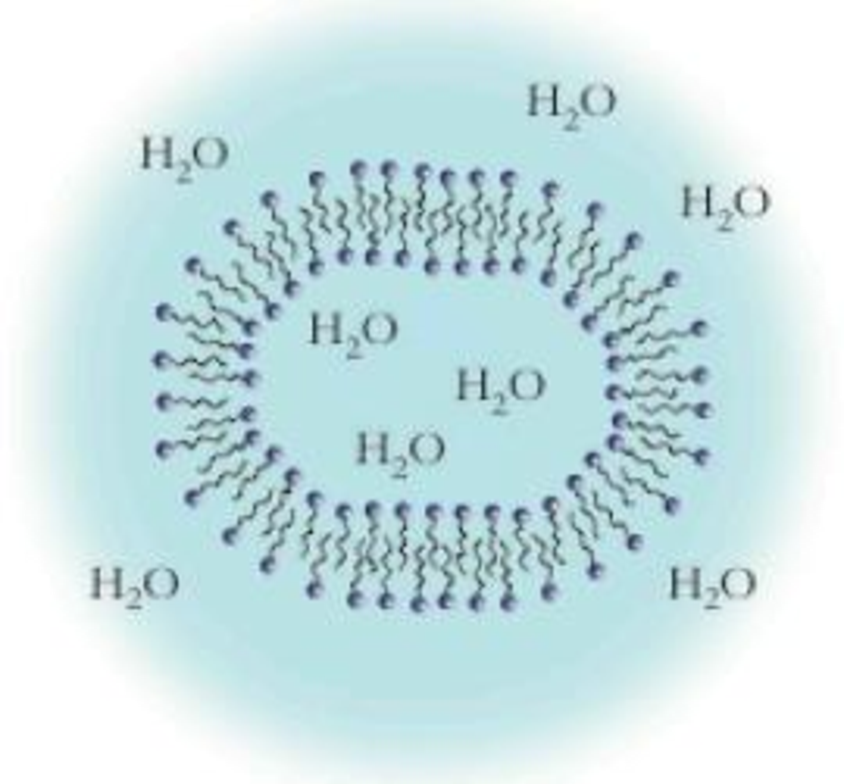
Fatty acid molecules can also align to form a bilipid layer that extends in three dimensions. Shown below is a cross section of this structure. This is similar to the micelle shown in Figure 16.23, though notably different because it contains an inner compartment of water. What is this structure called? (Hint: It forms the basis of all life as we know it.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When united,they form ribosomes?
What is Function nucleus?
( ) is the gelatinous matrix or base material of the nucleus
Describe the Exponential Behavior?
Describe the CaF2 structure in terms of a host framework of one type of atom with selective occupancy of particular interstices in that framework by the other type of atom.
Chapter 16 Solutions
Conceptual Physical Science (6th Edition)
Ch. 16 - Prob. 1RCQCh. 16 - Prob. 2RCQCh. 16 - Prob. 3RCQCh. 16 - Prob. 4RCQCh. 16 - How is a solution different from a suspension?Ch. 16 - How can a solution be separated from a suspension?Ch. 16 - What happens to the volume of a sugar solution as...Ch. 16 - Prob. 8RCQCh. 16 - What does it mean to say that a solution is...Ch. 16 - Is concentration typically given with the volume...
Ch. 16 - Why does the solubility of a gas solute in a...Ch. 16 - Why do sugar crystals dissolve faster when...Ch. 16 - Is sugar a polar or nonpolar substance?Ch. 16 - Which portion of a soap molecule is nonpolar?Ch. 16 - What is the difference between a soap and a...Ch. 16 - Prob. 16RCQCh. 16 - Why are soap molecules so attracted to calcium and...Ch. 16 - Why is treated water sprayed into the air before...Ch. 16 - What are two ways in which people disinfect water...Ch. 16 - What naturally occurring element has been...Ch. 16 - Why can wastewater treatment requirements in...Ch. 16 - What is the first step in treating raw sewage?Ch. 16 - Prob. 23RCQCh. 16 - Prob. 30TASCh. 16 - Prob. 31TASCh. 16 - Prob. 32TASCh. 16 - How much sodium chloride, in grams, is needed to...Ch. 16 - If water is added to 1 mole of sodium chloride in...Ch. 16 - A student is told to use 20.0 g of sodium chloride...Ch. 16 - Rank the following solutions in order of...Ch. 16 - Rank the following compounds in order of...Ch. 16 - Prob. 38TARCh. 16 - How might you separate a mixture of sand and salt?...Ch. 16 - Mixtures can be separated into their components by...Ch. 16 - Why can't the elements of a compound be separated...Ch. 16 - Many dry cereals are fortified with iron, which is...Ch. 16 - The Chemist's Classification of Matter 43....Ch. 16 - Classify each of the following as an element,...Ch. 16 - 45. Which of these boxes best represents a...Ch. 16 - Prob. 46ECh. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Which is more dense: air saturated with water...Ch. 16 - How many sugar molecules are there in a 2 M sugar...Ch. 16 - Prob. 51ECh. 16 - Which should weigh more: 100 mL of fresh water or...Ch. 16 - Explain why, for these three substances, the...Ch. 16 - The boiling point of 1,4-butanediol is 230C. Would...Ch. 16 - Based on atomic size, which would you expect to be...Ch. 16 - If nitrogen, N2, were pumped into your lungs at...Ch. 16 - Prob. 57ECh. 16 - Account for the observation that ethanol, C2H5OH,...Ch. 16 - At 10C, which is more concentrated: a saturated...Ch. 16 - Why is rain or snow called precipitation?Ch. 16 - Prob. 61ECh. 16 - Some bottled water is now advertised as containing...Ch. 16 - Two plastic bottles of fresh seltzer water are...Ch. 16 - Why can 500 mL of fresh water absorb more gaseous...Ch. 16 - Would you expect to find more dissolved oxygen in...Ch. 16 - Soaps, Detergents, and Hard Water Fatty acid...Ch. 16 - Fatty acid molecules can also align to form a...Ch. 16 - Prob. 68ECh. 16 - A scum forms on the surface of boiling hard water....Ch. 16 - Calcium and magnesium ions are more attracted to...Ch. 16 - Phosphate ions, PO43-, were once added to...Ch. 16 - Oils at the top of a tree have a higher...Ch. 16 - Why is distilling water so relatively expensive?Ch. 16 - What reverses with reverse osmosis?Ch. 16 - Why is it significantly less costly to purify...Ch. 16 - Prob. 76ECh. 16 - Many homeowners get their drinking; water piped up...Ch. 16 - Is the decomposition of food by bacteria in our...Ch. 16 - Where does most of the solid mass of raw sewage...Ch. 16 - Why is flushing a toilet with clean water from a...Ch. 16 - Why are people so willing to buy bottled water...Ch. 16 - It is possible to tow icebergs to coastal cities...Ch. 16 - Someone argues that he or she doesn't drink tap...Ch. 16 - Prob. 2RATCh. 16 - The air in your house is an example of a (a)...Ch. 16 - Half-frozen fruit punch is always sweeter than the...Ch. 16 - Why is sodium chloride, NaCl, insoluble in...Ch. 16 - Fish don't live very long in water that has just...Ch. 16 - Prob. 7RATCh. 16 - What is an advantage of using chlorine gas to...Ch. 16 - Why do red blood cells, which contain an aqueous...Ch. 16 - A stagnant pond smells worse than a babbling brook...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Express the unit vectors in terms of (that is, derive Eq. 1.64). Check your answers several ways Also work o...
Introduction to Electrodynamics
Choose the best answer to each of the following. Explain your reasoning. The leading hypothesis for the origin ...
The Cosmic Perspective Fundamentals (2nd Edition)
31. The 2.0 kg, uniform, horizontal rod in Figure P7.31 is seen from the side. What is the gravitational torque...
College Physics: A Strategic Approach (3rd Edition)
3. What is free-fall, and why does it make you weightless? Briefly describe why astronauts are weightless in th...
The Cosmic Perspective (8th Edition)
14. A rifle is aimed horizontally at a target 50 m away. The bullet hits the target 2.0 cm below the aim point....
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Star B has an apparent magnitude of 0, which tells us how bright it appears from Earth at its true location. St...
Lecture- Tutorials for Introductory Astronomy
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- what are some examples of how genes control physical structures and functions of organisms?arrow_forwardUsing the data table provided below for fN vs. T for a specific protein, answer the following: Plot fN vs. T for this data set using excel and provide the excel plot. Be sure to label the axes and provide the associated unitsarrow_forwardWhich of the following statements about the "tree of life" is correct? Before Carl Woese, all cellular life forms were classified into five kingdoms: Monera, Fungi, Plants, Animals, and Protists. Using rRNA genes for phylogenetic reconstruction, Carl Woese uncovered a previously unrecognized group that was thought to be bacteria. Afterward, all cellular life forms were classified into three domains: Archaea, Bacteria, and Eucarya. It is found that the relationship among Archaea, Bacteria, and Eucarya changes, depending on which gene was used for phylogenetic reconstruction. All of the abovearrow_forward
- What is astoroid?arrow_forwardLabel It Using the word bank, label the parts of an animal cell. Each word will be used once. mitochondrion golgi bodies nucleus endoplasmic reticulum cell membrane lysosome vacuole ribosome 1. 2. 3. 4. 5. 6. 7. 8.arrow_forwardWhich of these organelles is involved in the structural stability of cells (i.e. is part of the cytoskeleton)? Mark all that apply. Group of answer choices actin (8 nm) microfilaments cilia flagella free ribosomes Golgi complex (Golgi apparatus) intermediate (10 nm) filaments lysosomes microtubules (25 nm) mitochondria peroxisomes proteasomes rough endoplasmic reticulum smooth endoplasmic reticulum spliceosomesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Foundations of Astronomy (MindTap Course List)PhysicsISBN:9781337399920Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
 Stars and Galaxies (MindTap Course List)PhysicsISBN:9781337399944Author:Michael A. SeedsPublisher:Cengage Learning
Stars and Galaxies (MindTap Course List)PhysicsISBN:9781337399944Author:Michael A. SeedsPublisher:Cengage Learning
 Horizons: Exploring the Universe (MindTap Course ...PhysicsISBN:9781305960961Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Horizons: Exploring the Universe (MindTap Course ...PhysicsISBN:9781305960961Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning Stars and GalaxiesPhysicsISBN:9781305120785Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning
Stars and GalaxiesPhysicsISBN:9781305120785Author:Michael A. Seeds, Dana BackmanPublisher:Cengage Learning

Foundations of Astronomy (MindTap Course List)
Physics
ISBN:9781337399920
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning


Stars and Galaxies (MindTap Course List)
Physics
ISBN:9781337399944
Author:Michael A. Seeds
Publisher:Cengage Learning


Horizons: Exploring the Universe (MindTap Course ...
Physics
ISBN:9781305960961
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning

Stars and Galaxies
Physics
ISBN:9781305120785
Author:Michael A. Seeds, Dana Backman
Publisher:Cengage Learning
General Relativity: The Curvature of Spacetime; Author: Professor Dave Explains;https://www.youtube.com/watch?v=R7V3koyL7Mc;License: Standard YouTube License, CC-BY