
Conceptual Physical Science (6th Edition)
6th Edition
ISBN: 9780134060491
Author: Paul G. Hewitt, John A. Suchocki, Leslie A. Hewitt
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 58E
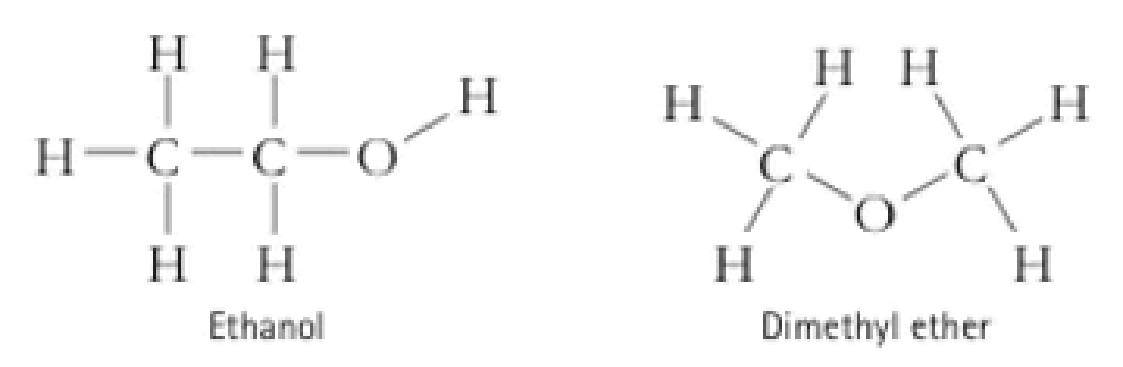
Account for the observation that ethanol, C2H5OH, dissolves readily in water, but dimethyl ether, CH3OCH3, which has the same number and kinds of atoms, does not.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the situation in the figure below; a neutral conducting ball hangs from the ceiling by an insulating string, and a charged insulating rod is going to be placed nearby.
A. First, if the rod was not there, what statement best describes the charge distribution of the ball?
1) Since it is a conductor, all the charges are on the outside of the ball. 2) The ball is neutral, so it has no positive or negative charges anywhere. 3) The positive and negative charges are separated from each other, but we don't know what direction the ball is polarized. 4) The positive and negative charges are evenly distributed everywhere in the ball.
B. Now, when the rod is moved close to the ball, what happens to the charges on the ball?
1) There is a separation of charges in the ball; the side closer to the rod becomes positively charged, and the opposite side becomes negatively charged. 2) Negative charge is drawn from the ground (via the string), so the ball acquires a net negative charge. 3)…
answer question 5-9
AMPS
VOLTS
OHMS
5) 50 A
110 V
6) .08 A
39 V
7) 0.5 A
60
8) 2.5 A
110 V
Chapter 16 Solutions
Conceptual Physical Science (6th Edition)
Ch. 16 - Prob. 1RCQCh. 16 - Prob. 2RCQCh. 16 - Prob. 3RCQCh. 16 - Prob. 4RCQCh. 16 - How is a solution different from a suspension?Ch. 16 - How can a solution be separated from a suspension?Ch. 16 - What happens to the volume of a sugar solution as...Ch. 16 - Prob. 8RCQCh. 16 - What does it mean to say that a solution is...Ch. 16 - Is concentration typically given with the volume...
Ch. 16 - Why does the solubility of a gas solute in a...Ch. 16 - Why do sugar crystals dissolve faster when...Ch. 16 - Is sugar a polar or nonpolar substance?Ch. 16 - Which portion of a soap molecule is nonpolar?Ch. 16 - What is the difference between a soap and a...Ch. 16 - Prob. 16RCQCh. 16 - Why are soap molecules so attracted to calcium and...Ch. 16 - Why is treated water sprayed into the air before...Ch. 16 - What are two ways in which people disinfect water...Ch. 16 - What naturally occurring element has been...Ch. 16 - Why can wastewater treatment requirements in...Ch. 16 - What is the first step in treating raw sewage?Ch. 16 - Prob. 23RCQCh. 16 - Prob. 30TASCh. 16 - Prob. 31TASCh. 16 - Prob. 32TASCh. 16 - How much sodium chloride, in grams, is needed to...Ch. 16 - If water is added to 1 mole of sodium chloride in...Ch. 16 - A student is told to use 20.0 g of sodium chloride...Ch. 16 - Rank the following solutions in order of...Ch. 16 - Rank the following compounds in order of...Ch. 16 - Prob. 38TARCh. 16 - How might you separate a mixture of sand and salt?...Ch. 16 - Mixtures can be separated into their components by...Ch. 16 - Why can't the elements of a compound be separated...Ch. 16 - Many dry cereals are fortified with iron, which is...Ch. 16 - The Chemist's Classification of Matter 43....Ch. 16 - Classify each of the following as an element,...Ch. 16 - 45. Which of these boxes best represents a...Ch. 16 - Prob. 46ECh. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Which is more dense: air saturated with water...Ch. 16 - How many sugar molecules are there in a 2 M sugar...Ch. 16 - Prob. 51ECh. 16 - Which should weigh more: 100 mL of fresh water or...Ch. 16 - Explain why, for these three substances, the...Ch. 16 - The boiling point of 1,4-butanediol is 230C. Would...Ch. 16 - Based on atomic size, which would you expect to be...Ch. 16 - If nitrogen, N2, were pumped into your lungs at...Ch. 16 - Prob. 57ECh. 16 - Account for the observation that ethanol, C2H5OH,...Ch. 16 - At 10C, which is more concentrated: a saturated...Ch. 16 - Why is rain or snow called precipitation?Ch. 16 - Prob. 61ECh. 16 - Some bottled water is now advertised as containing...Ch. 16 - Two plastic bottles of fresh seltzer water are...Ch. 16 - Why can 500 mL of fresh water absorb more gaseous...Ch. 16 - Would you expect to find more dissolved oxygen in...Ch. 16 - Soaps, Detergents, and Hard Water Fatty acid...Ch. 16 - Fatty acid molecules can also align to form a...Ch. 16 - Prob. 68ECh. 16 - A scum forms on the surface of boiling hard water....Ch. 16 - Calcium and magnesium ions are more attracted to...Ch. 16 - Phosphate ions, PO43-, were once added to...Ch. 16 - Oils at the top of a tree have a higher...Ch. 16 - Why is distilling water so relatively expensive?Ch. 16 - What reverses with reverse osmosis?Ch. 16 - Why is it significantly less costly to purify...Ch. 16 - Prob. 76ECh. 16 - Many homeowners get their drinking; water piped up...Ch. 16 - Is the decomposition of food by bacteria in our...Ch. 16 - Where does most of the solid mass of raw sewage...Ch. 16 - Why is flushing a toilet with clean water from a...Ch. 16 - Why are people so willing to buy bottled water...Ch. 16 - It is possible to tow icebergs to coastal cities...Ch. 16 - Someone argues that he or she doesn't drink tap...Ch. 16 - Prob. 2RATCh. 16 - The air in your house is an example of a (a)...Ch. 16 - Half-frozen fruit punch is always sweeter than the...Ch. 16 - Why is sodium chloride, NaCl, insoluble in...Ch. 16 - Fish don't live very long in water that has just...Ch. 16 - Prob. 7RATCh. 16 - What is an advantage of using chlorine gas to...Ch. 16 - Why do red blood cells, which contain an aqueous...Ch. 16 - A stagnant pond smells worse than a babbling brook...
Additional Science Textbook Solutions
Find more solutions based on key concepts
If someone at the other end of a room smokes a cigarette, you may breathe in some smoke. The movement of smoke ...
Campbell Essential Biology with Physiology (5th Edition)
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Which one of the following is not a fuel produced by microorganisms? a. algal oil b. ethanol c. hydrogen d. met...
Microbiology: An Introduction
Match each of the following items with all the terms it applies to:
Human Physiology: An Integrated Approach (8th Edition)
53. This reaction was monitored as a function of time:
A plot of In[A] versus time yields a straight ...
Chemistry: Structure and Properties (2nd Edition)
16. A 200 g mass attached to a horizontal spring oscillates at a frequency of 2.0 Hz. At , the mass is at and ...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- The drawing shows an edge-on view of two planar surfaces that intersect and are mutually perpendicular. Surface (1) has an area of 1.90 m², while surface (2) has an area of 3.90 m². The electric field in the drawing is uniform and has a magnitude of 215 N/C. Find the magnitude of the electric flux through surface (1 and 2 combined) if the angle 8 made between the electric field with surface (2) is 30.0°. Solve in Nm²/C 1 Ө Surface 2 Surface 1arrow_forwardPROBLEM 5 What is the magnitude and direction of the resultant force acting on the connection support shown here? F₁ = 700 lbs F2 = 250 lbs 70° 60° F3 = 700 lbs 45° F4 = 300 lbs 40° Fs = 800 lbs 18° Free Body Diagram F₁ = 700 lbs 70° 250 lbs 60° F3= = 700 lbs 45° F₁ = 300 lbs 40° = Fs 800 lbs 18°arrow_forwardPROBLEM 3 Cables A and B are Supporting a 185-lb wooden crate. What is the magnitude of the tension force in each cable? A 20° 35° 185 lbsarrow_forward
- The determined Wile E. Coyote is out once more to try to capture the elusive Road Runner of Loony Tunes fame. The coyote is strapped to a rocket, which provide a constant horizontal acceleration of 15.0 m/s2. The coyote starts off at rest 79.2 m from the edge of a cliff at the instant the roadrunner zips by in the direction of the cliff. If the roadrunner moves with constant speed, find the minimum velocity the roadrunner must have to reach the cliff before the coyote. (proper sig fig in answer)arrow_forwardPROBLEM 4 What is the resultant of the force system acting on the connection shown? 25 F₁ = 80 lbs IK 65° F2 = 60 lbsarrow_forwardThree point-like charges in the attached image are placed at the corners of an equilateral triangle as shown in the figure. Each side of the triangle has a length of 38.0 cm, and the point (C) is located half way between q1 and q3 along the side. Find the magnitude of the electric field at point (C). Let q1 = −2.80 µC, q2 = −3.40 µC, and q3 = −4.50 µC. Thank you.arrow_forward
- STRUCTURES I Homework #1: Force Systems Name: TA: PROBLEM 1 Determine the horizontal and vertical components of the force in the cable shown. PROBLEM 2 The horizontal component of force F is 30 lb. What is the magnitude of force F? 6 10 4 4 F = 600lbs F = ?arrow_forwardThe determined Wile E. Coyote is out once more to try to capture the elusive Road Runner of Loony Tunes fame. The coyote is strapped to a rocket, which provide a constant horizontal acceleration of 15.0 m/s2. The coyote starts off at rest 79.2 m from the edge of a cliff at the instant the roadrunner zips by in the direction of the cliff. If the roadrunner moves with constant speed, find the minimum velocity the roadrunner must have to reach the cliff before the coyote. (proper sig fig)arrow_forwardHello, I need some help with calculations for a lab, it is Kinematics: Finding Acceleration Due to Gravity. Equations: s=s0+v0t+1/2at2 and a=gsinθ. The hypotenuse,r, is 100cm (given) and a height, y, is 3.5 cm (given). How do I find the Angle θ1? And, for distance traveled, s, would all be 100cm? For my first observations I recorded four trials in seconds: 1 - 2.13s, 2 - 2.60s, 3 - 2.08s, & 4 - 1.95s. This would all go in the coloumn for time right? How do I solve for the experimental approximation of the acceleration? Help with trial 1 would be great so I can use that as a model for the other trials. Thanks!arrow_forward
- After the countdown at the beginning of a Mario Kart race, Bowser slams on the gas, taking off from rest. Bowser get up to a full speed of 25.5 m/s due to an acceleration of 10.4 m/s2. A)How much time does it take to reach full speed? B) How far does Bowser travel while accelerating?arrow_forwardThe drawing in the image attached shows an edge-on view of two planar surfaces that intersect and are mutually perpendicular. Side 1 has an area of 1.90 m^2, Side 2 has an area of 3.90 m^2, the electric field in magnitude is around 215 N/C. Please find the electric flux magnitude through side 1 and 2 combined if the angle (theta) made between the electric field with side 2 is 30.0 degrees. I believe side 1 is 60 degrees but could be wrong. Thank you.arrow_forwardAfter the countdown at the beginning of a Mario Kart race, Bowser slams on the gas, taking off from rest. Bowser get up to a full speed of 25.5 m/s due to an acceleration of 10.4 m/s2.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning


An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning

Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY