
Bundle: Chemistry: An Atoms First Approach, Loose-leaf Version, 2nd + OWLv2 with Student Solutions Manual, 4 terms (24 months) Printed Access Card
2nd Edition
ISBN: 9781337086431
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16Q
Consider the following potential energy plots:
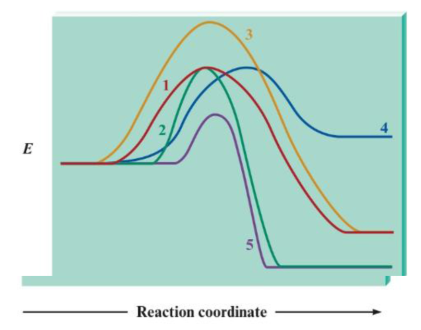
a. Rank the reactions from fastest to slowest and explain your answer. If any reactions have equal rates, explain why.
b. Label the reactions as endothermic or exothermic, and support your answer.
c. Rank the exothermic reactions from greatest to least change in potential energy. and support your answer.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Choose the best reagent for carrying out the following reactions from the list below. Place the letter of
the reagent(s) in the box over the reaction arrow. Use only one letter per box.
OH
OH
CH
CH
CH3
CHS
CH3
f
OH
OCH 3
H
A.
NaH, then CHI
B.
C.
m-ClC6H4COзH
D.
E.
warm H2SO4/H₂O
F.
G.
H₂/Pd
H.
I.
Cl₂, H₂O
J.
NaOCH3, CH3OH
CH3MgBr in ether, then H3O+
Hg(O2CCF3)2, CH3OH
PCC, CH2Cl2
LiAlH4 in ether, then H3O+
What is the product of the reaction of 2,4-pentanedione with phenylhydrazine?
In the reaction of naphthalene with CrO3 in acetic acid. Indicate
whether a different product is obtained if carried out at 25°C or with
heating (A).
Chapter 16 Solutions
Bundle: Chemistry: An Atoms First Approach, Loose-leaf Version, 2nd + OWLv2 with Student Solutions Manual, 4 terms (24 months) Printed Access Card
Ch. 16 - Define the following: a. spontaneous process b....Ch. 16 - What is the second law of thermodynamics? For any...Ch. 16 - Prob. 3RQCh. 16 - Prob. 4RQCh. 16 - Prob. 5RQCh. 16 - What is the standard free energy change, G, for a...Ch. 16 - If you calculate a value for G for a reaction...Ch. 16 - Consider the equation G = G + RT ln(Q). What is...Ch. 16 - Even if G is negative, the reaction may not occur....Ch. 16 - Prob. 10RQ
Ch. 16 - For the process A(l) A(g), which direction is...Ch. 16 - Prob. 2ALQCh. 16 - Gas A2 reacts with gas B2 to form gas AB at a...Ch. 16 - Prob. 4ALQCh. 16 - Prob. 5ALQCh. 16 - Prob. 6ALQCh. 16 - Predict the sign of S for each of the following...Ch. 16 - Is Ssurr favorable or unfavorable for exothermic...Ch. 16 - At 1 atm, liquid water is heated above 100C. For...Ch. 16 - Prob. 10ALQCh. 16 - The synthesis of glucose directly from CO2 and H2O...Ch. 16 - When the environment is contaminated by a toxic or...Ch. 16 - Entropy has been described as times arrow....Ch. 16 - Prob. 14QCh. 16 - A mixture of hydrogen gas and chlorine gas remains...Ch. 16 - Consider the following potential energy plots: a....Ch. 16 - Prob. 17QCh. 16 - Given the following illustration, what can be said...Ch. 16 - The third law of thermodynamics states that the...Ch. 16 - Prob. 20QCh. 16 - Prob. 21QCh. 16 - Prob. 22QCh. 16 - Monochloroethane (C2H5Cl) can be produced by the...Ch. 16 - Prob. 24QCh. 16 - Which of the following processes are spontaneous?...Ch. 16 - Which of the following processes are spontaneous?...Ch. 16 - Prob. 27ECh. 16 - Consider the following illustration of six...Ch. 16 - Consider the following energy levels, each capable...Ch. 16 - Prob. 30ECh. 16 - Choose the substance with the larger positional...Ch. 16 - Which of the following involve an increase in the...Ch. 16 - Predict the sign of Ssurr for the following...Ch. 16 - Prob. 34ECh. 16 - Given the values of H and S, which of the...Ch. 16 - At what temperatures will the following processes...Ch. 16 - Ethanethiol (C2H5SH; also called ethyl mercaptan)...Ch. 16 - For mercury, the enthalpy of vaporization is 58.51...Ch. 16 - For ammonia (NH3), the enthalpy of fusion is 5.65...Ch. 16 - The enthalpy of vaporization of ethanol is 38.7...Ch. 16 - Predict the sign of S for each of the following...Ch. 16 - Prob. 42ECh. 16 - Prob. 43ECh. 16 - For each of the following pairs, which substance...Ch. 16 - Predict the sign of S and then calculate S for...Ch. 16 - Predict the sign of S and then calculate S for...Ch. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Prob. 49ECh. 16 - Two crystalline forms of white phosphorus are...Ch. 16 - Consider the reaction 2O(g)O2(g) a. Predict the...Ch. 16 - Prob. 52ECh. 16 - Prob. 53ECh. 16 - The major industrial use of hydrogen is in the...Ch. 16 - Prob. 55ECh. 16 - At 100C and 1.00 atm, H = 40.6 kJ/mol for the...Ch. 16 - Prob. 57ECh. 16 - Prob. 58ECh. 16 - Prob. 59ECh. 16 - Prob. 60ECh. 16 - Consider the reaction...Ch. 16 - Consider the reaction 2POCl3(g)2PCl3(g)+O2(g) a....Ch. 16 - Prob. 63ECh. 16 - Consider two reactions for the production of...Ch. 16 - Prob. 65ECh. 16 - Prob. 66ECh. 16 - Consider the reaction 2NO2(g)N2O4(g) For each of...Ch. 16 - Prob. 68ECh. 16 - One of the reactions that destroys ozone in the...Ch. 16 - Hydrogen sulfide can be removed from natural gas...Ch. 16 - Consider the following reaction at 25.0C:...Ch. 16 - The standard free energies of formation and the...Ch. 16 - Calculate G forH2O(g)+12O2(g)H2O2(g) at 600. K,...Ch. 16 - The Ostwald process for the commercial production...Ch. 16 - Cells use the hydrolysis of adenosine...Ch. 16 - One reaction that occurs in human metabolism is...Ch. 16 - Prob. 77ECh. 16 - Consider the following reaction at 298 K:...Ch. 16 - Prob. 79ECh. 16 - The equilibrium constant K for the reaction...Ch. 16 - Prob. 81AECh. 16 - Some water is placed in a coffee-cup calorimeter....Ch. 16 - Consider the following system at equilibrium at...Ch. 16 - Calculate the entropy change for the vaporization...Ch. 16 - As O2(l) is cooled at 1 atm, it freezes at 54.5 K...Ch. 16 - Prob. 86AECh. 16 - Using the following data, calculate the value of...Ch. 16 - Prob. 88AECh. 16 - Carbon monoxide is toxic because it bonds much...Ch. 16 - Prob. 90AECh. 16 - Prob. 91AECh. 16 - Use the equation in Exercise 79 to determine H and...Ch. 16 - Consider the reaction...Ch. 16 - Consider the following diagram of free energy (G)...Ch. 16 - Prob. 95CWPCh. 16 - For rubidium Hvapo=69.0KJ/mol at 686C, its boiling...Ch. 16 - Given the thermodynamic data below, calculate S...Ch. 16 - Prob. 98CWPCh. 16 - Prob. 99CWPCh. 16 - Consider the dissociation of a weak acid HA (Ka =...Ch. 16 - Prob. 101CWPCh. 16 - The equilibrium constant for a certain reaction...Ch. 16 - For the following reactions at constant pressure,...Ch. 16 - The standard enthalpy of formation of H2O(l) at...Ch. 16 - Prob. 105CPCh. 16 - Liquid water at 25C is introduced into an...Ch. 16 - Using data from Appendix 4, calculate H, G, and K...Ch. 16 - Prob. 108CPCh. 16 - Prob. 109CPCh. 16 - Prob. 110CPCh. 16 - Prob. 111CPCh. 16 - Prob. 112CPCh. 16 - If wet silver carbonate is dried in a stream of...Ch. 16 - Carbon tetrachloride (CCl4) and benzene (C6H6)...Ch. 16 - Sodium chloride is added to water (at 25C) until...Ch. 16 - Prob. 116CPCh. 16 - Prob. 117CPCh. 16 - Prob. 118IPCh. 16 - Prob. 119IPCh. 16 - Prob. 120IPCh. 16 - Consider a sample containing 5.00 moles of a...Ch. 16 - Impure nickel, refined by smelting sulfide ores in...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Rub your hands together vigorously. What happens? Discuss the energy transfers and transformations that take...
College Physics: A Strategic Approach (3rd Edition)
Give the IUPAC name for each compound.
Organic Chemistry
Describe the evolution of mammals, tracing their synapsid lineage from early amniote ancestors to true mammals....
Loose Leaf For Integrated Principles Of Zoology
What process causes the Mediterranean intermediate Water MIW to become more dense than water in the adjacent At...
Applications and Investigations in Earth Science (9th Edition)
Why do scientists think that all forms of life on earth have a common origin?
Genetics: From Genes to Genomes
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- QUESTION: Fill in the answers in the empty green boxes 1. Step 2 2. Step 3 3. Step 4 (SUM) 4. Step 5 (df) (GIVEN) 5. Determine S y/x value *The data values have been provided in the worksheet attached in the first image*arrow_forwardIf the symbol A is placed in a reaction, at what temperature does it take place?arrow_forwardBy malonic or acetylacetic synthesis, synthesize 3-methyl-4-oxopentanoic acid (indicate the formulas of the compounds).arrow_forward
- oalmitic acid is a 16 carbon acid. In a balanced equation, the products of the sponification of tripalmitin (glyceryl tripalmitate are blank.arrow_forwardWrite the esterification reaction mechanism of salicylic acid and acetic acid to produce aspirin (acetylsalicylic acid). Note: salicylic acid will act as the alcoholarrow_forwardWhat type of interaction would you expect between the following R groups in the tertiary structure of a protein? O -CH2-CO and -CH2-CH2-CH2-CH2-NH3+ a. disulfide bonds b. salt bridges c. hydrogen bonds HO abios vist anisinoo tedt bigil s ai loistaslor sale! 10 OUT d. hydrophobic interactions e. peptide bondsarrow_forward
- 4. True or false: This skeletal structure represents a saturated fatty acid. Ini to 0 fale) me OH faistong starrow_forwardBy malonic or acetylacetic synthesis, synthesize 5-Methyl-2-hexanone (with the formulas of the compounds).arrow_forwardQUESTION: Answer Question 5: 'Calculating standard error of regression' by filling in all the empty green boxes *The values are all provided in the first photo attached*arrow_forward
- Draw the formula for 3-chlorobenzoic acetic anhydride.arrow_forwardBy malonic or acetylacetic synthesis, synthesize 2-methylbutanoic acid (indicate the formulas of the compounds).arrow_forwardObtain 2-methylbutanoic acid by malonic or acetylacetic synthesis (indicate the formulas of the compounds involved).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY