
Concept explainers
(a)
Interpretation: The second resonance structure for given species and its hybrid is to be drawn. The two resonance structure and the hybrid are to be ranked in order of increasing stability.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.7P
The second resonance structure for given species and its hybrid are,
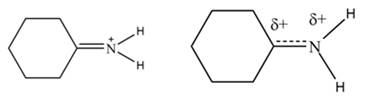
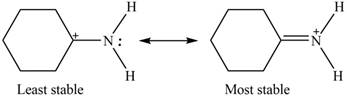
The increasing order of stability for the given resonance structure is,
Explanation of Solution
The given species is shown below.
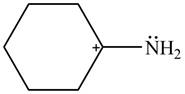
Figure 1
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
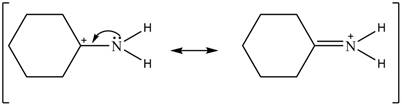
Figure 2
Resonance hybrid is the combination of all resonating structures. Thus, resonance hybrid for the given species is shown below.
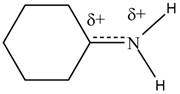
Figure 3
Better resonance structure is the one which has major contribution in the hybrid and others are minor contributors. Major contributor is the resonance structure which possesses more bonds and fewer charges.
The Figure 2 shows that the left side structure does not contain double bond. Thus, the stability of left side structure is less than the right side structure.
Therefore, the increasing order of stability for the given resonance structure is shown below.
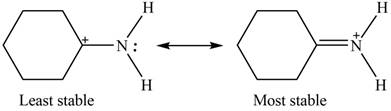
Figure 4
The second resonance structure for given species and its hybrid is shown in Figure 2 and 3. The increasing order of stability for the given resonance structure is shown in Figure 4.
(b)
Interpretation: The second resonance structure for given species and its hybrid is to be drawn. The two resonance structure and the hybrid are to be ranked in order of increasing stability.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.7P
The second resonance structure for given species and its hybrid are,
The increasing order of stability for the given resonance structure is,
Explanation of Solution
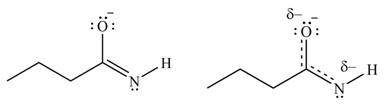
The given species is shown below.
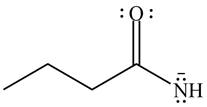
Figure 5
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
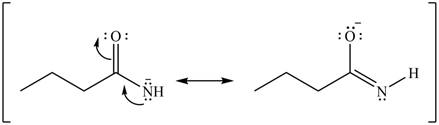
Figure 6
Resonance hybrid is the combination of all resonating structures. Thus, resonance hybrid for the given species is shown below.
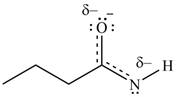
Figure 7
Better resonance structure is the one which has major contribution in the hybrid and others are minor contributors. Major contributor is the resonance structure in which negative charge is on more electronegative atom. The electronegativity of oxygen is more than nitrogen.
The Figure 6 shows that in the left side structure, negative charge is on nitrogen atom, whereas in right side structure, negative charge is on oxygen atom.. Thus, the stability of left side structure is less than the right side structure.
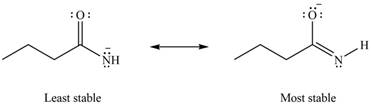
Therefore, the increasing order of stability for the given resonance structure is shown below.
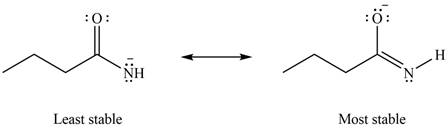
Figure 8
The second resonance structure for given species and its hybrid is shown in Figure 6 and 7. The increasing order of stability for the given resonance structure is shown in Figure 8.
(c)
Interpretation: The second resonance structure for given species and its hybrid is to be drawn. The two resonance structure and the hybrid are to be ranked in order of increasing stability.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.7P
The second resonance structure for given carbocation and its hybrid are,
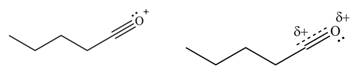
The increasing order of stability for the given resonance structure is,
Explanation of Solution
The given species is shown below.
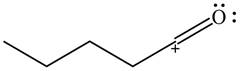
Figure 9
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
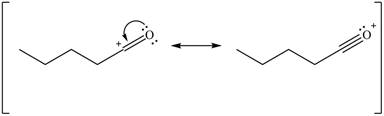
Figure 10
Resonance hybrid is the combination of all resonating structures. Thus, resonance hybrid for the given species is shown below.
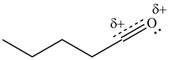
Figure 11
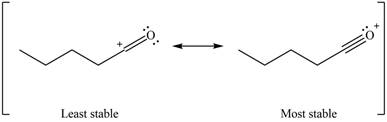
Better resonance structure is the one which has major contribution in the hybrid and others are minor contributors. Major contributor is the resonance structure which possesses more bonds and fewer charges.
Therefore, the increasing order of stability for the given resonance structure is shown below.

Figure 12
The second resonance structure for given carbocation and its hybrid is shown in Figure 10 and 11. The increasing order of stability for the given resonance structure is shown in Figure 12.
(d)
Interpretation: The second resonance structure for given species and its hybrid is to be drawn. The two resonance structure and the hybrid are to be ranked in order of increasing stability.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.7P
The second resonance structure for given carbocation and its hybrid are,
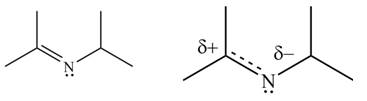
The increasing order of stability for the given resonance structure is,
Explanation of Solution
The given species is shown below.
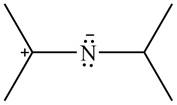
Figure 13
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
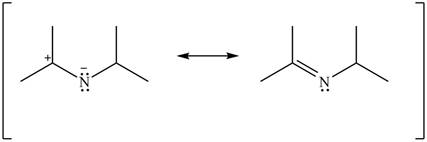
Figure 14
Resonance hybrid is the combination of all resonating structures. Thus, resonance hybrid for the given species is shown below.
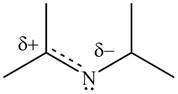
Figure 15
Better resonance structure is the one which has major contribution in the hybrid and others are minor contributors. Major contributor is the resonance structure which possesses more bonds and fewer charges.
The given Figure 2 shows that the left side structure does not contain double bond. Thus, the stability of left side structure is less than the right side structure.
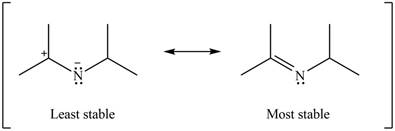
Therefore, the increasing order of stability for the given resonance structure is shown below.

Figure 16
The second resonance structure for given carbocation and its hybrid is shown in Figure 14 and 15. The increasing order of stability for the given resonance structure is shown in Figure 16.
Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEMISTRY
- Identify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forwardRank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forward
- Rank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forwardWhy isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forward
- What is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forwardComplete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forward
- Q Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forwardb. ὋΗ CH3CH2OH H2SO4arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





