
EBK CHEMISTRY
12th Edition
ISBN: 9780133911312
Author: Timberlake
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Question
Chapter 15.5, Problem 15.39QAP
Interpretation Introduction
Interpretation:
Draw the condensed formula for the glycerol phospholipid cephalin that contains two molecules of palmitic acid and the aminoalcohal ethanolamine.
Concept introduction:
Palmitic acid and glycerol are connected by ester bond formation, remaining one group of glycerol is connected to phosphate Palmitic acid:
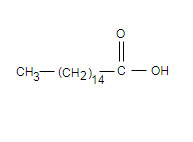
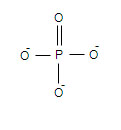
Aminoalcohal ethanolamine:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
11
1 Which one of the following compounds would show a
proton NMR signal at the highest chemical shift? (7pts)
cl
@amitabh
CI CI
d)
Cl
CICI
None
H2SO4 (cat.), H₂O
100 °C
NH₂
Chapter 15 Solutions
EBK CHEMISTRY
Ch. 15.1 - Prob. 15.1QAPCh. 15.1 - Prob. 15.2QAPCh. 15.1 - Lipids are not soluble in water. Are lipids polar...Ch. 15.1 - Which of the following solvents might be used to...Ch. 15.2 - Describe some similarities and differences in the...Ch. 15.2 - Prob. 15.6QAPCh. 15.2 - Prob. 15.7QAPCh. 15.2 - Draw the line-angle formula for each of the...Ch. 15.2 - Prob. 15.9QAPCh. 15.2 - For each of the following fatty acids, give the...
Ch. 15.2 - Prob. 15.11QAPCh. 15.2 - How does the double bond influence the dispersion...Ch. 15.2 - Prob. 15.13QAPCh. 15.2 - Prob. 15.14QAPCh. 15.2 - Prob. 15.15QAPCh. 15.2 - Prob. 15.16QAPCh. 15.2 - Prob. 15.17QAPCh. 15.2 - Prob. 15.18QAPCh. 15.3 - Draw the condensed structural formula for the...Ch. 15.3 - Prob. 15.20QAPCh. 15.3 - Prob. 15.21QAPCh. 15.3 - Draw the condensed structural formula for a mixed...Ch. 15.3 - Prob. 15.23QAPCh. 15.3 - Prob. 15.24QAPCh. 15.3 - Prob. 15.25QAPCh. 15.3 - Prob. 15.26QAPCh. 15.4 - Identify each of the following processes as...Ch. 15.4 - Identify each of the following processes as...Ch. 15.4 - Prob. 15.28QAPCh. 15.4 - Prob. 15.29QAPCh. 15.4 - Prob. 15.30QAPCh. 15.4 - Prob. 15.31QAPCh. 15.4 - Prob. 15.32QAPCh. 15.4 - Prob. 15.33QAPCh. 15.4 - Prob. 15.34QAPCh. 15.4 - Prob. 15.35QAPCh. 15.4 - Draw the condensed structural formula for all the...Ch. 15.5 - Prob. 15.37QAPCh. 15.5 - Prob. 15.38QAPCh. 15.5 - Prob. 15.39QAPCh. 15.5 - Prob. 15.40QAPCh. 15.5 - Prob. 15.41QAPCh. 15.5 - Prob. 15.42QAPCh. 15.5 - Prob. 15.43QAPCh. 15.5 - Prob. 15.44QAPCh. 15.6 - Draw the structure for the steroid nucleus.Ch. 15.6 - Draw the structure for cholesterol.Ch. 15.6 - Prob. 15.47QAPCh. 15.6 - Prob. 15.48QAPCh. 15.6 - Prob. 15.49QAPCh. 15.6 - Prob. 15.50QAPCh. 15.6 - Prob. 15.51QAPCh. 15.6 - Prob. 15.52QAPCh. 15.6 - Prob. 15.53QAPCh. 15.6 - What are the similarities and differences between...Ch. 15.6 - Prob. 15.55QAPCh. 15.6 - Prob. 15.56QAPCh. 15.7 - What is the function if the lipid by layer in a...Ch. 15.7 - Prob. 15.58QAPCh. 15.7 - Prob. 15.59QAPCh. 15.7 - How do the unsaturated fatty acids in the...Ch. 15.7 - Prob. 15.61QAPCh. 15.7 - Prob. 15.62QAPCh. 15.7 - Prob. 15.63QAPCh. 15.7 - 15.66 Identify the type of transport described by...Ch. 15 - Prob. 15.65UTCCh. 15 - Prob. 15.66UTCCh. 15 - Prob. 15.67UTCCh. 15 - Prob. 15.68UTCCh. 15 - Prob. 15.69AQAPCh. 15 - Prob. 15.70AQAPCh. 15 - Prob. 15.71AQAPCh. 15 - Prob. 15.72AQAPCh. 15 - Prob. 15.73AQAPCh. 15 - Prob. 15.74AQAPCh. 15 - Identify each of the following as a fatty acid,...Ch. 15 - Identify each of the following as a fatty acid,...Ch. 15 - 15.81 Identify the components (1 to 6 ) contained...Ch. 15 - Prob. 15.78AQAPCh. 15 - Prob. 15.79AQAPCh. 15 - Prob. 15.80AQAPCh. 15 - Draw the condensed structural formula for a...Ch. 15 - sunflower seed oil can be used to make margarine....Ch. 15 - 15.89 A sink drain can become clogged with solid...Ch. 15 - 15.90 One of the triacylglycerols in olive oil is...Ch. 15 - The plastic known as PETE...Ch. 15 - Using the Internet, look up the condensed...Ch. 15 - The insect repellent DEET is an amide that can be...Ch. 15 - Glyceryl trimyristate (trimyristin) is found in...Ch. 15 - Prob. 31CICh. 15 - Prob. 32CI
Knowledge Booster
Similar questions
- X Draw the major products of the elimination reaction below. If elimination would not occur at a significant rate, check the box under the drawing area instead. ది www. Cl + OH Elimination will not occur at a significant rate. Click and drag to start drawing a structure.arrow_forwardNonearrow_forward1A H 2A Li Be Use the References to access important values if needed for this question. 8A 3A 4A 5A 6A 7A He B C N O F Ne Na Mg 3B 4B 5B 6B 7B 8B-1B 2B Al Si P 1B 2B Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe * Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Ha ****** Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Analyze the following reaction by looking at the electron configurations given below each box. Put a number and a symbol in each box to show the number and kind of the corresponding atom or ion. Use the smallest integers possible. cation anion + + Shell 1: 2 Shell 2: 8 Shell 3: 1 Shell 1 : 2 Shell 2 : 6 Shell 1 : 2 Shell 2: 8 Shell 1: 2 Shell 2: 8arrow_forward
- Nonearrow_forwardIV. Show the detailed synthesis strategy for the following compounds. a. CH3CH2CH2CH2Br CH3CH2CCH2CH2CH3arrow_forwardDo the electrons on the OH participate in resonance with the ring through a p orbital? How many pi electrons are in the ring, 4 (from the two double bonds) or 6 (including the electrons on the O)?arrow_forward
- Predict and draw the product of the following organic reaction:arrow_forwardNonearrow_forwardRedraw the molecule below as a skeletal ("line") structure. Be sure to use wedge and dash bonds if necessary to accurately represent the direction of the bonds to ring substituents. Cl. Br Click and drag to start drawing a structure. : ☐ ☑ Parrow_forward
- K m Choose the best reagents to complete the following reaction. L ZI 0 Problem 4 of 11 A 1. NaOH 2. CH3CH2CH2NH2 1. HCI B OH 2. CH3CH2CH2NH2 DII F1 F2 F3 F4 F5 A F6 C CH3CH2CH2NH2 1. SOCl2 D 2. CH3CH2CH2NH2 1. CH3CH2CH2NH2 E 2. SOCl2 Done PrtScn Home End FA FQ 510 * PgUp M Submit PgDn F11arrow_forwardNonearrow_forwardPlease provide a mechanism of synthesis 1,4-diaminobenzene, start from a benzene ring.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY