
CHEMISTRY:MOLECULAR..(LL)-PRINT..W/CODE
7th Edition
ISBN: 9781119457282
Author: JESPERSEN
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 77RQ
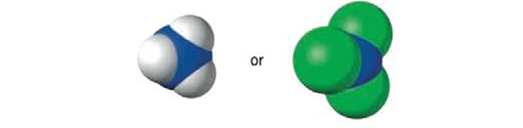
Which of the following compounds is the stronger base? Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which of the following anions is the stronger base? Explain your choice.
Which species is the stronger base and why?
Which is a stronger base, S2 - or Se2 -? Explain.
Chapter 15 Solutions
CHEMISTRY:MOLECULAR..(LL)-PRINT..W/CODE
Ch. 15 - Which of the following are conjugate acid-base...Ch. 15 - Write the formula of the conjugate base for each...Ch. 15 - Sodium cyanide solution, when poured into excess...Ch. 15 - One kind of baking powder contains sodium...Ch. 15 - Which of the following are amphoteric and which...Ch. 15 - The anion of sodium monohydrogen phosphate,...Ch. 15 -
Given that is a stronger acid than what is the...Ch. 15 - Given that HClO is a weaker acid than determine...Ch. 15 - Order the following groups of acids from the...Ch. 15 - Using only the periodic cable, choose the stronger...
Ch. 15 - Prob. 11PECh. 15 - Explain why one acid is weaker than the other in...Ch. 15 - In each pair, explain why one is a stronger acid...Ch. 15 - In each pair, explain why one is a weaker acid...Ch. 15 - How would you expect the acidities of the...Ch. 15 - List these acids in terms of increasing acidity:...Ch. 15 - Identify the Lewis acid and Lewis base in each...Ch. 15 - Is the fluoride ion more likely to behave as a...Ch. 15 - Brnsted-Lowry Acids and Bases How is a...Ch. 15 - Brnsted-Lowry Acids and Bases How are the formulas...Ch. 15 - Brnsted-Lowry Acids and Bases Is H2SO4 the...Ch. 15 - Brnsted-Lowry Acids and Bases What is meant by the...Ch. 15 - Brnsted-Lowry Acids and Bases Define the term...Ch. 15 - Strengths of Bronsted-Lowry Acids and Bases
15.6...Ch. 15 - Strengths of Brønsted-Lowry Acids and Bases
15.7...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases The...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Acetic...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Nitric...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases HCIO4...Ch. 15 - Strengths of Brnsted-Lowry Acids and Bases Formic...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids What are...Ch. 15 - Periodic Trends in the Strength of Acids Within...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids Within...Ch. 15 - Periodic Trends in the Strength of Acids Explain...Ch. 15 - Periodic Trends in the Strength of Acids Astatine,...Ch. 15 - Periodic Trends in the Strength of Acids
15.21...Ch. 15 - Periodic Trends in the Strength of Acids
15.22...Ch. 15 - Periodic Trends in the Strength of Acids Which of...Ch. 15 - Periodic Trends in the Strength of Acids Which of...Ch. 15 - Lewis Acids and Bases Define Lewis acid and Lewis...Ch. 15 - Lewis Acids and Bases In terms of atomic orbitals,...Ch. 15 - Lewis Acids and Bases
15.27 Explain why the...Ch. 15 - Lewis Acids and Bases Methylamine has the formula...Ch. 15 - Use Lewis structures to show the Lewis acid-base...Ch. 15 - Lewis Acids and Bases
15.30 Explain why the oxide...Ch. 15 - Lewis Acids and Bases The molecule SbF5 is able to...Ch. 15 - Lewis Acids and Bases In the reaction of calcium...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 35RQCh. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 40RQCh. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Acid-Base Properties of the Elements and Their...Ch. 15 - Prob. 43RQCh. 15 - Advanced Ceramics and Acid-Base Chemistry What is...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry What is...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry
15.46...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry How does...Ch. 15 - Advanced Ceramics and Acid-Base Chemistry
15.48...Ch. 15 - Brønsted-Lowry Acids and Bases
15.49 Write the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.50 Write the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.51 Write the...Ch. 15 - Brnsted-Lowry Acids and Bases Write the formula...Ch. 15 - Brønsted-Lowry Acids and Bases
15.53 Identify the...Ch. 15 - Brønsted-Lowry Acids and Bases
15.54 Identify the...Ch. 15 - Periodic Trends in the Strengths of Acids Choose...Ch. 15 - Periodic Trends in the Strengths of Acids Choose...Ch. 15 - Choose the stronger acid and give your reason:...Ch. 15 - Choose the stronger acid and give your reason:...Ch. 15 - Choose the stronger acid:...Ch. 15 - Choose the stronger acid:...Ch. 15 - Lewis Acids and Bases Use Lewis symbols co diagram...Ch. 15 - Lewis Acids and Bases Use Lewis symbols to diagram...Ch. 15 - *15.63 Beryllium chloride, , exists in the solid...Ch. 15 - Aluminum chloride, AlCl3, forms molecules with...Ch. 15 - Use Lewis structures to diagram the reaction...Ch. 15 - Use Lewis structures to diagram the reaction...Ch. 15 - Use Lewis structures to show how the following...Ch. 15 - *15.68 Use Lewis structures to show how the...Ch. 15 - Acid-Base Properties of Elements and Their...Ch. 15 - Acid-Base Properties of Elements and Their Oxides...Ch. 15 - Prob. 71RQCh. 15 - Prob. 72RQCh. 15 - What is the formula of the conjugate acid of...Ch. 15 - *15.74 Using liquid ammonia as a solvent, sodium...Ch. 15 - In liquid SO2asasolvent,SOCl2reactswithNa2SO3 in a...Ch. 15 - *15.76 The following space-filling model depicts...Ch. 15 - Which of the following compounds is the stronger...Ch. 15 - Which of the two molecules below is the stronger...Ch. 15 - 15.79 Write equations that illustrate the...Ch. 15 - Hydrogen peroxide is a stronger Brnsted-Lowry acid...Ch. 15 - Sodium hydroxide, NaOH, is basic. Aluminum...Ch. 15 - Hydrazine, N2H4, is a weaker Brnsted-Lowry base...Ch. 15 - Identify the two Brnsted-Lowry acids and two bases...Ch. 15 - In the reaction in the preceding exercise, the...Ch. 15 - How would you expect the degree of ionization of...Ch. 15 - Prob. 86RQCh. 15 - A mixture is prepared containing 0.10 M of each of...Ch. 15 - 15.88 Are all Arrhenius acids Brønsted-Lowry...Ch. 15 - How could you determine whether HBr is a stronger...Ch. 15 - 15.90 Alcohols are organic compounds that have an...Ch. 15 - Acid rain, acid mine runoff, and acid leaching of...Ch. 15 - 15.92 Using just Figure 7.30, find the five most...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Report on copper plating experiment needs to be created. Concept introduction: By passing electricity one metal...
Living By Chemistry: First Edition Textbook
Arrange the following atoms and ions in order of increasing ionic radius: F, S2-, Cl, and Se2-.
F < S2- < Cl <...
Chemistry: The Central Science (14th Edition)
Answer the following questions for each compound: a. How many signals are in its 13C NMR spectrum? b. Which sig...
Organic Chemistry (8th Edition)
Calculate the elevation of the water table at each well location and write the approximate elevation on the lin...
Applications and Investigations in Earth Science (9th Edition)
There are 25 individuals in population 1, all with genotype AA, and there are 40 individuals in population 2, a...
Campbell Biology (11th Edition)
27. Consider the reaction.
Express the rate of the reaction in terms of the change in concentration of each of...
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Summarize the relationship between pKa and base strength by completing the followingsentences: a. For a given base, the higher the pKa of its conjugate acid, the stronger or weaker the base. b. For a given base, the lower the pKa of its conjugate acid, the stronger or weaker the base.arrow_forwardSeveral acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forwardComplete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reaction. (a) (b) (c) (d)arrow_forward
- (E) Label each of the following as strong acid, strong base, or neither.arrow_forwardAs we shall see in Chapter 19, hydrogens on a carbon adjacent to a carbonyl group are far more acidic than those not adjacent to a carbonyl group. The anion derived from acetone, for example, is more stable than is the anion derived from ethane. Account for the greater stability of the anion from acetone.arrow_forwardThe following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forward
- For the previous four questions, label each molecule that appears in the question or your answer asstrong acid, strong base, weak acid, or weak base.arrow_forwardList the following bases in order of their decreasing strength strongest base first: CN,H2O,HSO3,ClO,Cl.arrow_forwardThe following reactions illustrate Brnsted acid-base behavior. Complete each equation. a.HI(aq)+?H3O+(aq)+I(aq) b.NH3(l)+?NH4++NH2 c.H2C2O4(aq)+H2O(l)?+HC2O4(aq) d.H2N2O2(aq)+H2O(l)H3O+(aq)+? e.?+H2O(l)H3O+(aq)+CO32(aq)arrow_forward
- Predict the position of equilibrium for this acid-base reaction.arrow_forwardSummarize the relationship between pKa and acid strength by completing the following sentences: a. The higher the pKa of an acid, the stronger or weaker the acid. b. The lower the pKa of an acid, the stronger or weaker the acid.arrow_forwardA very strong base can remove a proton from methylamine:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY