
Concept explainers
(a)
Interpretation: The product(s) formed by the reaction of given
Concept introduction: The reaction of hydrogen halide with alkene results in the formation of
Answer to Problem 15.23P
The product formed by the reaction of given alkene with
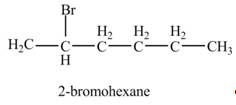
The product formed by the reaction of given alkene with
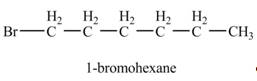
Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.
![]()
Figure 1
The steps followed by electrophilic addition reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
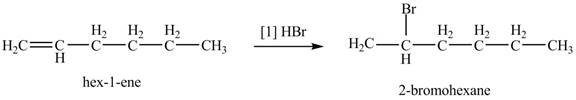
Figure 2
The product formed by the reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
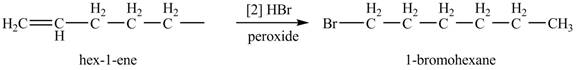
Figure 3
The product formed by the reaction of given alkene with
The product(s) formed by the reaction of given alkene with
(b)
Interpretation: The product(s) formed by the reaction of given alkene with
Concept introduction: The reaction of hydrogen halide with alkene results in the formation of alkyl halide. This type of reaction is an electrophilic addition of hydrogen halide. Electrophilic addition reactions are those in which breaking of pi bond take place to form new sigma bond.
Answer to Problem 15.23P
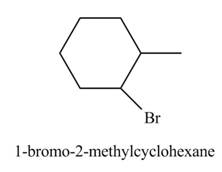
The product formed by the reaction of given alkene with
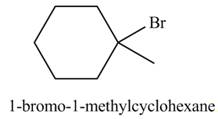
The product formed by the reaction of given alkene with
Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.
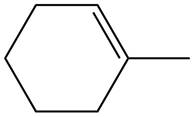
Figure 4
The steps followed by electrophilic addition reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
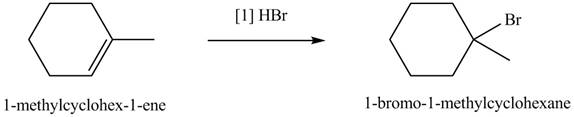
Figure 5
The product formed by the reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
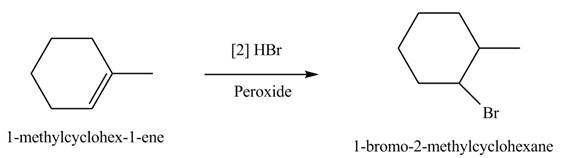
Figure 6
The product formed by the reaction of given alkene with
The product(s) formed by the reaction of given alkene with
(c)
Interpretation: The product(s) formed by the reaction of given alkene with
Concept introduction: The reaction of hydrogen halide with alkene results in the formation of alkyl halide. This type of reaction is an electrophilic addition of hydrogen halide. Electrophilic addition reactions are those in which breaking of pi bond take place to form new sigma bond.
Answer to Problem 15.23P
The product formed by the reaction of given alkene with
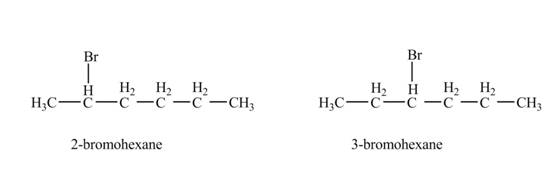
The product formed by the reaction of given alkene with

Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.

Figure 7
The steps followed by electrophilic addition reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
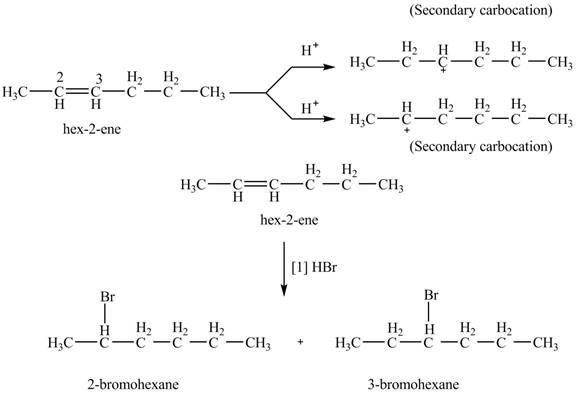
Figure 8
The reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
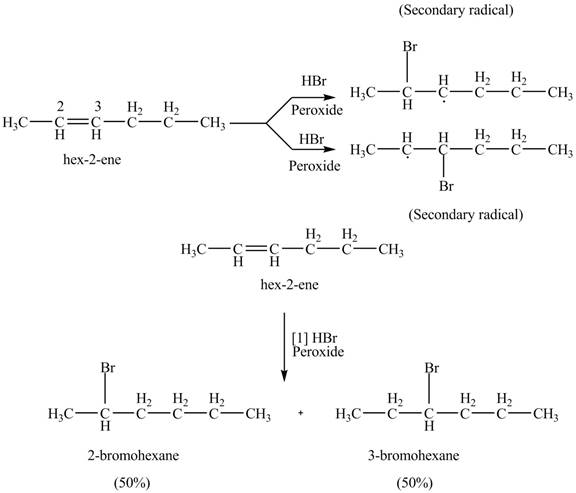
Figure 9
The reaction of given alkene with
The product(s) formed by the reaction of given alkene with
Want to see more full solutions like this?
Chapter 15 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- Calculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.000259 M HClO4arrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. NaN₃arrow_forward
- A. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardCan I please get help with this.arrow_forward
- Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forwardPlease help me with identifying these.arrow_forwardCan I please get help with this?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





