
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
8th Edition
ISBN: 9780134595450
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.4, Problem 5P
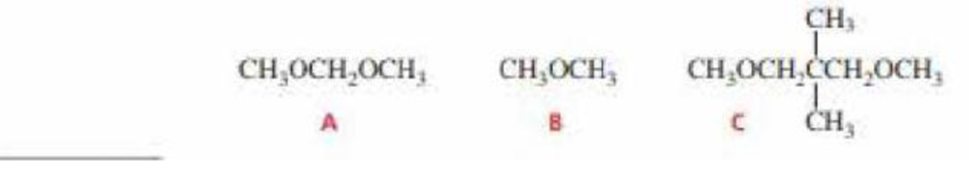
How could you distinguish the 1H NMR spectra of the following compounds?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
achieve.macmillanlearning.com
Canvas
EA eac
h Hulu
YouTube
G 3 methyl cyclobutanol - Google Search
Ranking Phenol Acidity
Course -236 - Organic Chemistry - Mac...
←
Assessment
Completed 10 of 22 Questions
1 +
Netflix
paramount plus
chem hw
Galdehyde reaction with grignard reagent...
b My Questions | bartleby
M Inbox - chenteislegit@gmail.com - Gmail
Due: Fri, Jan 31
Resources
Solution
Penalized
? Hint
Submit Answer
Use retrosynthetic analysis to suggest two paths to synthesize 2-methyl-3-hexanol using the Grignard reaction. (Click and drag
the appropriate image to the correct position in the reactions.)
Route 1
Aldehyde 1
or
+98
Aldehyde 2
Route 2
Q6
+100
Solved in 1 attempt
Q7
+95
Solved in 2 attempts
Q8
+98
Unlimited attempts
possible
+
+
Grignard 1
OH
H3O+
Grignard 2
Answer Bank
Q9
+90
MgBr
Unlimited attempts
possible
CH3CH2CH2MgBr
Q10
Unlimited attempts
Q11
?
?
+100
in 1 attempt
2-methyl-3-hexanol
CH3CH2MgBr
H
H
о
H
Attempt 3
2) (4 pt) After the reaction was completed, the student collected the following data. Crude
product data is the data collected after the reaction is finished, but before the product
is purified. "Pure" product data is the data collected after attempted purification using
recrystallization.
Student B's data:
Crude product data
"Pure"
product data
after
recrystallization
Crude mass: 0.93 g grey solid
Crude mp: 96-106 °C
Crude % yield:
Pure mass: 0.39 g white solid
Pure mp: 111-113 °C
Pure % yield:
a) Calculate the crude and pure percent yields for the student's reaction.
b) Summarize what is indicated by the crude and pure melting points.
Don't used hand raiting
Chapter 14 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
Ch. 14.1 - Prob. 1PCh. 14.1 - Prob. 2PCh. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How could you distinguish the 1H NMR spectra of...Ch. 14.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 14.5 - Prob. 7PCh. 14.5 - Prob. 8PCh. 14.5 - Prob. 9PCh. 14.5 - Where would you expect to find the 1H NMR signal...
Ch. 14.6 - Prob. 11PCh. 14.7 - Prob. 12PCh. 14.7 - Prob. 13PCh. 14.7 - Without referring to Table 14.1, label the proton...Ch. 14.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 14.9 - How would integration distinguish the 1H NMR...Ch. 14.9 - Which of the following compounds is responsible...Ch. 14.10 - Prob. 19PCh. 14.10 - Explain how the following compounds, each with the...Ch. 14.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 14.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 14.12 - Indicate the number of signals and the...Ch. 14.12 - Explain the relative chemical shifts of the...Ch. 14.12 - How can their 1H NMR spectra distinguish the...Ch. 14.12 - Identify each compound from its molecular formula...Ch. 14.12 - Predict the splitting patterns for the signals...Ch. 14.12 - Describe the 1H NMR spectrum you would expect for...Ch. 14.12 - Propose structures that are consistent with the...Ch. 14.13 - Prob. 30PCh. 14.13 - Identify the compound with molecular formula...Ch. 14.14 - Prob. 32PCh. 14.15 - a. For the following compounds, which pairs of...Ch. 14.15 - How would the 1H NMR spectra for the four...Ch. 14.17 - Explain why the chemical shift of the OH proton of...Ch. 14.17 - Prob. 38PCh. 14.17 - Prob. 39PCh. 14.17 - Prob. 40PCh. 14.20 - Answer the following questions for each compound:...Ch. 14.20 - Prob. 42PCh. 14.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 14.20 - Identify each compound below from its molecular...Ch. 14.22 - Prob. 45PCh. 14.22 - What does cross peak X in Figure 14.34 tell you?Ch. 14 - Prob. 47PCh. 14 - Draw a spitting diagram for the Hb proton and give...Ch. 14 - Label each set of chemically equivalent protons,...Ch. 14 - Determine the ratios of the chemically...Ch. 14 - How can 1H NMR distinguish between the compounds...Ch. 14 - Prob. 52PCh. 14 - Match each of the 1H NMR spectra with one of the...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Prob. 55PCh. 14 - Prob. 56PCh. 14 - Compound A, with molecular formula C4H9Cl, shows...Ch. 14 - Would it be better to use 1H NMR or 13C NMR...Ch. 14 - There are four esters with molecular formula...Ch. 14 - Identify the compound with molecular formula C6H14...Ch. 14 - An alkyl halide reacts with an alkoxide ion to...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 65PCh. 14 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 14 - The 1H NMR spectra of two compounds, each with...Ch. 14 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 14 - Sketch the following spectra that would be...Ch. 14 - How can 1H NMR be used to prove that the addition...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 14 - Calculate the amount of energy (in calories)...Ch. 14 - The following 1H NMR spectra are four compounds,...Ch. 14 - When compound A (C5H12O) is treated with HBr, it...Ch. 14 - Identity the compound with molecular formula...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 78PCh. 14 - Identify each of the following compounds from its...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A DEPT NMR spectrum is shown for a molecule with the molecular formula of C5H12O. Draw the structure that best fits this data. 200 180 160 140 120 100 一盆 00 40 8- 20 ppm 0 Qarrow_forwardDon't used hand raitingarrow_forwardShown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H. H. +N=C H H H Cl: Click and drag to start drawing a structure. : ? g B S olo Ar B Karrow_forward
- Don't used hand raitingarrow_forwardS Shown below is the major resonance structure for a molecule. Draw the second best resonance structure of the molecule. Include all non-zero formal charges. H H = HIN: H C. :0 H /\ H H Click and drag to start drawing a structure. ×arrow_forwardPlease help me figure out these calculation and what should be plotted. These are notes for my chemistry class.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY