
Organic Chemistry As a Second Language: First Semester Topics
4th Edition
ISBN: 9781119110668
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 14.4, Problem 14.21P
Interpretation Introduction
Interpretation:
The products that is expected to be formed when the given compound is treated with
Concept Introduction:
Ether are compounds that contain an oxygen atom that is sandwiched between two R groups. The R groups may be alkyl, vinyl or aryl.

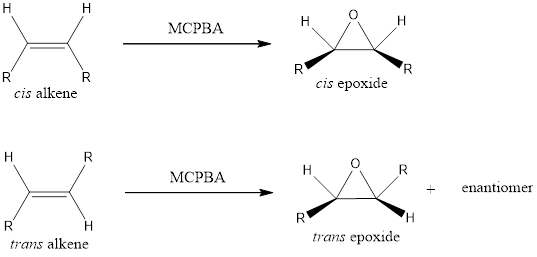
Epoxide formation from an alkene is a stereospecific process. This means a cis alkene gets converted to cis epoxide and trans alkene gets converted to trans epoxide on treatment with peroxy acids.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Pls help asap
What reagents do you need to
complete the transformation in order
(show all work)
→
No₂ Br
Pls help asap
Chapter 14 Solutions
Organic Chemistry As a Second Language: First Semester Topics
Ch. 14.1 - Prob. 14.1PCh. 14.1 - Prob. 14.2PCh. 14.1 - Prob. 14.3PCh. 14.1 - Prob. 14.4PCh. 14.1 - Prob. 14.5PCh. 14.1 - Prob. 14.6PCh. 14.1 - Prob. 14.7PCh. 14.2 - Prob. 14.9PCh. 14.2 - Prob. 14.10PCh. 14.2 - Prob. 14.11P
Ch. 14.2 - Prob. 14.12PCh. 14.2 - Prob. 14.13PCh. 14.3 - Prob. 14.14PCh. 14.3 - Prob. 14.15PCh. 14.3 - Prob. 14.16PCh. 14.3 - Prob. 14.17PCh. 14.4 - Prob. 14.18PCh. 14.4 - Prob. 14.19PCh. 14.4 - Prob. 14.20PCh. 14.4 - Prob. 14.21PCh. 14.4 - Prob. 14.22PCh. 14.4 - Prob. 14.23PCh. 14.4 - Prob. 14.24PCh. 14.4 - Prob. 14.25PCh. 14.5 - Prob. 14.27PCh. 14.5 - Prob. 14.28PCh. 14.5 - Prob. 14.29PCh. 14.5 - Prob. 14.30PCh. 14.5 - Prob. 14.31PCh. 14.5 - Prob. 14.32P
Knowledge Booster
Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY