
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 3LGP
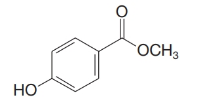
The
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
potential energy
Br
b) Translate the Newman projection below to its wedge-and-dash drawing.
F
H.
OH
CH3
CI
c) Isopentane (2-methylbutane) is a compound containing a branched carbon chain. Draw
a Newman projection of six conformations about the C2-C3 bond of isopentane. On the
curve of potential energy versus angle of internal rotation for isopentane, label each
energy maximum and minimum with one of the conformations.
0°
。
F
A
B
D
C
angle of internal rotation
E
F
360° (=0°)
JDownl
Q7: Identify the functional groups in these molecules
a)
CH 3
b) Aspirin:
HO
'N'
Capsaicin
HO
O
CH3
CH 3
Q2: Name the following alkanes
Chapter 14 Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Ch. 14 - PRACTICE PROBLEM 14.1 Provide a name for each of...Ch. 14 - Prob. 2PPCh. 14 - Prob. 3PPCh. 14 - Practice Problem 14.4 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.5 Apply the polygon-and-circle...Ch. 14 - Practice Problem 14.6 1,3,5-Cycloheptatriene is...Ch. 14 - Prob. 7PPCh. 14 - Prob. 8PPCh. 14 - Practice Problem 14.9 In 1967 R. Breslow (of...Ch. 14 - Prob. 10PP
Ch. 14 - Practice Problem 14.11 In addition to a signal...Ch. 14 - PRACTICE PROBLEM 14.12
Azulene has an appreciable...Ch. 14 - Practice Problem 14.13 (a) The -Sh group is...Ch. 14 - Practice Problem 14.14
Explain how NMR...Ch. 14 - PRACTICE PROBLEM 14.15 Four benzenoid compounds,...Ch. 14 - Prob. 16PCh. 14 - Write structural formulas and give acceptable...Ch. 14 - Prob. 18PCh. 14 - Prob. 19PCh. 14 - Prob. 20PCh. 14 - Which of the hydrogen atoms shown below is more...Ch. 14 - 14.22 The rings below are joined by a double bond...Ch. 14 - Prob. 23PCh. 14 - 14.24 (a) In 1960 T. Katz (Columbia University)...Ch. 14 - Prob. 25PCh. 14 - Prob. 26PCh. 14 - 14.27 5-Chloro-1,3-cyclopentadiene (below)...Ch. 14 - Prob. 28PCh. 14 - Furan possesses less aromatic character than...Ch. 14 - 14.30 For each of the pairs below, predict...Ch. 14 - Assign structures to each of the compounds A, B,...Ch. 14 - Prob. 32PCh. 14 - Give a structure for compound F that is consistent...Ch. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - The IR and 1H NMR spectra for compound X(C8H10)...Ch. 14 - Prob. 37PCh. 14 - Prob. 38PCh. 14 - 14.39 Given the following information, predict the...Ch. 14 - Consider these reactions: The intermediate A is a...Ch. 14 - Prob. 41PCh. 14 - Compound E has the spectral features given below....Ch. 14 - Draw all of the molecular orbitals for...Ch. 14 - Prob. 1LGPCh. 14 - Prob. 2LGPCh. 14 - 3. The NMR signals for the aromatic hydrogens of...Ch. 14 - Prob. 4LGPCh. 14 - Prob. 5LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Glycine has pK2 values of 2.34 and 9.60. At what pH does glycine exist in the forms shown?
Organic Chemistry (8th Edition)
30. Consider the unbalanced equation for the reaction of aluminum with sulfuric acid:
a. Balance the equation...
Introductory Chemistry (6th Edition)
What is the inducer for the lac operon?
Microbiology: Principles and Explorations
The genotype of F1, individuals in a tetrahybrid cross is AaBbCcDd. Assuming lndependent assortment of these fo...
Campbell Biology (11th Edition)
3. What is free-fall, and why does it make you weightless? Briefly describe why astronauts are weightless in th...
The Cosmic Perspective (8th Edition)
Police Captain Jeffers has suffered a myocardial infarction. a. Explain to his (nonmedically oriented) family w...
Human Physiology: An Integrated Approach (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Complete the following table in your laboratory notebook. Substance Formula Methanol CH3OH Ethanol C2H5OH 1-Propanol C3H7OH 1-Butanol C4H9OH Pentane C5H12 Hexane C6H14 Water H₂O Acetone C3H60 Structural Formula Molecular Weight (g/mol) Hydrogen Bond (Yes or No)arrow_forwardQ1: Compare the relative acidity in each pair of compounds. Briefly explain. (a) CH3OH vs NH 3 (b) HF vs CH3COOH (c) NH3 vs CH4 (d) HCI vs HI (e) CH3COOH vs CH3SH (f) H₂C=CH2 vs CH3 CH3 (g) compare the acidity of the two bolded hydrogens O. H N- (h) compare the acidity of the two bolded hydrogens, draw resonance structures to explain H H Harrow_forwardQ3: Rank the following molecules in order of decreasing boiling point: (a) 3-methylheptane; (b) octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.arrow_forward
- Q5: Conformations of Alkanes a) Draw a Newman Projection of the compound below about the C2-C3 bond. H3C Cli... H IIIH Br CH3arrow_forwardThe ability of atoms to associate with each other depends ona) the electronic structure and its spatial orientation.b) the electron affinity.c) The other two answers are correct.arrow_forwardWhat is the final volume after you reach the final temperature? I put 1.73 but the answer is wrong not sure why The initial volume of gas is 1.60 LL , the initial temperature of the gas is 23.0 °C°C , and the system is in equilibrium with an external pressure of 1.2 bar (given by the sum of a 1 bar atmospheric pressure and a 0.2 bar pressure due to a brick that rests on top of the piston). Then, as you did in Exercise 1, you heat the gas slowly until the temperature reaches 48.2 °Carrow_forward
- Q4: Identify the type of Carbon ( methyl, primary, secondary, etc. ) indicated by this arrow.arrow_forwardQ3: Curved Arrows, Lewis Acids & Bases, Nucleophiles and Electrophiles Considering the following reactions: a) Predict the products to complete the reactions. b) Use curved electron-pushing arrows to show the mechanism for the reaction in the forward direction. Redraw some of the compounds to explicitly illustrate all bonds that are broken and all bonds that are formed. c) Label Lewis acids and bases, nucleophiles and electrophiles in the reactions. A. S + AICI 3 B. + H₂Oarrow_forward3. A thermometer is placed in a test tube of chipped ice at -5.0 °C. The temperature is recorded at the time intervals shown below until room temperature is reached. Plot the data given below on graph paper and explain all flat, horizontal portions of the curve. Plot time on the X-axis! Time (min) Temperature (°C) 0 -5.0 2 -2.5 4 -1.0 6 0.0 10 0.0 15 0.0 20 0.0 25 0.0 30 1.5 35 4.0 40 8.0 45 11.5 50 15.0 55 17.5 60 19.0 65 20.0 70 20.0 75 20.0 80 20.0arrow_forward
- Naming the Alkanes a) Write the IUPAC nomenclature of the compound below b) Draw 4-isopropyl-2,4,5-trimethylheptane, identify the primary, secondary, tertiary, and quaternary carbons. c) Rank pentane, neopentane and isopentane for boiling point. pentane: H3C-CH2-CH2-CH2-CH3 neopentane: CH3 H3C-Ċ-CH3 I CH3 isopentane: CH3 H3C-CH2-CH-CH3arrow_forwardWhich will evaporate faster, 1-Butanol or Pentane? Explain your choice.arrow_forwardUsing the equation below, what is the rate of this reaction if the rate of disappearance of H2 is 0.44 M/sec? H2 + Br2 → 2HBrarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY