
Organic Chemistry, Binder Ready Version
2nd Edition
ISBN: 9781118454312
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 14, Problem 32PP
Interpretation Introduction
Interpretation: To construct all the possible constitutional isomers of ether for the molecular formula
Concept Introduction:
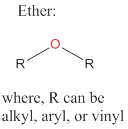
Ethers: Organic compounds that exhibit an oxygen atom bonded to two R groups, where each R groups can be an alkyl, aryl, or vinyl group.
HDI calculation: The HDI value calculated from the molecular formula is used to predict the degree of unsaturation in the structure.
To identify: construct all the possible constitutional isomers of a molecular formula
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the pH of a 0.01m solution of acetic acid use pka of 4.75
What is the product of the reaction?
F3C.
CF3
OMe
NaOH / H₂O
What is the product of the reaction?
F3C.
CF3
OMe
NaOH / H₂O
Chapter 14 Solutions
Organic Chemistry, Binder Ready Version
Ch. 14.2 - Prob. 1LTSCh. 14.2 - Prob. 1PTSCh. 14.2 - Prob. 2ATSCh. 14.2 - Prob. 3ATSCh. 14.4 - Prob. 4CCCh. 14.5 - Prob. 2LTSCh. 14.5 - Prob. 5PTSCh. 14.5 - Prob. 6ATSCh. 14.5 - Prob. 7ATSCh. 14.5 - Prob. 8CC
Ch. 14.5 - Prob. 9CCCh. 14.5 - Prob. 10CCCh. 14.6 - Prob. 11CCCh. 14.7 - Prob. 12CCCh. 14.7 - Prob. 13CCCh. 14.8 - Prob. 3LTSCh. 14.8 - Prob. 14PTSCh. 14.8 - Prob. 15ATSCh. 14.9 - Prob. 16CCCh. 14.10 - Prob. 4LTSCh. 14.10 - Prob. 17PTSCh. 14.10 - Prob. 18ATSCh. 14.10 - Prob. 19ATSCh. 14.10 - Prob. 5LTSCh. 14.10 - Prob. 20PTSCh. 14.10 - Prob. 21ATSCh. 14.11 - Prob. 22CCCh. 14.11 - Prob. 23CCCh. 14.12 - Prob. 6LTSCh. 14.12 - Prob. 24PTSCh. 14.12 - Prob. 25ATSCh. 14.12 - Prob. 7LTSCh. 14.12 - Prob. 26PTSCh. 14.12 - Prob. 27ATSCh. 14.12 - Prob. 28ATSCh. 14.12 - Prob. 29ATSCh. 14 - Prob. 30PPCh. 14 - Prob. 31PPCh. 14 - Prob. 32PPCh. 14 - Prob. 33PPCh. 14 - Prob. 34PPCh. 14 - Prob. 35PPCh. 14 - Prob. 36PPCh. 14 - Prob. 37PPCh. 14 - Prob. 38PPCh. 14 - Prob. 39PPCh. 14 - Prob. 40PPCh. 14 - Prob. 41PPCh. 14 - Prob. 42PPCh. 14 - Prob. 43PPCh. 14 - Prob. 44PPCh. 14 - Prob. 45PPCh. 14 - Prob. 46PPCh. 14 - Prob. 47PPCh. 14 - Prob. 48PPCh. 14 - Prob. 49PPCh. 14 - Prob. 50PPCh. 14 - Prob. 51IPCh. 14 - Prob. 52IPCh. 14 - Prob. 53IPCh. 14 - Prob. 54IPCh. 14 - Prob. 55IPCh. 14 - Prob. 56IPCh. 14 - Prob. 57IPCh. 14 - Prob. 58IPCh. 14 - Prob. 59IPCh. 14 - Prob. 60IPCh. 14 - Prob. 61IPCh. 14 - Prob. 62IPCh. 14 - Prob. 63IP
Knowledge Booster
Similar questions
- What would you expect to be the major product obtained from the following reaction? Please explain what is happening here. Provide a detailed explanation and a drawing showing how the reaction occurs. The correct answer to this question is V.arrow_forwardPlease answer the question for the reactions, thank youarrow_forwardWhat is the product of the following reaction? Please include a detailed explanation of what is happening in this question. Include a drawing showing how the reagent is reacting with the catalyst to produce the correct product. The correct answer is IV.arrow_forward
- Please complete the reactions, thank youarrow_forwardConsider the synthesis. What is compound Y? Please explain what is happening in this question. Provide a detailed explanation and a drawing to show how the compound Y creates the product. The correct answer is D.arrow_forwardWhat would be the major product of the following reaction? Please include a detailed explanation of what is happening in this question. Include steps and a drawing to show this reaction proceeds and how the final product is formed. The correct answer is B. I put answer D and I don't really understand what is going on in the question.arrow_forward
- What is the product of the following reaction? Please explain what is happening in this question. Provide a detailed explanation and a drawing showing how the reagent is reacting with the catalysts to product the correct product. The correct answer is B.arrow_forwardWhat is the missing intermediate 1 and the final product 2. Please include a detailed explanation explaining the steps of malonic ester synthesis. Please include drawings of the intermediate and how it occurs and how the final product is former.arrow_forwardWhat would be the reagents and conditions above and below the arrow that will complete the proposed acetoacetic ester synthesis? If it cannot be done efficiently, then I will choose that answer. There could be 2 or 4 reagents involved. Please provide a detailed explanation and drawings showing how it would proceed with the correct reagents.arrow_forward
- For benzene, the ∆H° of vaporization is 30.72 kJ/mol and the ∆S° of vaporization is 86.97 J/mol・K. At 1.00 atm and 228.0 K, what is the ∆G° of vaporization for benzene, in kJ/mol?arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reaction. it is spontaneous only at High T, it is spontaneous at low T it is nonspontaneous at all T it is spontanrous at all T. it is non spontaneous only at low T.arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reactionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY