
Study Guide And Selected Solutions Manual For Chemistry Format: Paperback
13th Edition
ISBN: 9780134553986
Author: Timberlake, Karen C
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.2, Problem 13.18PP
Identify each of the following as D or L:
a.
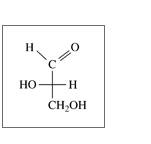
b.
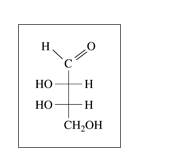
c.
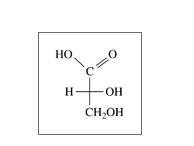
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Drawing Arrows
1
I
I
1
heat
1
51
MO
+
Drag To
Und Settings
Done
0 0
Jan 31
3:5
Don't used hand raiting
Gramicidin A can adopt more than one structure; NMR spectroscopy has revealed an “end-to-end” dimer form, and x-ray crystallography has revealed an “anti-parallel double- helical” form. Briefly outline and describe an experimentalapproach/strategy to investigate WHICH configuration (“end-to-end dimer” vs “anti-paralleldouble helical”) gramicidin adopts in an actual lipid bilayer.
Chapter 13 Solutions
Study Guide And Selected Solutions Manual For Chemistry Format: Paperback
Ch. 13.1 - Prob. 13.1PPCh. 13.1 - Prob. 13.2PPCh. 13.1 - Prob. 13.3PPCh. 13.1 - Prob. 13.4PPCh. 13.1 - Prob. 13.5PPCh. 13.1 - Prob. 13.6PPCh. 13.1 - Prob. 13.7PPCh. 13.1 - Prob. 13.8PPCh. 13.1 - Prob. 13.9PPCh. 13.1 - Prob. 13.10PP
Ch. 13.2 - Prob. 13.11PPCh. 13.2 - Prob. 13.12PPCh. 13.2 - Prob. 13.13PPCh. 13.2 - Prob. 13.14PPCh. 13.2 - Prob. 13.15PPCh. 13.2 - Prob. 13.16PPCh. 13.2 - Identify each of the following as D or L: a. b. c.Ch. 13.2 - Identify each of the following as D or L: a. b. c.Ch. 13.2 - Identify the chiral carbon in each of the...Ch. 13.2 - Prob. 13.20PPCh. 13.3 - Prob. 13.21PPCh. 13.3 - Prob. 13.22PPCh. 13.3 - Prob. 13.23PPCh. 13.3 - Prob. 13.24PPCh. 13.3 - Prob. 13.25PPCh. 13.3 - Prob. 13.26PPCh. 13.3 - Prob. 13.27PPCh. 13.3 - Prob. 13.28PPCh. 13.3 - Prob. 13.29PPCh. 13.3 - Prob. 13.30PPCh. 13.4 - What are the kind and number of atoms in the ring...Ch. 13.4 - What are the kind and number of atoms in the ring...Ch. 13.4 - Prob. 13.33PPCh. 13.4 - Prob. 13.34PPCh. 13.4 - Prob. 13.35PPCh. 13.4 - Identify each of the following as the a or ß...Ch. 13.5 - Prob. 13.37PPCh. 13.5 - Prob. 13.38PPCh. 13.5 - Prob. 13.39PPCh. 13.5 - Prob. 13.40PPCh. 13.6 - Prob. 13.41PPCh. 13.6 - Prob. 13.42PPCh. 13.6 - Prob. 13.43PPCh. 13.6 - Prob. 13.44PPCh. 13.6 - Prob. 13.45PPCh. 13.6 - Prob. 13.46PPCh. 13.7 - Describe the similarities and differences in the...Ch. 13.7 - Prob. 13.48PPCh. 13.7 - Prob. 13.49PPCh. 13.7 - Prob. 13.50PPCh. 13.7 - Prob. 13.51PPCh. 13.7 - Prob. 13.52PPCh. 13.7 - Prob. 13.53PPCh. 13.7 - Prob. 13.54PPCh. 13 - Prob. 13.55UTCCh. 13 - Prob. 13.56UTCCh. 13 - Prob. 13.57UTCCh. 13 - Prob. 13.58UTCCh. 13 - Prob. 13.59UTCCh. 13 - Prob. 13.60UTCCh. 13 - Prob. 13.61APPCh. 13 - Prob. 13.62APPCh. 13 - Prob. 13.63APPCh. 13 - Prob. 13.64APPCh. 13 - Prob. 13.65APPCh. 13 - Prob. 13.66APPCh. 13 - Prob. 13.67APPCh. 13 - Prob. 13.68APPCh. 13 - Prob. 13.69APPCh. 13 - Prob. 13.70APPCh. 13 - Prob. 13.71APPCh. 13 - Prob. 13.72APPCh. 13 - Prob. 13.73APPCh. 13 - Prob. 13.74APPCh. 13 - Prob. 13.75CPCh. 13 - Prob. 13.76CPCh. 13 - 13.77 Gentiobiose is found in saffron. (13.4,...Ch. 13 - Prob. 13.78CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Don't used hand raitingarrow_forwardCHEM2323 Problem 2-24 Tt O e: ל Predict the product(s) of the following acid/base reactions. Draw curved arrows to show the formation and breaking of bonds. If the bonds needed are not drawn out, you should redraw them. + BF3 (a) (b) HI + (c) OH -BF Problem 2-25 Use curved arrows and a proton (H+) to draw the protonated form of the following Lewis bases. Before starting, add all missing lone pairs. (a) (b) :0: (c) N 1 CHEM2323 PS CH02 Name:arrow_forwardCHEM2323 Problem 2-26 Tt O PS CH02 Name: Use the curved-arrow formalism to show how the electrons flow in the resonance form on the left to give the one on the right. (Draw all lone pairs first) (a) NH2 NH2 + (b) Problem 2-27 Double bonds can also act like Lewis bases, sharing their electrons with Lewis acids. Use curved arrows to show how each of the following double bonds will react with H-Cl and draw the resulting carbocation. (a) H2C=CH2 (b) (c) Problem 2-28 Identify the most electronegative element in each of the following molecules: (a) CH2FCI F Problem 2-29 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li F 0 0 Use the electronegativity table in Figure 2.3 to predict which bond in the following pairs is more polar and indicate the direction of bond polarity for each compound. (a) H3C-Cl or Cl-CI (b) H3C-H or H-CI (c) HO-CH3 or (CH3)3Si-CH3 (d) H3C-Li or Li-OHarrow_forward
- Reagan is doing an atomic absorption experiment that requires a set of zinc standards in the 0.4-1.6 ppm range. A 1000 ppm Zn solution was prepared by dissolving the necessary amount of solid Zn(NO3)2 in water. The standards can be prepared by diluting the 1000 ppm Zn solution. Table 1 shows one possible set of serial dilutions (stepwise dilution of a solution) that Reagan could perform to make the necessary standards. Solution A was prepared by diluting 5.00 ml of the 1000 ppm Zn standard to 50.00 ml. Solutions C-E are called "calibration standards" because they will be used to calibrate the atomic absorption spectrometer. a. Compare the solution concentrations expressed as ppm Zn and ppm Zn(NO3)2. Compare the concentrations expressed as M Zn and M Zn(NO3)2 - Which units allow easy conversion between chemical species (e.g. Zn and Zn(NO3)2)? - Which units express concentrations in numbers with easily expressed magnitudes? - Suppose you have an analyte for which you don't know the molar…arrow_forwardNonearrow_forwardHow will you prepare the following buffers? 2.5 L of 1.5M buffer, pH = 10.5 from NH4Cl and NH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY