
BURDGE CHEMISTRY VALUE ED (LL)
4th Edition
ISBN: 9781259995958
Author: VALUE EDITION
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 136AP
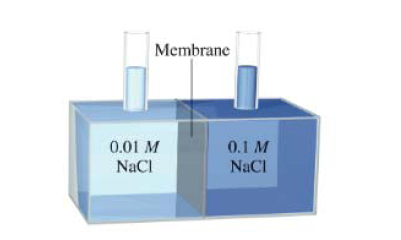
In the apparatus shown, what will happen if the membrane is (a) permeable to both water and the
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
>
each pair of substrates below, choose the one that will react faster in a substitution reaction, assuming that:
1. the rate of substitution doesn't depend on nucleophile concentration and
2. the products are a roughly 50/50 mixture of enantiomers.
Substrate A
Substrate B
Faster Rate
X
Ś
CI
(Choose one)
(Choose one)
CI
Br
Explanation
Check
Br
(Choose one)
© 2025 McGraw Hill LLC. All Rights F
NMR spectrum of ethyl acetate has signals whose chemical shifts are indicated below. Which hydrogen or set of hydrogens corresponds to the signal at
4.1 ppm? Select the single best answer.
The
H
O
HỌC—C—0—CH, CH,
2
A
ethyl acetate
H NMR: 1.3 ppm, 2.0 ppm, 4.1 ppm
Check
OA
B
OC
ch
B
C
Save For Later
Submit Ass
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center |
How many signals do you expect in the H NMR spectrum for this molecule?
Br Br
Write the answer below.
Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H
atoms that would contribute to the same signal as the H already highlighted red
Note for advanced students: In this question, any multiplet is counted as one signal.
1
Number of signals in the 'H NMR spectrum.
For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to
the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
Check
For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute
to the same signal as the H atom already highlighted red.
If no other H atoms will contribute, check the box at right.
O
✓
No additional Hs to color in top
molecule
ง
No additional Hs to color in bottom…
Chapter 13 Solutions
BURDGE CHEMISTRY VALUE ED (LL)
Ch. 13.1 - Prob. 1PPACh. 13.1 - Prob. 1PPBCh. 13.1 - Prob. 1PPCCh. 13.2 - Prob. 1CPCh. 13.2 - Prob. 2CPCh. 13.2 - Practice Problem ATTEMPT
Determine (a) the...Ch. 13.2 - Practice Problem BUILD
Determine the molality of...Ch. 13.2 - Prob. 1PPCCh. 13.3 - Practice Problem ATTEMPT
An aqueous solution that...Ch. 13.3 - Practice Problem BUILD
Determine the percent...
Ch. 13.3 - Practice Problem CONCEPTUALIZE
The diagrams...Ch. 13.3 - Prob. 1CPCh. 13.3 - What is the molality of a solution prepared by...Ch. 13.3 - Prob. 3CPCh. 13.3 - Prob. 4CPCh. 13.4 - Practice ProblemATTEMPT Calculate the...Ch. 13.4 - Prob. 1PPBCh. 13.4 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.4 - The solubility of N2 in water at 25°C and an N 2...Ch. 13.4 - Calculate the molar concentration of O 2 in water...Ch. 13.5 - Practice ProblemATTEMPT Calculate the vapor...Ch. 13.5 - Prob. 1PPBCh. 13.5 - Practice ProblemCONCEPTUALIZE The diagrams...Ch. 13.5 - 13.5.1 A solution contains 75.0 g of glucose...Ch. 13.5 - Determine the boiling point and the freezing point...Ch. 13.5 - 13.5.3 Calculate the osmotic pressure of a...Ch. 13.5 - 13.5.4 A 1.00-m solution of has a freezing point...Ch. 13.6 - Prob. 1PPACh. 13.6 - Prob. 1PPBCh. 13.6 - Practice Problem CONCEPTUALIZE
The diagrams...Ch. 13.6 - 13.6.1 A solution made by dissolving 14.2 g of...Ch. 13.6 - Prob. 2CPCh. 13.7 - Practice ProblemATTEMPT The freezing-point...Ch. 13.7 - Practice ProblemBUILD Using the experimental van't...Ch. 13.7 - Practice Problem CONCEPTUALIZE
The diagram...Ch. 13.8 - Practice ProblemATTEMPT Determine the osmotic...Ch. 13.8 - Practice Problem BUILD
Determine the...Ch. 13.8 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.9 - Practice Problem ATTEMPT
Calculate the molar mass...Ch. 13.9 - Practice Problem BUILD
What mass of naphthalene...Ch. 13.9 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.10 - Practice Problem ATTEMPT A solution made by...Ch. 13.10 - Practice Problem BUILD What mass of insulin must...Ch. 13.10 - Practice ProblemCONCEPTUALIZE The first diagram...Ch. 13.11 - Practice Problem ATTEMPT An aqueous solution that...Ch. 13.11 - Practice Problem BUILD
An aqueous solution that is...Ch. 13.11 - Practice Problem CONCEPTUALIZE The diagrams...Ch. 13 - Which of the following processes is accompanied by...Ch. 13 - 13.2
For each of the processes depicted here,...Ch. 13 - 13.3
For each of the processes depicted here,...Ch. 13 - Prob. 4KSPCh. 13 - Describe and give examples of an unsaturated...Ch. 13 - Prob. 2QPCh. 13 - Prob. 3QPCh. 13 - Prob. 4QPCh. 13 - Prob. 5QPCh. 13 - As you know, some solution processes are...Ch. 13 - Prob. 7QPCh. 13 - 13.8 Describe the factors that affect the...Ch. 13 - Prob. 9QPCh. 13 - Prob. 10QPCh. 13 - Prob. 11QPCh. 13 - Prob. 12QPCh. 13 - Prob. 13QPCh. 13 - Prob. 14QPCh. 13 - Prob. 15QPCh. 13 - Prob. 16QPCh. 13 - Prob. 17QPCh. 13 - Prob. 18QPCh. 13 - Prob. 19QPCh. 13 - Prob. 20QPCh. 13 - 13.21 The alcohol content of hard liquor is...Ch. 13 - Prob. 22QPCh. 13 - Prob. 23QPCh. 13 - 13.24 The density of an aqueous solution...Ch. 13 - Prob. 25QPCh. 13 - Prob. 26QPCh. 13 - Prob. 27QPCh. 13 - What is thermal pollution? Why is it harmful to...Ch. 13 - Prob. 29QPCh. 13 - A student is observing two beakers of water. One...Ch. 13 - Prob. 31QPCh. 13 - Prob. 32QPCh. 13 - The solubility of KNO 3 is 155 g per 100 g of...Ch. 13 - Prob. 34QPCh. 13 - 13.35 The solubility of in water at What is its...Ch. 13 - Prob. 36QPCh. 13 - Prob. 37QPCh. 13 - Prob. 38QPCh. 13 - Prob. 39QPCh. 13 - Prob. 40QPCh. 13 - Prob. 41QPCh. 13 - Prob. 42QPCh. 13 - Prob. 43QPCh. 13 - Prob. 44QPCh. 13 - Prob. 45QPCh. 13 - 13.46 Write the equations relating boiling-point...Ch. 13 - Prob. 47QPCh. 13 - Prob. 48QPCh. 13 - Prob. 49QPCh. 13 - Prob. 50QPCh. 13 - Prob. 51QPCh. 13 - Prob. 52QPCh. 13 - Prob. 53QPCh. 13 - What are ion pairs? What effect does ion-pair...Ch. 13 - Prob. 55QPCh. 13 - Prob. 56QPCh. 13 - 13.57 A solution is prepared by dissolving 396 g...Ch. 13 - Prob. 58QPCh. 13 - Prob. 59QPCh. 13 - Prob. 60QPCh. 13 - Prob. 61QPCh. 13 - Prob. 62QPCh. 13 - Prob. 63QPCh. 13 - 13.64 How many liters of the antifreeze ethylene...Ch. 13 - Prob. 65QPCh. 13 - Prob. 66QPCh. 13 - Prob. 67QPCh. 13 - Prob. 68QPCh. 13 - 13.69 Both and are used to melt ice on roads and...Ch. 13 - Prob. 70QPCh. 13 - Prob. 71QPCh. 13 - Prob. 72QPCh. 13 - Prob. 73QPCh. 13 - Calculate the difference in osmotic pressure (in...Ch. 13 - 13.75 Which of the following aqueous solutions has...Ch. 13 - Prob. 76QPCh. 13 - 13.77 Arrange the following solutions in order of...Ch. 13 - Prob. 78QPCh. 13 - Indicate which compound in each of the following...Ch. 13 - Prob. 80QPCh. 13 - Prob. 81QPCh. 13 - Prob. 82QPCh. 13 - Prob. 83QPCh. 13 - The elemental analysis of an organic solid...Ch. 13 - 13.85 A solution of 2.50 g of a compound having...Ch. 13 - 13.86 The molar mass of benzoic acid determined...Ch. 13 - 13.87 A solution containing 0.8330 g of a polymer...Ch. 13 - Prob. 88QPCh. 13 - A solution of 6.85 g of a carbohydrate in 100.0 g...Ch. 13 - Prob. 90QPCh. 13 - Prob. 91QPCh. 13 - Prob. 92QPCh. 13 - Prob. 93QPCh. 13 - Prob. 94QPCh. 13 - Prob. 95APCh. 13 - Prob. 96APCh. 13 - 13.97 Acetic acid is a polar molecule and can form...Ch. 13 - Prob. 98APCh. 13 - Prob. 99APCh. 13 - Prob. 100APCh. 13 - Prob. 101APCh. 13 - Prob. 102APCh. 13 - Prob. 103APCh. 13 - Prob. 104APCh. 13 - Prob. 105APCh. 13 - A solution of 1.00 g of anhydrous aluminum...Ch. 13 - Explain why reverse osmosis is (theoretically)...Ch. 13 - A 1.32-g sample of a mixture of cyclohexane ( C 6...Ch. 13 - Prob. 109APCh. 13 - Prob. 110APCh. 13 - Prob. 111APCh. 13 - Prob. 112APCh. 13 - Prob. 113APCh. 13 - Prob. 114APCh. 13 - Prob. 115APCh. 13 - Iodine ( I 2 ) is only sparingly soluble in water...Ch. 13 - Concentrated hydrochloric acid is usually...Ch. 13 - Explain each of the following statements: (a) The...Ch. 13 - A mixture of NaCl and sucrose ( C 12 H 22 O 12 )...Ch. 13 - Prob. 120APCh. 13 - At 27°C, the vapor pressure of pure water is 23.76...Ch. 13 - A nonvolatile organic compound Z was used to make...Ch. 13 - Prob. 123APCh. 13 - Prob. 124APCh. 13 - Prob. 125APCh. 13 - Prob. 126APCh. 13 - Prob. 127APCh. 13 - Prob. 128APCh. 13 - Prob. 129APCh. 13 - Prob. 130APCh. 13 - Prob. 131APCh. 13 - Consider the three mercury manometers shown in the...Ch. 13 - Prob. 133APCh. 13 - Prob. 134APCh. 13 - Prob. 135APCh. 13 - 13.136 In the apparatus shown, what will happen if...Ch. 13 - Prob. 137APCh. 13 - Prob. 138APCh. 13 - Lysozyme is an enzyme that cleaves bacterial cell...Ch. 13 - Prob. 140APCh. 13 - Prob. 141APCh. 13 - Prob. 142APCh. 13 - Prob. 143APCh. 13 - Prob. 144APCh. 13 - Prob. 145APCh. 13 - What masses of sodium chloride, magnesium...Ch. 13 - Prob. 147APCh. 13 - Prob. 148APCh. 13 - Prob. 149APCh. 13 - Hemoglobin, the oxygen-transport protein, binds...Ch. 13 - Prob. 151APCh. 13 - 13.152 The vapor pressure of ethanol and the...Ch. 13 - Prob. 153APCh. 13 - A mixture of two volatile liquids is said to be...Ch. 13 - A mixture of two volatile liquids is said to be...Ch. 13 - Prob. 3SEPPCh. 13 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- in the kinetics experiment, what were the values calculated? Select all that apply.a) equilibrium constantb) pHc) order of reactiond) rate contstantarrow_forwardtrue or false, given that a 20.00 mL sample of NaOH took 24.15 mL of 0.141 M HCI to reach the endpoint in a titration, the concentration of the NaOH is 1.17 M.arrow_forwardin the bromothymol blue experiment, pKa was measured. A closely related compound has a Ka of 2.10 x 10-5. What is the pKa?a) 7.1b) 4.7c) 2.0arrow_forward
- calculate the equilibrium concentration of H2 given that K= 0.017 at a constant temperature for this reaction. The inital concentration of HBr is 0.050 M.2HBr(g) ↔ H2(g) + Br2(g)a) 4.48 x 10-2 M b) 5.17 x 10-3 Mc) 1.03 x 10-2 Md) 1.70 x 10-2 Marrow_forwardtrue or falsegiven these two equilibria with their equilibrium constants:H2(g) + CI2(l) ↔ 2HCI(g) K= 0.006 CI2(l) ↔ CI2(g) K= 0.30The equilibrium contstant for the following reaction is 1.8H2(g) + CI2 ↔ 2HCI(g)arrow_forwardI2(g) + CI2(g) ↔ 2ICIK for this reaction is 81.9. Find the equilibrium concentration of I2 if the inital concentration of I2 and CI2 are 0.010 Marrow_forward
- true or false,the equilibrium constant for this reaction is 0.50.PCI5(g) ↔ PCI3(g) + CI2(g)Based on the above, the equilibrium constant for the following reaction is 0.25.2PCI5(g) ↔. 2PCI3(g) + 2CI2(g)arrow_forwardtrue or false, using the following equilibrium, if carbon dioxide is added the equilibrium will shift toward the productsC(s) + CO2(g) ↔ 2CO(g)arrow_forward2S2O2/3- (aq) + I2 (aq) ---> S4O2/6- (aq) +2I- (aq) Experiment I2 (M) S2O3- (M) Initital Rate (M/s) 1 0.01 0.01 0.0004 2 0.01 0.02 0.0004 3 0.02 0.01 0.0008 Calculate the overall order for this reaction using the table data a) 3b) 0c) 2d) 1arrow_forward
- the decomposition of N2O5 is the first order with a half-life of 1.98 minutes. If the inital concentration of N2O5 is 0.200 M, what is the concentration after 6 minutes?a) 0.612 Mb) 0.035 Mc) 0.024 Md) 0.100 Marrow_forward20.00 mL of 0.150 M HCI is titrated with 0.075 M NaOH. What volume of NaOH is needed?a) 50 mLb) 20 mLc) 40 mLd) 26.66 mLarrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCI. What is the molarity of the HCI?a) 0.150 Mb) 0.079 Mc) 0.025 Md) 0.050 Marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY