
Fundamentals of Chemical Engineering Thermodynamics (MindTap Course List)
1st Edition
ISBN: 9781111580704
Author: Kevin D. Dahm, Donald P. Visco
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12.7, Problem 25P
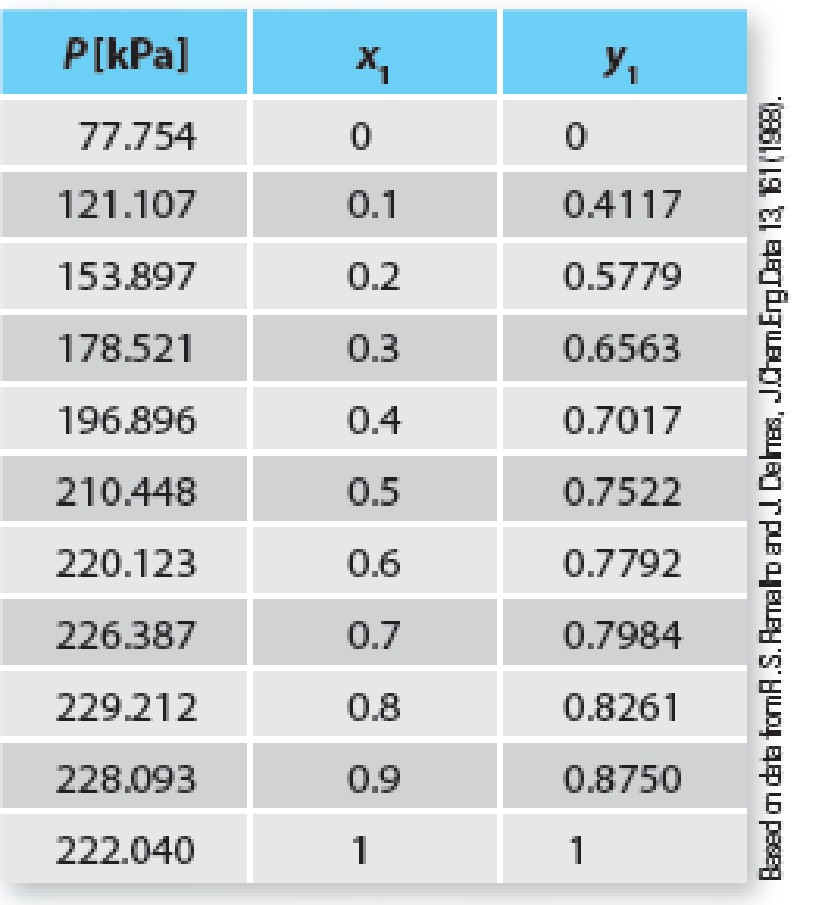
Predict the Pxy behavior for a mixture of cyclohexane (1) + 1-butanol (2) at 383.15 K using the Peng-Robinson equation of state. Compare the predictions to the experimental data given in Table P12-25. Determine a “best fit” binary interaction parameter (k12) that best matches the equation of state pressures to the experimental pressures. Plot those new predictions on the same curve. Comment on your results.
Table P12-25 Vapor-liquid equilibrium of cyclohexane (1) + 1-butanol (2) at 383.15K.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 12 Solutions
Fundamentals of Chemical Engineering Thermodynamics (MindTap Course List)
Ch. 12.6 - Prob. 1ECh. 12.6 - Derive Equation 12.31. which is the expression for...Ch. 12.6 - For an equimolar acetone (1) + methyl ethyl ketone...Ch. 12.6 - For an equimolar mixture of n-hexane (1) + benzene...Ch. 12.6 - For an equimolar mixture of water (1) + chloroform...Ch. 12.6 - For a binary mixture you desire to produce a Txy...Ch. 12.6 - Prob. 8ECh. 12.6 - The binary interaction parameters for the...Ch. 12.6 - You need to determine the binary interaction...Ch. 12.7 - An equimolar mixture of methane and propane is...
Ch. 12.7 - Consider the propane (1) + n-butane (2) system at...Ch. 12.7 - Consider the 1,1,1- trifluoroethane [R-143a] (1) +...Ch. 12.7 - Predict the Pxy behavior for a mixture of propane...Ch. 12.7 - Predict the Pxy behavior for a mixture of...Ch. 12.7 - Consider the pentafluorethane [R-125] (1) +...Ch. 12.7 - You work in a developing nation for a large...Ch. 12.7 - Prob. 20PCh. 12.7 - Predict the Txy behavior for a mixture of ethanol...Ch. 12.7 - Predict the Txy behavior for a mixture of acetone...Ch. 12.7 - Predict the Pxy behavior for a mixture of diethyl...Ch. 12.7 - Predict the Pxy behavior for a mixture of...Ch. 12.7 - Predict the Pxy behavior for a mixture of acetone...Ch. 12.7 - You are interested in the location of the...Ch. 12.7 - Prob. 28PCh. 12.7 - In Problem 12-18 in this section, you used a...Ch. 12.7 - Use - approach to model the vapor-liquid...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The
Homogeneous and Heterogeneous Equilibrium - Chemical Equilibrium - Chemistry Class 11; Author: Ekeeda;https://www.youtube.com/watch?v=8V9ozZSKl9E;License: Standard YouTube License, CC-BY