
Fundamentals of Chemical Engineering Thermodynamics (MindTap Course List)
1st Edition
ISBN: 9781111580704
Author: Kevin D. Dahm, Donald P. Visco
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12.7, Problem 24P
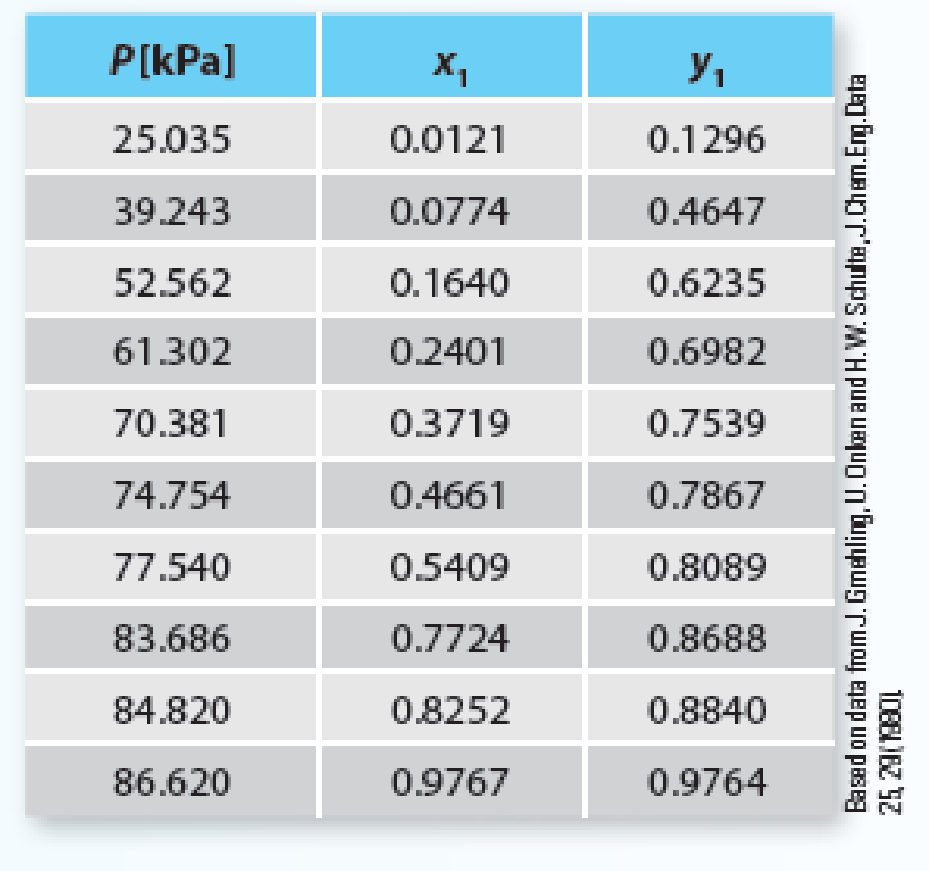
Predict the Pxy behavior for a mixture of diethyl ether (1) + methanol (2) at 303.15 K using the Peng-Robinson equation of state. Compare the predictions to the experimental data given in Table P12-24. Using the OBJ_P objective function, calculate an optimal binary interaction parameter. Is the Peng-Robinson equation of state a reasonable model for this system at this state? Please explain.
Table P12-24 Vapor-liquid equilibrium of diethyl ether (1) + methanol (2) at 303.15K.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 12 Solutions
Fundamentals of Chemical Engineering Thermodynamics (MindTap Course List)
Ch. 12.6 - Prob. 1ECh. 12.6 - Derive Equation 12.31. which is the expression for...Ch. 12.6 - For an equimolar acetone (1) + methyl ethyl ketone...Ch. 12.6 - For an equimolar mixture of n-hexane (1) + benzene...Ch. 12.6 - For an equimolar mixture of water (1) + chloroform...Ch. 12.6 - For a binary mixture you desire to produce a Txy...Ch. 12.6 - Prob. 8ECh. 12.6 - The binary interaction parameters for the...Ch. 12.6 - You need to determine the binary interaction...Ch. 12.7 - An equimolar mixture of methane and propane is...
Ch. 12.7 - Consider the propane (1) + n-butane (2) system at...Ch. 12.7 - Consider the 1,1,1- trifluoroethane [R-143a] (1) +...Ch. 12.7 - Predict the Pxy behavior for a mixture of propane...Ch. 12.7 - Predict the Pxy behavior for a mixture of...Ch. 12.7 - Consider the pentafluorethane [R-125] (1) +...Ch. 12.7 - You work in a developing nation for a large...Ch. 12.7 - Prob. 20PCh. 12.7 - Predict the Txy behavior for a mixture of ethanol...Ch. 12.7 - Predict the Txy behavior for a mixture of acetone...Ch. 12.7 - Predict the Pxy behavior for a mixture of diethyl...Ch. 12.7 - Predict the Pxy behavior for a mixture of...Ch. 12.7 - Predict the Pxy behavior for a mixture of acetone...Ch. 12.7 - You are interested in the location of the...Ch. 12.7 - Prob. 28PCh. 12.7 - In Problem 12-18 in this section, you used a...Ch. 12.7 - Use - approach to model the vapor-liquid...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The
Homogeneous and Heterogeneous Equilibrium - Chemical Equilibrium - Chemistry Class 11; Author: Ekeeda;https://www.youtube.com/watch?v=8V9ozZSKl9E;License: Standard YouTube License, CC-BY