
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
10th Edition
ISBN: 9781305957510
Author: ZUMDAHL, Steven S.; Zumdahl, Susan A.; DeCoste, Donald J.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12, Problem 67E
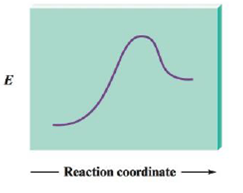
For the following reaction profile, indicate
a. the positions of reactants and products.
b. the activation energy.
c. ∆E for the reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Please help me calculate the undiluted samples ppm concentration.
My calculations were 280.11 ppm. Please see if I did my math correctly using the following standard curve.
Link: https://mnscu-my.sharepoint.com/:x:/g/personal/vi2163ss_go_minnstate_edu/EVSJL_W0qrxMkUjK2J3xMUEBHDu0UM1vPKQ-bc9HTcYXDQ?e=hVuPC4
Provide an IUPAC name for each of the compounds shown.
(Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to
commas, dashes, etc.)
H₁₂C
C(CH3)3
C=C
H3C
CH3
CH3CH2CH
CI
CH3
Submit Answer
Retry Entire Group
2 more group attempts remaining
Previous
Next
Arrange the following compounds / ions in increasing nucleophilicity (least to
most nucleophilic)
CH3NH2
CH3C=C:
CH3COO
1
2
3
5
Multiple Choice 1 point
1, 2, 3
2, 1, 3
3, 1, 2
2, 3, 1
The other answers are not correct
0000
Chapter 12 Solutions
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
Ch. 12 - Define reaction rate. Distinguish between the...Ch. 12 - Distinguish between the differential rate law and...Ch. 12 - One experimental procedure that can be used to...Ch. 12 - The initial rate for a reaction is equal to the...Ch. 12 - Consider the zero-, first-, and second-order...Ch. 12 - Derive expressions for the half-life of zero-,...Ch. 12 - Prob. 7RQCh. 12 - What two requirements must be met to call a...Ch. 12 - Prob. 9RQCh. 12 - Give the Arrhenius equation. Take the natural log...
Ch. 12 - Why does a catalyst increase the rate of a...Ch. 12 - Define stability from both a kinetic and...Ch. 12 - Describe at least two experiments you could...Ch. 12 - Make a graph of [A] versus time for zero-, first-,...Ch. 12 - How does temperature affect k, the rate constant?...Ch. 12 - Consider the following statements: In general, the...Ch. 12 - For the reaction A+BC, explain at least two ways...Ch. 12 - A friend of yours states, A balanced equation...Ch. 12 - Provide a conceptual rationale for the differences...Ch. 12 - The rate constant (k) depends on which of the...Ch. 12 - Table 11-2 illustrates how the average rate of a...Ch. 12 - The rate law for a reaction can be determined only...Ch. 12 - The plot below shows the number of collisions with...Ch. 12 - For the reaction O2(g)+2NO(g)2NO2(g) the observed...Ch. 12 - Each of the statements given below is false....Ch. 12 - Define what is meant by unimolecular and...Ch. 12 - The type of rate law for a reaction, either the...Ch. 12 - The initial rate of a reaction doubles as the...Ch. 12 - Hydrogen reacts explosively with oxygen. However,...Ch. 12 - The central idea of the collision model is that...Ch. 12 - Consider the following energy plots for a chemical...Ch. 12 - Prob. 21QCh. 12 - Prob. 22QCh. 12 - The combustion of carbohydrates and the combustion...Ch. 12 - Would the slope of a ln(k) versus 1/T plot (with...Ch. 12 - Consider the reaction 4PH3(g)P4(g)+6H2(g) If, in a...Ch. 12 - In the Haber process for the production of...Ch. 12 - At 40C, H2O2 (aq) will decompose according to the...Ch. 12 - Consider the general reaction aA+bBcC and the...Ch. 12 - What are the units for each of the following if...Ch. 12 - The rate law for the reaction...Ch. 12 - The reaction 2NO(g)+Cl2(g)2NOCl(g) was studied at...Ch. 12 - The reaction 2I-(aq)+S2O82-(aq)I2(aq)+2SO42-(aq)...Ch. 12 - The decomposition of nitrosyl chloride was...Ch. 12 - The following data were obtained for the gas-phase...Ch. 12 - The reaction I(aq)+OCl(aq)IO(aq)+Cl(aq) was...Ch. 12 - The reaction 2NO(g)+O2(g)2NO2(g) was studied. and...Ch. 12 - The rote of the reaction between hemoglobin (Hb)...Ch. 12 - The following data were obtained for the reaction...Ch. 12 - The decomposition of hydrogen peroxide was...Ch. 12 - A certain reaction has the following general form:...Ch. 12 - The rate of the reaction NO2(g)+CO(g)NO(g)+CO2(g)...Ch. 12 - A certain reaction has the following general form:...Ch. 12 - The decomposition of ethanol (C2H5OH) on an...Ch. 12 - At 500 K in the presence of a copper surface,...Ch. 12 - The dimerization of butadiene 2C4H6(g)C8H12(g) was...Ch. 12 - The rate of the reaction O(g)+NO2(g)NO(g)+O2(g)...Ch. 12 - Experimental data for the reaction A2B+C have been...Ch. 12 - Prob. 48ECh. 12 - The reaction AB+C is known to be zero order in A...Ch. 12 - The decomposition of hydrogen iodide on finely...Ch. 12 - Prob. 51ECh. 12 - A first-order reaction is 75.0% complete in 320....Ch. 12 - The rate law for the decomposition of phosphine...Ch. 12 - DDT (molar mass = 354.49 g/mol) was a widely used...Ch. 12 - The rate law for the reaction...Ch. 12 - Prob. 57ECh. 12 - Theophylline is a pharmaceutical drug that is...Ch. 12 - You and a coworker have developed a molecule...Ch. 12 - Consider the hypothetical reaction A+B+2C2D+3E...Ch. 12 - Write the rate laws for the following elementary...Ch. 12 - A possible mechanism for the decomposition of...Ch. 12 - A proposed mechanism for a reaction is...Ch. 12 - The mechanism for the gas-phase reaction of...Ch. 12 - Is the mechanism NO+Cl2l1NOCl2NOCl2+NOl22NOCl...Ch. 12 - The reaction 2NO(g) + O2(g) 2NO2(g) exhibits the...Ch. 12 - For the following reaction profile, indicate a....Ch. 12 - Draw a rough sketch of the energy profile for each...Ch. 12 - The activation energy for the reaction...Ch. 12 - The activation energy for some reaction...Ch. 12 - The rate constant for the gas-phase decomposition...Ch. 12 - The reaction (CH3)3CBr+OH(CH3)3COH+Br in a certain...Ch. 12 - The activation energy for the decomposition of...Ch. 12 - A first-order reaction has rate constants of 4.6 ...Ch. 12 - A certain reaction has an activation energy of...Ch. 12 - Prob. 76ECh. 12 - Which of the following reactions would you expect...Ch. 12 - Prob. 78ECh. 12 - One mechanism for the destruction of ozone in the...Ch. 12 - One of the concerns about the use of Freons is...Ch. 12 - Assuming that the mechanism for the hydrogenation...Ch. 12 - The decomposition of NH3 to N2 and H2 was studied...Ch. 12 - The decomposition of many substances on the...Ch. 12 - Prob. 84ECh. 12 - A popular chemical demonstration is the magic...Ch. 12 - Prob. 86ECh. 12 - Consider the following representation of the...Ch. 12 - The reaction H2SeO3(aq) + 6I-(aq) + 4H+(aq) Se(s)...Ch. 12 - Consider two reaction vessels, one containing A...Ch. 12 - Sulfuryl chloride (SO2Cl2) decomposes to sulfur...Ch. 12 - For the reaction 2N2O5(g)4NO2(g)+O2(g) the...Ch. 12 - Experimental values for the temperature dependence...Ch. 12 - At 620. K butadiene dimerizes at a moderate rate....Ch. 12 - For a first order gas phase reaction A products,...Ch. 12 - Cobra venom helps the snake secure food by binding...Ch. 12 - Iodomethane (CH3I) is a commonly used reagent in...Ch. 12 - Experiments during a recent summer on a number of...Ch. 12 - The activation energy of a certain uncatalyzed...Ch. 12 - Consider the following initial rate data for the...Ch. 12 - Consider a hypothetical reaction between A and B:...Ch. 12 - Consider the reaction 3A+B+CD+E where the rate law...Ch. 12 - The thiosulfate ion (S2O32) is oxidized by iodine...Ch. 12 - The reaction A(aq)+B(aq)products(aq) was studied,...Ch. 12 - A certain substance, initially present at 0.0800...Ch. 12 - A reaction of the form aAProducts gives a plot of...Ch. 12 - A certain reaction has the form aAProducts At a...Ch. 12 - Which of the following statement(s) is( are) true?...Ch. 12 - Consider the hypothetical reaction A2(g) + B2(g) ...Ch. 12 - Experiments have shown that the average frequency...Ch. 12 - Consider a reaction of the type aA products, in...Ch. 12 - A study was made of the effect of the hydroxide...Ch. 12 - Two isomers (A and B) of a given compound dimerize...Ch. 12 - The reaction NO(g)+O3NO2(g)+O2(g) was studied by...Ch. 12 - Prob. 114CPCh. 12 - In the gas phase, the production of phosgene from...Ch. 12 - Most reactions occur by a series of steps. The...Ch. 12 - You are studying the kinetics of the reaction...Ch. 12 - The decomposition of NO2(g) occurs by the...Ch. 12 - The following data were collected in two studies...Ch. 12 - Consider the following hypothetical data collected...Ch. 12 - Consider the hypothetical reaction A+B+2C2D+3E In...Ch. 12 - Hydrogen peroxide and the iodide ion react in...Ch. 12 - Sulfuryl chloride undergoes first-order...Ch. 12 - Upon dissolving InCl(s) in HCl, In+(aq) undergoes...Ch. 12 - The decomposition of iodoethane in the gas phase...Ch. 12 - Consider the following reaction: CH3X+YCH3Y+X At...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Describe the evolution of mammals, tracing their synapsid lineage from early amniote ancestors to true mammals....
Loose Leaf For Integrated Principles Of Zoology
60. The solar system is 25,000 light years from the center of our Milky Way galaxy. One light year is the dista...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
What are the cervical and lumbar enlargements?
Principles of Anatomy and Physiology
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- curved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the cured electron-pushing arrows for thw following reaction or mechanistic steps. be sure to account for all bond-breaking and bond making stepsarrow_forwardUsing the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.arrow_forwardSynthesis of Dibenzalacetone [References] Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below. Question 1 1 pt Question 2 1 pt Question 3 1 pt H Question 4 1 pt Question 5 1 pt Question 6 1 pt Question 7 1pt Question 8 1 pt Progress: 7/8 items Que Feb 24 at You do not have to consider stereochemistry. . Draw the enolate ion in its carbanion form. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. ⚫ Separate multiple reactants using the + sign from the drop-down menu. ? 4arrow_forward
- Shown below is the mechanism presented for the formation of biasplatin in reference 1 from the Background and Experiment document. The amounts used of each reactant are shown. Either draw or describe a better alternative to this mechanism. (Note that the first step represents two steps combined and the proton loss is not even shown; fixing these is not the desired improvement.) (Hints: The first step is correct, the second step is not; and the amount of the anhydride is in large excess to serve a purpose.)arrow_forwardHi I need help on the question provided in the image.arrow_forwardDraw a reasonable mechanism for the following reaction:arrow_forward
- Draw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M HCl is titrated with 37.75 mL of NaOH. What is the molarity of the NaOH?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY