
ESSENTIAL UNIV.PHYS.-MOD.MASTERING(18W)
4th Edition
ISBN: 9780136780984
Author: Wolfson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 33P
You work for a garden equipment company, and you’re designing a new garden cart. Specifications to be listed include the horizontal force that must be applied to push the fully loaded cart (mass 55 kg. 60-cm-diameter wheels) up an abrupt 8.0-cm step, as shown in Fig. 12.19. What do you specify for the force?
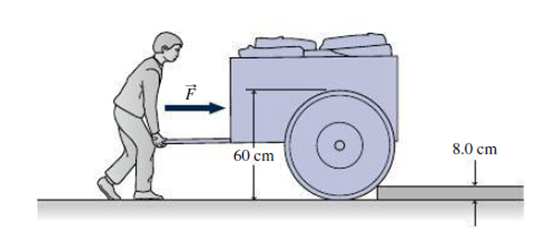
FIGURE 12.19 Problem 27
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A cylinder with a piston contains 0.153 mol of
nitrogen at a pressure of 1.83×105 Pa and a
temperature of 290 K. The nitrogen may be
treated as an ideal gas. The gas is first compressed
isobarically to half its original volume. It then
expands adiabatically back to its original volume,
and finally it is heated isochorically to its original
pressure.
Part A
Compute the temperature at the beginning of the adiabatic expansion.
Express your answer in kelvins.
ΕΠΙ ΑΣΦ
T₁ =
?
K
Submit
Request Answer
Part B
Compute the temperature at the end of the adiabatic expansion.
Express your answer in kelvins.
Π ΑΣΦ
T₂ =
Submit
Request Answer
Part C
Compute the minimum pressure.
Express your answer in pascals.
ΕΠΙ ΑΣΦ
P =
Submit
Request Answer
?
?
K
Pa
Learning Goal:
To understand the meaning and the basic applications of
pV diagrams for an ideal gas.
As you know, the parameters of an ideal gas are
described by the equation
pV = nRT,
where p is the pressure of the gas, V is the volume of
the gas, n is the number of moles, R is the universal gas
constant, and T is the absolute temperature of the gas. It
follows that, for a portion of an ideal gas,
pV
= constant.
Τ
One can see that, if the amount of gas remains constant,
it is impossible to change just one parameter of the gas:
At least one more parameter would also change. For
instance, if the pressure of the gas is changed, we can
be sure that either the volume or the temperature of the
gas (or, maybe, both!) would also change.
To explore these changes, it is often convenient to draw a
graph showing one parameter as a function of the other.
Although there are many choices of axes, the most
common one is a plot of pressure as a function of
volume: a pV diagram.
In this problem, you…
Learning Goal:
To understand the meaning and the basic applications of
pV diagrams for an ideal gas.
As you know, the parameters of an ideal gas are
described by the equation
pV = nRT,
where p is the pressure of the gas, V is the volume of
the gas, n is the number of moles, R is the universal gas
constant, and T is the absolute temperature of the gas. It
follows that, for a portion of an ideal gas,
pV
= constant.
T
One can see that, if the amount of gas remains constant,
it is impossible to change just one parameter of the gas:
At least one more parameter would also change. For
instance, if the pressure of the gas is changed, we can
be sure that either the volume or the temperature of the
gas (or, maybe, both!) would also change.
To explore these changes, it is often convenient to draw a
graph showing one parameter as a function of the other.
Although there are many choices of axes, the most
common one is a plot of pressure as a function of
volume: a pV diagram.
In this problem, you…
Chapter 12 Solutions
ESSENTIAL UNIV.PHYS.-MOD.MASTERING(18W)
Ch. 12.1 - The figure shows three pairs of forces acting on...Ch. 12.2 - Prob. 12.2GICh. 12.3 - The figure shows a person in static equilibrium...Ch. 12.4 - Prob. 12.4GICh. 12 - Give an example of an object on which the net...Ch. 12 - Prob. 2FTDCh. 12 - Pregnant women often assume a posture with their...Ch. 12 - When you carry a bucket of water with one hand,...Ch. 12 - Is a ladder more likely to slip when you stand...Ch. 12 - Does choosing a pivot point in an equilibrium...
Ch. 12 - If you take the pivot point at the application...Ch. 12 - A short dog and a tall person are standing on a...Ch. 12 - Prob. 9FTDCh. 12 - A body is subject to three forces; F1=1i+2jN,...Ch. 12 - To demonstrate that the choice of pivot point...Ch. 12 - In Fig. 12.11 the forces shown all have the same...Ch. 12 - Figure 12.12a shows a thin, uniform square plate...Ch. 12 - Repeat the preceding problem for the equilateral...Ch. 12 - A 23-m-long log of irregular cross section lies...Ch. 12 - A 60-kg uniform board 2.4 m long is supported by a...Ch. 12 - Where should the child in Fig. 12.14 sit if the...Ch. 12 - A 4.2-m-long beam is supported by a cable at its...Ch. 12 - Figure 12.15 shows how a scale with a capacity of...Ch. 12 - A portion of a roller-coaster track is described...Ch. 12 - Prob. 21ECh. 12 - Prob. 22ECh. 12 - Prob. 23ECh. 12 - Example 12.2: Climbers attempting to cross a...Ch. 12 - Example 12.4: Consider the potential-energy curve...Ch. 12 - Prob. 27ECh. 12 - Prob. 28ECh. 12 - Youre a highway safety engineer, and youre asked...Ch. 12 - Figure 12.17a shows an outstretched arm with mass...Ch. 12 - A uniform sphere of radius R is supported by a...Ch. 12 - You work for a garden equipment company, and youre...Ch. 12 - Figure 12.20 shows the fool and lower leg of a...Ch. 12 - A uniform 5.0-kg ladder is leaning against a...Ch. 12 - The boom in the crane of Fig. 12.21 is free to...Ch. 12 - A uniform board of length L and weight W is...Ch. 12 - Figure 12.23 shows a 1250-kg car that has slipped...Ch. 12 - You are headwaiter at a new restaurant, and your...Ch. 12 - You are headwaiter at a new restaurant, and your...Ch. 12 - Prob. 41PCh. 12 - A crane in a marble quarry is mounted on the...Ch. 12 - A rectangular block measures w w L, where L is...Ch. 12 - The potential energy as a function of position for...Ch. 12 - A rectangular block of mass m measures w w L,...Ch. 12 - A 160-kg highway sign of uniform density is 2.3 m...Ch. 12 - Prob. 47PCh. 12 - Prob. 48PCh. 12 - A uniform, solid cube of mass m and side s is in...Ch. 12 - An isosceles triangular block of mass m and height...Ch. 12 - Youre investigating ladder safety for the Consumer...Ch. 12 - A 2.0-m-long rod has density in kilograms per...Ch. 12 - What horizontal force applied at its highest point...Ch. 12 - A rectangular block twice as high as it is wide is...Ch. 12 - What condition on the coefficient of friction in...Ch. 12 - A uniform solid cone of height h and base diameter...Ch. 12 - Prove the statement in Section 12.1 that the...Ch. 12 - Three identical books of length L are stacked over...Ch. 12 - A uniform pole of mass M is at rest on an incline...Ch. 12 - For what angle does the situation in Problem 53...Ch. 12 - Figure 12.31 shows a popular system for mounting...Ch. 12 - The nuchal ligament is a thick, cordlike structure...Ch. 12 - A 4.2-kg plant hangs from the bracket shown in...Ch. 12 - The wheel in Fig. 12.34 has mass M and is weighted...Ch. 12 - An interstellar spacecraft from an advanced...Ch. 12 - Prob. 66PCh. 12 - Engineers designing a new semiconductor device...Ch. 12 - Youve been hired by your states environmental...Ch. 12 - Youve been hired by your states environmental...Ch. 12 - Youve been hired by your states environmental...Ch. 12 - Youve been hired by your states environmental...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For the following fragment of DNA, determine the number of hydrogen bonds and the number of phosphodiester bond...
Genetic Analysis: An Integrated Approach (3rd Edition)
DRAW IT In human spermatogenesis, mitosis of a stem cell gives rise to one cell that remains a stem cell and on...
Campbell Biology (11th Edition)
Fibrous connective tissue consists of ground substance and fibers that provide strength, support, and flexibili...
Human Biology: Concepts and Current Issues (8th Edition)
You microscopically examine scrapings from a case of Acan-thamoeba keratitis. You expect to see a. nothing. b. ...
Microbiology: An Introduction
Which coastal area experiences the largest tidal range difference in height between the high tide and low tide?...
Applications and Investigations in Earth Science (9th Edition)
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- ■ Review | Constants A cylinder with a movable piston contains 3.75 mol of N2 gas (assumed to behave like an ideal gas). Part A The N2 is heated at constant volume until 1553 J of heat have been added. Calculate the change in temperature. ΜΕ ΑΣΦ AT = Submit Request Answer Part B ? K Suppose the same amount of heat is added to the N2, but this time the gas is allowed to expand while remaining at constant pressure. Calculate the temperature change. AT = Π ΑΣΦ Submit Request Answer Provide Feedback ? K Nextarrow_forward4. I've assembled the following assortment of point charges (-4 μC, +6 μC, and +3 μC) into a rectangle, bringing them together from an initial situation where they were all an infinite distance away from each other. Find the electric potential at point "A" (marked by the X) and tell me how much work it would require to bring a +10.0 μC charge to point A if it started an infinite distance away (assume that the other three charges remains fixed). 300 mm -4 UC "A" 0.400 mm +6 UC +3 UC 5. It's Friday night, and you've got big party plans. What will you do? Why, make a capacitor, of course! You use aluminum foil as the plates, and since a standard roll of aluminum foil is 30.5 cm wide you make the plates of your capacitor each 30.5 cm by 30.5 cm. You separate the plates with regular paper, which has a thickness of 0.125 mm and a dielectric constant of 3.7. What is the capacitance of your capacitor? If you connect it to a 12 V battery, how much charge is stored on either plate? =arrow_forwardLearning Goal: To understand the meaning and the basic applications of pV diagrams for an ideal gas. As you know, the parameters of an ideal gas are described by the equation pV = nRT, where p is the pressure of the gas, V is the volume of the gas, n is the number of moles, R is the universal gas constant, and T is the absolute temperature of the gas. It follows that, for a portion of an ideal gas, PV T = constant. One can see that, if the amount of gas remains constant, it is impossible to change just one parameter of the gas: At least one more parameter would also change. For instance, if the pressure of the gas is changed, we can be sure that either the volume or the temperature of the gas (or, maybe, both!) would also change. To explore these changes, it is often convenient to draw a graph showing one parameter as a function of the other. Although there are many choices of axes, the most common one is a plot of pressure as a function of volume: a pV diagram. In this problem, you…arrow_forward
- A-e pleasearrow_forwardTwo moles of carbon monoxide (CO) start at a pressure of 1.4 atm and a volume of 35 liters. The gas is then compressed adiabatically to 1/3 this volume. Assume that the gas may be treated as ideal. Part A What is the change in the internal energy of the gas? Express your answer using two significant figures. ΕΠΙ ΑΣΦ AU = Submit Request Answer Part B Does the internal energy increase or decrease? internal energy increases internal energy decreases Submit Request Answer Part C ? J Does the temperature of the gas increase or decrease during this process? temperature of the gas increases temperature of the gas decreases Submit Request Answerarrow_forwardYour answer is partially correct. Two small objects, A and B, are fixed in place and separated by 2.98 cm in a vacuum. Object A has a charge of +0.776 μC, and object B has a charge of -0.776 μC. How many electrons must be removed from A and put onto B to make the electrostatic force that acts on each object an attractive force whose magnitude is 12.4 N? e (mea is the es a co le E o ussian Number Tevtheel ed Media ! Units No units → answe Tr2Earrow_forward
- 4 Problem 4) A particle is being pushed up a smooth slot by a rod. At the instant when 0 = rad, the angular speed of the arm is ė = 1 rad/sec, and the angular acceleration is = 2 rad/sec². What is the net force acting on the 1 kg particle at this instant? Express your answer as a vector in cylindrical coordinates. Hint: You can express the radial coordinate as a function of the angle by observing a right triangle. (20 pts) Ꮎ 2 m Figure 3: Particle pushed by rod along vertical path.arrow_forward4 Problem 4) A particle is being pushed up a smooth slot by a rod. At the instant when 0 = rad, the angular speed of the arm is ė = 1 rad/sec, and the angular acceleration is = 2 rad/sec². What is the net force acting on the 1 kg particle at this instant? Express your answer as a vector in cylindrical coordinates. Hint: You can express the radial coordinate as a function of the angle by observing a right triangle. (20 pts) Ꮎ 2 m Figure 3: Particle pushed by rod along vertical path.arrow_forwardplease solve and answer the question correctly. Thank you!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Glencoe Physics: Principles and Problems, Student...
Physics
ISBN:9780078807213
Author:Paul W. Zitzewitz
Publisher:Glencoe/McGraw-Hill

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

University Physics Volume 1
Physics
ISBN:9781938168277
Author:William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:OpenStax - Rice University

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Static Equilibrium: concept; Author: Jennifer Cash;https://www.youtube.com/watch?v=0BIgFKVnlBU;License: Standard YouTube License, CC-BY