
(a)
The shape of the molecule
N 2 O
(a)
Answer to Problem 32E

The molecule
Explanation of Solution
Given Info: Refer to Table 12.6 in the textbook to predict the shape of the molecule.
Explanation:
The shape of the molecule

The central atom has two surrounding regions.
The polarity of a molecule depends on the electronegativity of atoms in that molecule and the shape of the molecule. A molecule which has one polar bond is polar.
A molecule which has all nonpolar bonds is nonpolar.
The symmetric molecule will be nonpolar because the polarity of bonds is canceled out. Thus an asymmetric molecule is polar because the polarity of bonds is not canceled.
Therefore, the asymmetric molecule
Conclusion:
Therefore, the shape of the molecule

The molecule
(b)
The shape of the molecule
SO 4 2 −
(b)
Answer to Problem 32E
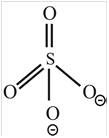
The molecule
Explanation of Solution
Given Info: Refer to Table 12.6 in the textbook to predict the shape of the molecule.
Explanation:
The shape of the molecule

The central atom has four surrounding regions.
The polarity of a molecule depends on the electronegativity of atoms in that molecule and the shape of the molecule. A molecule which has one polar bond is polar.
A molecule which has all nonpolar bonds is nonpolar.
The symmetric molecule will be nonpolar because the polarity of bonds is canceled out. Thus an asymmetric molecule is polar because the polarity of bonds is not canceled.
Therefore, the symmetric molecule
Conclusion:
Therefore, the shape of the molecule

The molecule
(c)
The shape of the molecule
GaH 3
(c)
Answer to Problem 32E
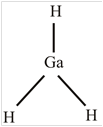
The molecule
Explanation of Solution
Given Info: Refer to Table 12.6 in the textbook to predict the shape of the molecule.
Explanation:
The shape of the molecule

The central atom has three surrounding regions.
The polarity of a molecule depends on the electronegativity of atoms in that molecule and the shape of the molecule. A molecule which has one polar bond is polar.
A molecule which has all nonpolar bonds is nonpolar.
The symmetric molecule will be nonpolar because the polarity of bonds is canceled out. Thus an asymmetric molecule is polar because the polarity of bonds is not canceled.
Therefore, the symmetric molecule
Conclusion:
The shape of the molecule

The molecule
(d)
The shape of the molecule
NH 2 Cl
(d)
Answer to Problem 32E
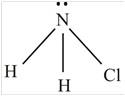
The molecule
Explanation of Solution
Given Info: Refer to Table 12.6 in the textbook to predict the shape of the molecule.
Explanation:
The shape of the molecule

The central atom has four surrounding regions.
The polarity of a molecule depends on the electronegativity of atoms in that molecule and the shape of the molecule. A molecule which has one polar bond is polar.
A molecule which has all nonpolar bonds is nonpolar.
The symmetric molecule will be nonpolar because the polarity of bonds is canceled out. Thus an asymmetric molecule is polar because the polarity of bonds is not canceled.
Therefore, the asymmetric molecule
Conclusion:
Therefore, the shape of the molecule

The molecule
Want to see more full solutions like this?
Chapter 12 Solutions
An Introduction to Physical Science
- Consider the circuit shown in the figure below. (Assume L = 5.20 m and R2 = 440 Ω.) (a) When the switch is in position a, for what value of R1 will the circuit have a time constant of 15.4 µs? (b) What is the current in the inductor at the instant the switch is thrown to position b?arrow_forwardCan someone helparrow_forwardCan someone help mearrow_forward
- A particle in a box between x=0 and x=6 has the wavefunction Psi(x)=A sin(2πx). How muchenergy is required for the electron to make a transition to Psi(x)= A’ sin(7π x/3). Draw anapproximate graph for the wavefunction. Find A and A'arrow_forwardA proton is moving with 10^8 m/s speed. Find the De Broglie wavelength associated with theproton and the frequency of that wave.arrow_forwardFind the wavelength of the photon if a (Li--) electron makes a transition from n=4 to n=3. Findthe Bohr radius for each state.arrow_forward
- A photon with wavelength 3000 nm hits a stationary electron. After the collision electron isscattered to 60 degrees. Find the wavelength and frequency of the scattered photon.arrow_forwardA metal has threshold frequency 10^15. Calculate the maximum kinetic energy of the ejectedelectron if a laser beam with wavelength 1.5 10^-7 m is projected on the metal.arrow_forwardDetermine the direction of the vector V, B, or ♬ that is missing from the pair of vectors shown in each scenario. Here, u is the velocity vector of a moving positive charge, B is a constant and uniform magnetic field, and F is the resulting force on the moving charge. 1. 2. 3. B OB F 4. ↑F F 5. 怔 ↑ ↑F Answer Bank 6. ↑ TE Farrow_forward
- Two point charges (+9.80 nC and -9.80 nC) are located 8.00 cm apart. Let U=0 when all of the charges are separated by infinite distances. What is the potential energy if a third point charge q=-4.20 nC is placed at point b? 8.00 cm 8.00 cm 4.00 +4.00 +4.00- cm cm cm HJarrow_forward! Required information Two chloride ions and two sodium ions are in water, the "effective charge" on the chloride ions (CI¯) is −2.00 × 10-21 C and that of the sodium ions (Na+) is +2.00 x 10-21 C. (The effective charge is a way to account for the partial shielding due to nearby water molecules.) Assume that all four ions are coplanar. CT Na+ Na+ 30.0° 45.0% с сг L. where a = 0.300 nm, b = 0.710 nm, and c = 0.620 nm. What is the direction of electric force on the chloride ion in the lower right-hand corner in the diagram? Enter the angle in degrees where positive indicates above the negative x-axis and negative indicates below the positive x-axis.arrow_forwardA pendulum has a 0.4-m-long cord and is given a tangential velocity of 0.2 m/s toward the vertical from a position 0 = 0.3 rad. Part A Determine the equation which describes the angular motion. Express your answer in terms of the variable t. Express coefficients in radians to three significant figures. ΜΕ ΑΣΦ vec (t)=0.3 cos (4.95t) + 0.101 sin (4.95t) Submit Previous Answers Request Answer × Incorrect; Try Again; 6 attempts remainingarrow_forward
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax



