
Interpretation:
For each transformation, the reducing agent used, out of
Concept introduction:
Lithium aluminium hydride and sodium borohydride are reducing agents.
Answer to Problem 1PP
Solution:
(a)
(b)
(c)
(d)
(e)
(a)
Explanation of Solution
The given transformation is as follows:
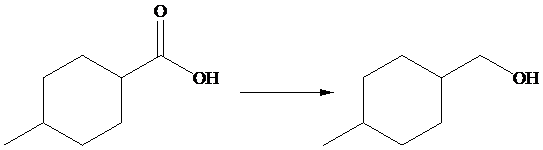
In the above transformation,
Hence,
(b)
The given transformation is as follows;
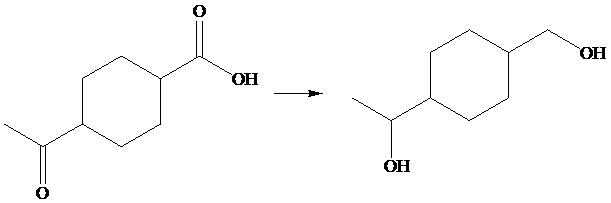
In the above transformation, carboxylic acid and ketone are converted into their corresponding alcohols. This transformation is done by using
Hence,
(c)
The given transformation is as follows:
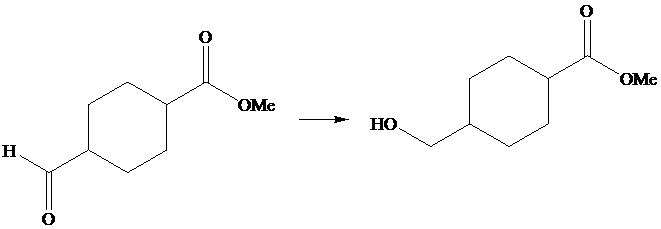
In the above transformation, aldehyde is converted into its corresponding alcohol, but there is no effect on the ester group. This transformation is done by using
Hence,
(d)
The given transformation is as follows:
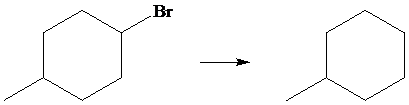
In the above transformation, the bromine atom is replaced by the hydrogen atom. This transformation is done by using
Hence,
(e)
The given transformation is as follows:
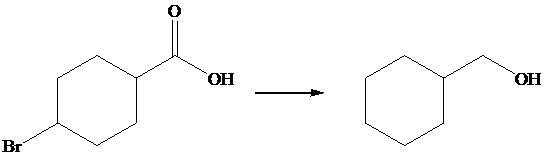
In the above transformation, the bromine atom is replaced by the hydrogen atom, and carboxylic acid is converted into its corresponding alcohol. This transformation is done by using
Hence,
Want to see more full solutions like this?
Chapter 12 Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
- K Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward7 Comment on the general features of the predicted (extremely simplified) ¹H- NMR spectrum of lycopene that is provided below. 00 6 57 PPM 3 2 1 0arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
