
(a)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
The given transformation can be carried out by oxymercuration-reduction. The detailed mechanism is as follows:
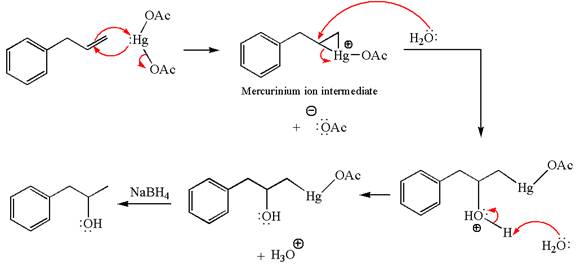
Explanation of Solution
The given equation is
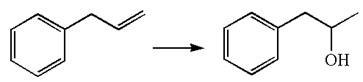
In the substrate, the alkene
The alkene substrate, on reaction with mercury
In the first step, the electron rich
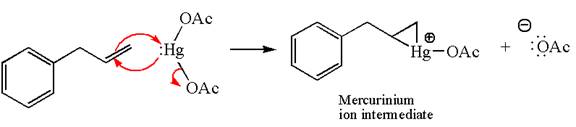
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
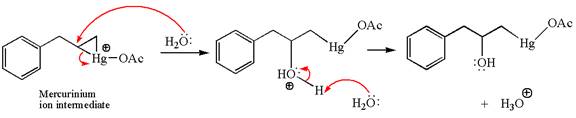
The product formed in the previous step is then subjected to reduction with sodium borohydride,
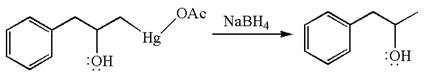
The preparation of the given compound is explained indicating the addition of water across the
(b)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
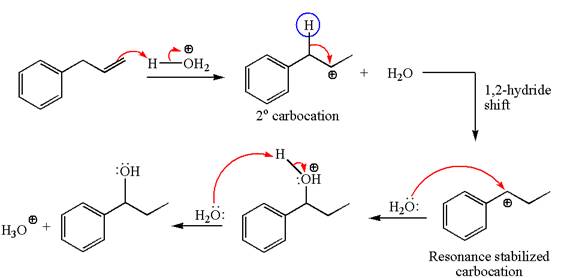
The given transformation can be carried out by acid-catalyzed hydration. The detailed mechanism is as follows:
Explanation of Solution
The given equation is
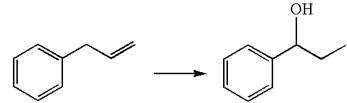
In the substrate, the alkene
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
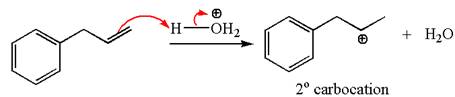
The secondary carbocation can be rearranged to more stable tertiary as well as resonance stabilized carbocation by
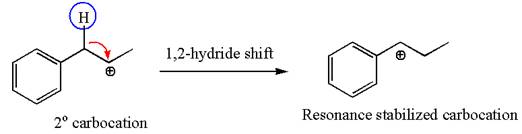
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
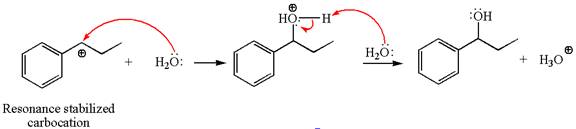
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through carbocation rearrangement.
(c)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
The given transformation can be carried out by acid catalyzed hydration. The detailed mechanism is as follows:
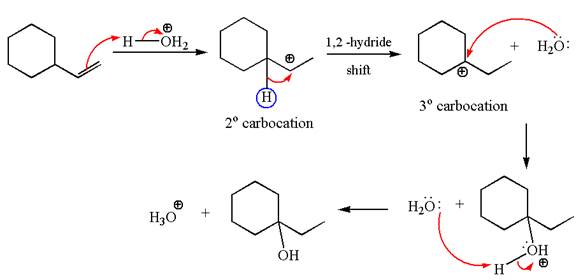
Explanation of Solution
The given equation is
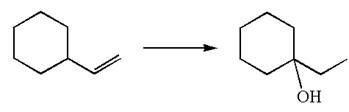
In the substrate, the alkene
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
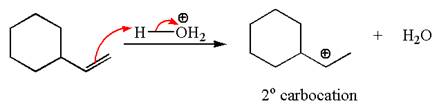
The secondary carbocation can be rearranged to more stable tertiary by
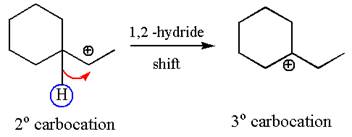
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
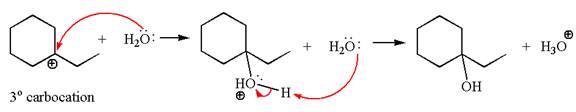
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through carbocation rearrangement.
(d)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water across the
Answer to Problem 12.44P
The given transformation can be carried out by oxymercuration-reduction. The detailed mechanism is as follows:
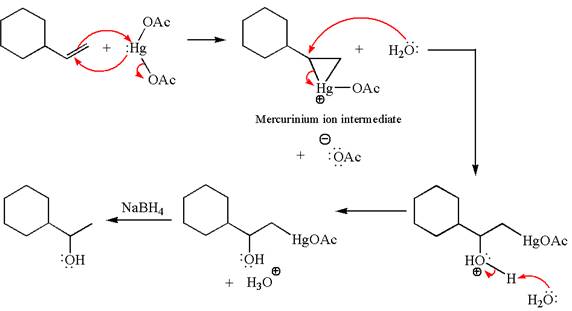
Explanation of Solution
The given equation is
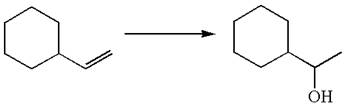
In the substrate, the alkene
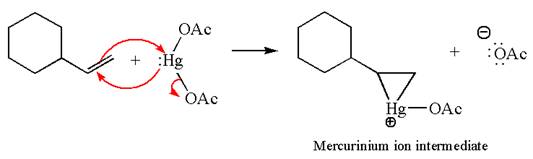
The alkene substrate, on reaction with mercury
In the first step, the electron rich
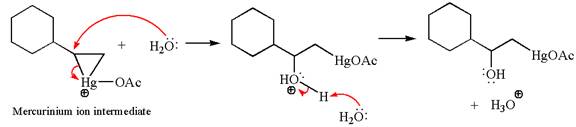
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
The product formed in the previous step is then subjected to reduction with sodium borohydride,
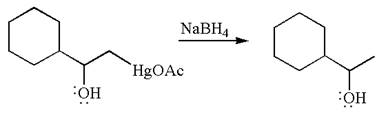
The preparation of the given compound is explained indicating the addition of water across the
Want to see more full solutions like this?
Chapter 12 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 5) Confidence interval. Berglund and Wichardt investigated the quantitative determination of Cr in high-alloy steels using a potentiometric titration of Cr(VI). Before the titration, samples of the steel were dissolved in acid and the chromium oxidized to Cr(VI) using peroxydisulfate. Shown here are the results (as %w/w Cr) for the analysis of a reference steel. 16.968, 16.922, 16.840, 16.883, 16.887, 16.977, 16.857, 16.728 Calculate the mean, the standard deviation, and the 95% confidence interval about the mean. What does this confidence interval mean?arrow_forwardIn the Nitrous Acid Test for Amines, what is the observable result for primary amines? Group of answer choices nitrogen gas bubbles form a soluble nitrite salt yellow oily layer of nitrosoaminearrow_forward3. a. Use the MS to propose at least two possible molecular formulas. For an unknown compound: 101. 27.0 29.0 41.0 50.0 52.0 55.0 57.0 100 57.5 58.0 58.5 62.0 63.0 64.0 65.0 74.0 40 75.0 76.0 20 20 40 60 80 100 120 140 160 180 200 220 m/z 99.5 68564810898409581251883040 115.0 116.0 77404799 17417M 117.0 12.9 118.0 33.5 119.0 36 133 0 1.2 157.0 2.1 159.0 16 169.0 219 170.0 17 171.0 21.6 172.0 17 181.0 1.3 183.0 197.0 100.0 198.0 200. 784 Relative Intensity 2 2 8 ō (ppm) 6 2arrow_forward
- Solve the structure and assign each of the following spectra (IR and C-NMR)arrow_forward1. For an unknown compound with a molecular formula of C8H100: a. What is the DU? (show your work) b. Solve the structure and assign each of the following spectra. 8 6 2 ō (ppm) 4 2 0 200 150 100 50 ō (ppm) LOD D 4000 3000 2000 1500 1000 500 HAVENUMBERI -11arrow_forward16. The proton NMR spectral information shown in this problem is for a compound with formula CioH,N. Expansions are shown for the region from 8.7 to 7.0 ppm. The normal carbon-13 spec- tral results, including DEPT-135 and DEPT-90 results, are tabulated: 7 J Normal Carbon DEPT-135 DEPT-90 19 ppm Positive No peak 122 Positive Positive cus и 124 Positive Positive 126 Positive Positive 128 No peak No peak 4° 129 Positive Positive 130 Positive Positive (144 No peak No peak 148 No peak No peak 150 Positive Positive してしarrow_forward
- 3. Propose a synthesis for the following transformation. Do not draw an arrow-pushing mechanism below, but make sure to draw the product of each proposed step (3 points). + En CN CNarrow_forwardShow work..don't give Ai generated solution...arrow_forwardLabel the spectrum with spectroscopyarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


