
(a)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
The given transformation can be carried out by oxymercuration-reduction. The detailed mechanism is as follows:
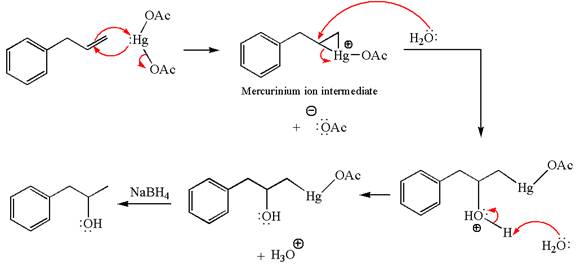
Explanation of Solution
The given equation is
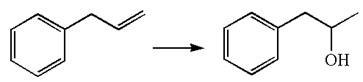
In the substrate, the alkene
The alkene substrate, on reaction with mercury
In the first step, the electron rich
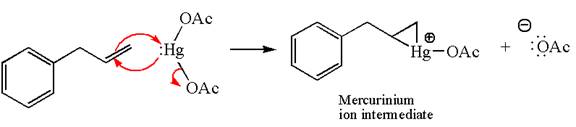
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
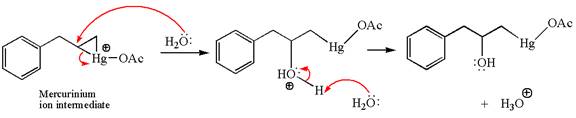
The product formed in the previous step is then subjected to reduction with sodium borohydride,
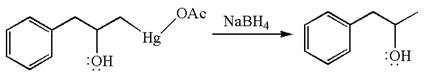
The preparation of the given compound is explained indicating the addition of water across the
(b)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
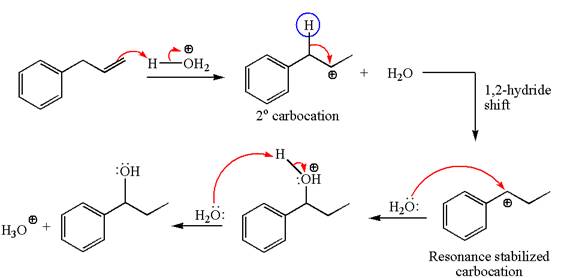
The given transformation can be carried out by acid-catalyzed hydration. The detailed mechanism is as follows:
Explanation of Solution
The given equation is
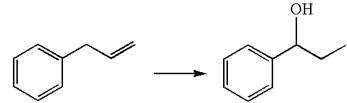
In the substrate, the alkene
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
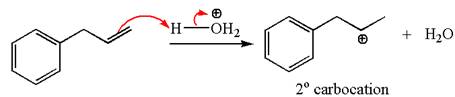
The secondary carbocation can be rearranged to more stable tertiary as well as resonance stabilized carbocation by
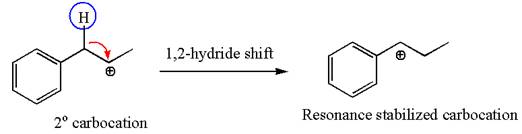
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
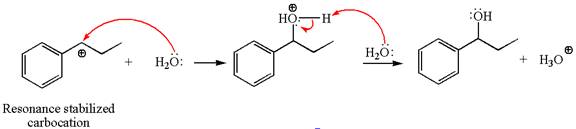
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through carbocation rearrangement.
(c)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water through the
Answer to Problem 12.44P
The given transformation can be carried out by acid catalyzed hydration. The detailed mechanism is as follows:
Explanation of Solution
The given equation is
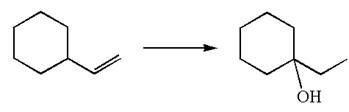
In the substrate, the alkene
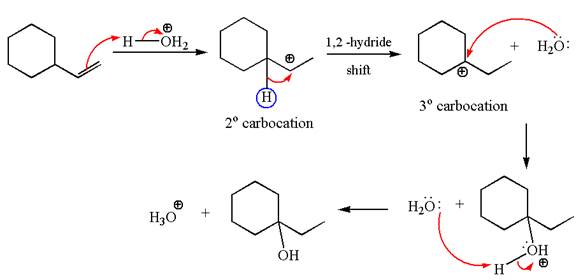
The first step is the formation of a secondary carbocation by proton transfer reaction. The proton transfers to the less substituted double bonded carbon.
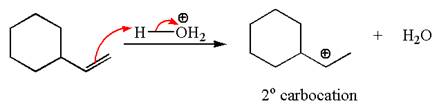
The secondary carbocation can be rearranged to more stable tertiary by
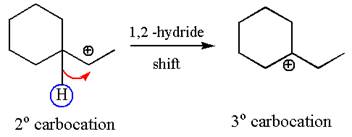
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
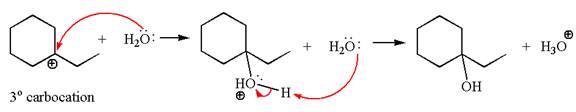
The detailed mechanism for the given reaction is drawn by suggesting that the reaction occurred through carbocation rearrangement.
(d)
Interpretation:
It is to be explained whether the given transformation would be a result of acid-catalyzed hydration or oxymercuration-reduction.
Concept introduction:
The acid-catalyzed hydration of an alkene is the electrophilic addition of water across the
The oxymercuration-reduction is also the reaction of addition of water across the
Answer to Problem 12.44P
The given transformation can be carried out by oxymercuration-reduction. The detailed mechanism is as follows:
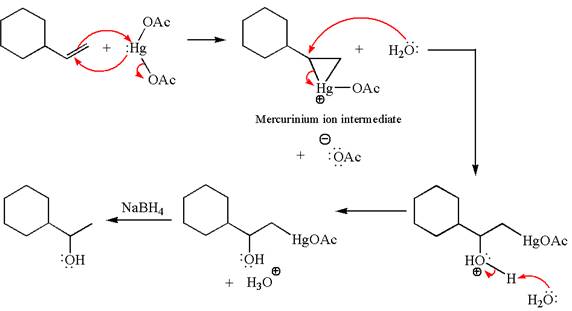
Explanation of Solution
The given equation is
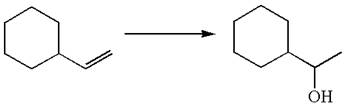
In the substrate, the alkene
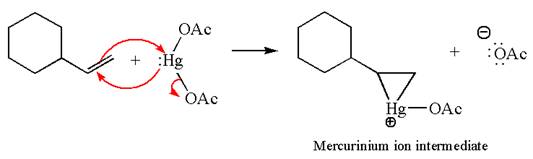
The alkene substrate, on reaction with mercury
In the first step, the electron rich
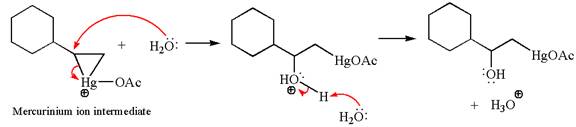
In the second step, the water molecule acts as a nucleophile on one of the carbons of the three-membered ring to open the ring, followed by deprotonation of the positively charged oxygen atom.
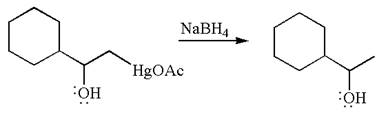
The product formed in the previous step is then subjected to reduction with sodium borohydride,
The preparation of the given compound is explained indicating the addition of water across the
Want to see more full solutions like this?
Chapter 12 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- You are a Quality Manager for a very well-known food ingredient company that produces umami powder, and you are responsible for setting specification limits. The net weight (in grams) of bags of unami powder is monitored by taking samples of six bags on an hourly basis during production. The label on every bag reports a contents of 1KG umami powder. The process mean is μ = 1012 g, and when the process is properly adjusted, it varies with σ = 11 g. QUESTION: Using all the available information, set the upper and lower specification limits.arrow_forward43) 10.00 ml of vinegar (active ingredient is acetic acid) is titrated to the endpoint using 19.32 ml of 0.250 M sodium hydroxide. What is the molarity of acetic acid in the vinegar? YOU MUST SHOW YOUR WORK. NOTE: MA x VA = MB x VBarrow_forward424 Repon Sheet Rates of Chemical Reactions : Rate and Order of 1,0, Deception B. Effect of Temperature BATH TEMPERATURE 35'c Yol of Oh نام Time 485 Buret rend ing(n) 12 194 16. 6 18 20 10 22 24 14 115 95 14738 2158235 8:26 CMS 40148 Total volume of 0, collected Barometric pressure 770-572 ml mm Hg Vapor pressure of water at bath temperature (see Appendix L) 42.2 Slope Compared with the rate found for solution 1, there is Using the ideal gas law, calculate the moles of O; collected (show calculations) times faster 10 Based on the moles of O, evolved, calculate the molar concentration of the original 3% 1,0, solution (sho calculations)arrow_forward
- Steps and explanations pleasearrow_forwardUse diagram to answer the following: 1.Is the overall rxn endo- or exothermic. Explain briefly your answer____________________2. How many steps in this mechanism?_____________3. Which is the rate determining step? Explain briefly your answer____________________4. Identify (circle and label) the reactants,the products and intermediate (Is a Cation, Anion, or a Radical?) Please explain and provide full understanding.arrow_forwardDraw the entire mechanism and add Curved Arrows to show clearly how electrons areredistributed in the process. Please explain and provide steps clearly.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning




