
EBK GENERAL CHEMISTRY: THE ESSENTIAL CO
7th Edition
ISBN: 9780100257047
Author: Chang
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 12, Problem 12.35QP
(a)
Interpretation Introduction
Interpretation:
The following types of crystals have to be discussed and examples have to be given for each type.
- (a) Ionic crystals (b) Covalent crystals (c) Molecular crystals (d) Metallic crystals.
Concept Introduction:
- A solid is anything that is firm and stable in shape.
Physics and Chemistry do provide clear cut explanation for the structure of solids. The firm and dense nature of solids is due to the strong intermolecular forces between their components. - On the basis of the arrangement of the components in a solid, there are two distinct types of solids - crystalline solids and amorphous solids. These two types differ in the arrangement of their respective components and so in their properties.
- Crystalline solids, also known as crystals have their own sub-classification. The types of crystals can be summarized as follows,
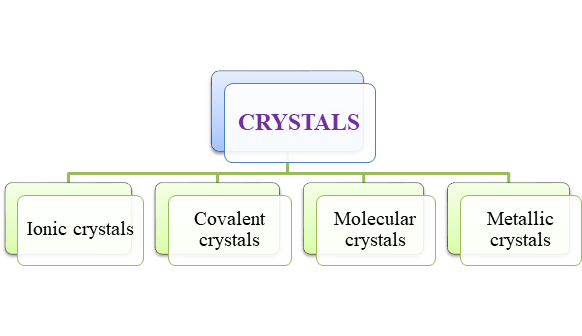 Figure 1
Figure 1 - Crystals have well-defined regular, compact, orderly arrangement of their components of very long range order. They are classified as – Ionic crystals, molecular crystals, covalent crystals and metallic crystals.
(b)
Interpretation Introduction
Interpretation:
The following types of crystals have to be discussed and examples have to be given for each type.
- (b) Ionic crystals (b) Covalent crystals (c) Molecular crystals (d) Metallic crystals.
Concept Introduction:
- A solid is anything that is firm and stable in shape. Physics and Chemistry do provide clear cut explanation for the structure of solids. The firm and dense nature of solids is due to the strong intermolecular forces between their components.
- On the basis of the arrangement of the components in a solid, there are two distinct types of solids - crystalline solids and amorphous solids. These two types differ in the arrangement of their respective components and so in their properties.
- Crystalline solids, also known as crystals have their own sub-classification. The types of crystals can be summarized as follows,
 Figure 1
Figure 1 - Crystals have well-defined regular, compact, orderly arrangement of their components of very long range order. They are classified as – Ionic crystals, molecular crystals, covalent crystals and metallic crystals.
(c)
Interpretation Introduction
Interpretation:
The following types of crystals have to be discussed and examples have to be given for each type.
- (c) Ionic crystals (b) Covalent crystals (c) Molecular crystals (d) Metallic crystals.
Concept Introduction:
- A solid is anything that is firm and stable in shape. Physics and Chemistry do provide clear cut explanation for the structure of solids. The firm and dense nature of solids is due to the strong intermolecular forces between their components.
- On the basis of the arrangement of the components in a solid, there are two distinct types of solids - crystalline solids and amorphous solids. These two types differ in the arrangement of their respective components and so in their properties.
- Crystalline solids, also known as crystals have their own sub-classification. The types of crystals can be summarized as follows,
 Figure 1
Figure 1 - Crystals have well-defined regular, compact, orderly arrangement of their components of very long range order. They are classified as – Ionic crystals, molecular crystals, covalent crystals and metallic crystals.
(d)
Interpretation Introduction
Interpretation:
The following types of crystals have to be discussed and examples have to be given for each type.
- (d) Ionic crystals (b) Covalent crystals (c) Molecular crystals (d) Metallic crystals.
Concept Introduction:
- A solid is anything that is firm and stable in shape. Physics and Chemistry do provide clear cut explanation for the structure of solids. The firm and dense nature of solids is due to the strong intermolecular forces between their components.
- On the basis of the arrangement of the components in a solid, there are two distinct types of solids - crystalline solids and amorphous solids. These two types differ in the arrangement of their respective components and so in their properties.
- Crystalline solids, also known as crystals have their own sub-classification. The types of crystals can be summarized as follows,
 Figure 1
Figure 1 - Crystals have well-defined regular, compact, orderly arrangement of their components of very long range order. They are classified as – Ionic crystals, molecular crystals, covalent crystals and metallic crystals.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
b) Certain cyclic compounds are known to be conformationally similar to carbohydrates, although they are not
themselves carbohydrates. One example is Compound C shown below, which could be imagined as adopting
four possible conformations. In reality, however, only one of these is particularly stable. Circle the conformation
you expect to be the most stable, and provide an explanation to justify your choice. For your explanation to be
both convincing and correct, it must contain not only words, but also "cartoon" orbital drawings contrasting the
four structures.
Compound C
Possible conformations (circle one):
Дет
Lab Data
The distance entered is out of the expected range.
Check your calculations and conversion factors.
Verify your distance. Will the gas cloud be closer to the cotton ball with HCI or NH3?
Did you report your data to the correct number of significant figures?
- X
Experimental Set-up
HCI-NH3
NH3-HCI
Longer Tube
Time elapsed (min)
5 (exact)
5 (exact)
Distance between cotton balls (cm)
24.30
24.40
Distance to cloud (cm)
9.70
14.16
Distance traveled by HCI (cm)
9.70
9.80
Distance traveled by NH3 (cm)
14.60
14.50
Diffusion rate of HCI (cm/hr)
116
118
Diffusion rate of NH3 (cm/hr)
175.2
175.2
How to measure distance and calculate rate
For the titration of a divalent metal ion (M2+) with EDTA, the stoichiometry of the reaction is typically:
1:1 (one mole of EDTA per mole of metal ion)
2:1 (two moles of EDTA per mole of metal ion)
1:2 (one mole of EDTA per two moles of metal ion)
None of the above
Chapter 12 Solutions
EBK GENERAL CHEMISTRY: THE ESSENTIAL CO
Ch. 12.2 - Prob. 1PECh. 12.2 - Prob. 2PECh. 12.2 - Prob. 1RCCh. 12.3 - Prob. 1RCCh. 12.4 - Prob. 1PECh. 12.4 - Prob. 1RCCh. 12.5 - Prob. 1PECh. 12.5 - Prob. 1RCCh. 12.6 - Prob. 1PECh. 12.6 - Prob. 1RC
Ch. 12.7 - Prob. 1RCCh. 12 - Prob. 12.1QPCh. 12 - Prob. 12.2QPCh. 12 - Prob. 12.3QPCh. 12 - Prob. 12.4QPCh. 12 - Prob. 12.5QPCh. 12 - Prob. 12.6QPCh. 12 - Prob. 12.7QPCh. 12 - Prob. 12.8QPCh. 12 - Prob. 12.9QPCh. 12 - Prob. 12.10QPCh. 12 - Prob. 12.11QPCh. 12 - Prob. 12.12QPCh. 12 - Prob. 12.13QPCh. 12 - 12.14 Diethyl ether has a boiling point of 34.5°C,...Ch. 12 - Prob. 12.15QPCh. 12 - Prob. 12.16QPCh. 12 - Prob. 12.17QPCh. 12 - 12.18 What kind of attractive forces must be...Ch. 12 - Prob. 12.19QPCh. 12 - Prob. 12.20QPCh. 12 - Prob. 12.21QPCh. 12 - Prob. 12.22QPCh. 12 - Prob. 12.23QPCh. 12 - Prob. 12.24QPCh. 12 - Prob. 12.25QPCh. 12 - Prob. 12.26QPCh. 12 - Prob. 12.27QPCh. 12 - Prob. 12.28QPCh. 12 - Prob. 12.29QPCh. 12 - Prob. 12.30QPCh. 12 - Prob. 12.31QPCh. 12 - Prob. 12.32QPCh. 12 - Prob. 12.33QPCh. 12 - Prob. 12.34QPCh. 12 - Prob. 12.35QPCh. 12 - 12.36 A solid is hard, brittle, and electrically...Ch. 12 - Prob. 12.37QPCh. 12 - Prob. 12.38QPCh. 12 - Prob. 12.39QPCh. 12 - Prob. 12.40QPCh. 12 - Prob. 12.41QPCh. 12 - Prob. 12.42QPCh. 12 - Prob. 12.43QPCh. 12 - Prob. 12.44QPCh. 12 - Prob. 12.45QPCh. 12 - Prob. 12.46QPCh. 12 - Prob. 12.47QPCh. 12 - Prob. 12.48QPCh. 12 - Prob. 12.49QPCh. 12 - Prob. 12.50QPCh. 12 - Prob. 12.51QPCh. 12 - Prob. 12.52QPCh. 12 - Prob. 12.53QPCh. 12 - Prob. 12.54QPCh. 12 - Prob. 12.55QPCh. 12 - Prob. 12.56QPCh. 12 - Prob. 12.57QPCh. 12 - Prob. 12.58QPCh. 12 - Prob. 12.59QPCh. 12 - Prob. 12.60QPCh. 12 - Prob. 12.61QPCh. 12 - Prob. 12.62QPCh. 12 - 12.63 What is the relationship between...Ch. 12 - Prob. 12.64QPCh. 12 - 12.65 Why is solid carbon dioxide called dry ice?
Ch. 12 - Prob. 12.66QPCh. 12 - 12.67 Referring to Figure 12.28, estimate the...Ch. 12 - Prob. 12.68QPCh. 12 - Prob. 12.69QPCh. 12 - Prob. 12.70QPCh. 12 - Prob. 12.71QPCh. 12 - Prob. 12.72QPCh. 12 - Prob. 12.73QPCh. 12 - Prob. 12.74QPCh. 12 - 12.75 These compounds are liquid at −10°C; their...Ch. 12 - 12.76 Freeze-dried coffee is prepared by freezing...Ch. 12 - Prob. 12.77QPCh. 12 - 12.78 Steam at 100°C causes more serious burns...Ch. 12 - 12.79 Vapor pressure measurements at several...Ch. 12 - Prob. 12.80QPCh. 12 - Prob. 12.81QPCh. 12 - Prob. 12.82QPCh. 12 - Prob. 12.83QPCh. 12 - Prob. 12.84QPCh. 12 - Prob. 12.85QPCh. 12 - Prob. 12.86QPCh. 12 - Prob. 12.87QPCh. 12 - Prob. 12.88QPCh. 12 - Prob. 12.89QPCh. 12 - 12.90 Name the kinds of attractive forces that...Ch. 12 - Prob. 12.91QPCh. 12 - Prob. 12.92QPCh. 12 - Prob. 12.93QPCh. 12 - Prob. 12.94QPCh. 12 - Prob. 12.95QPCh. 12 - Prob. 12.96QPCh. 12 - Prob. 12.97QPCh. 12 - Prob. 12.98QPCh. 12 - 12.99 The liquid-vapor boundary line in the phase...Ch. 12 - Prob. 12.100QPCh. 12 - Prob. 12.101QPCh. 12 - Prob. 12.102QPCh. 12 - Prob. 12.103QPCh. 12 - Prob. 12.104QPCh. 12 - Prob. 12.105QPCh. 12 - Prob. 12.106QPCh. 12 - 12.107 The following graph shows approximate plots...Ch. 12 - Prob. 12.108QPCh. 12 - Prob. 12.109QPCh. 12 - Prob. 12.110QPCh. 12 - Prob. 12.111QPCh. 12 - Prob. 12.112QPCh. 12 - Prob. 12.113QPCh. 12 - Prob. 12.114QPCh. 12 - 12.115 Use the concept of intermolecular forces to...Ch. 12 - Prob. 12.116QPCh. 12 - 12.117 What is the origin of dark spots on the...Ch. 12 - Prob. 12.118QPCh. 12 - 12.119 The electrical conductance of copper metal...Ch. 12 - Prob. 12.120SPCh. 12 - Prob. 12.121SPCh. 12 - Prob. 12.122SPCh. 12 - Prob. 12.123SPCh. 12 - Prob. 12.124SPCh. 12 - 12.125 The boiling point of methanol is 65.0°C and...Ch. 12 - Prob. 12.126SPCh. 12 - Prob. 12.127SPCh. 12 - Prob. 12.128SPCh. 12 - Prob. 12.129SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please help me solve this reaction.arrow_forwardIndicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forwardSynthesize 2-Ethyl-3-methyloxirane from dimethyl(propyl)sulfonium iodide using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- Synthesize 2-Hydroxy-2-phenylacetonitrile from phenylmethanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardSynthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forward
- If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.arrow_forwardSynthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forward
- We mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forwardIndicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=HCWwRh5CXYU;License: Standard YouTube License, CC-BY