
Without referring to your textbook or a periodic table, write the full electron configuration, the orbital box diagram, and the noble gas shorthand configuration for the elements with the following
msp;
(a)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 1.
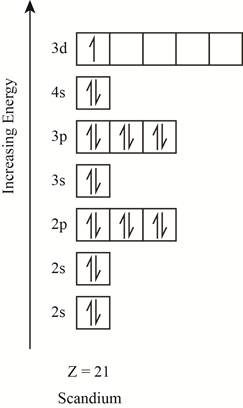
Figure 1
The electronic configuration of the given element can also be written as
(b)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 2.
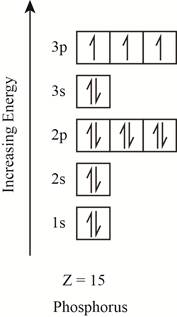
Figure 2
The electronic configuration of the given element can also be written as
(c)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 3.
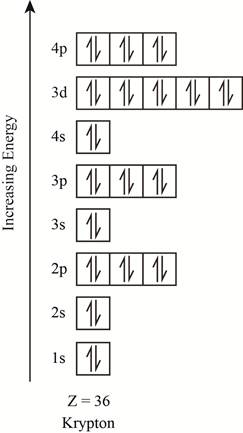
Figure 3
The electronic configuration of the given element can also be written as
(d)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 4.
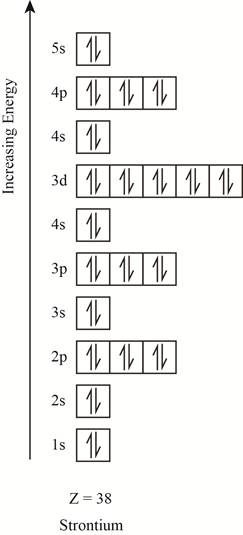
Figure 4
The electronic configuration of the given element can also be written as
(e)
Interpretation:
The electronic configuration of the given element, the orbital box diagram and the noble gas shorthand configuration for the elements are to be stated.
Concept Introduction:
The distribution of the electrons that exists in the atomic orbital of an atom is collectively known as electronic configuration. The description of every electron in an orbital is given by the electronic configuration of that atom.
Answer to Problem 97AP
The electronic configuration of the given element with
Explanation of Solution
The electronic configuration of the given element with
The orbitals in the orbital box diagram are arranged in increasing order of energy shells. The orbital box diagram is shown in figure 5.
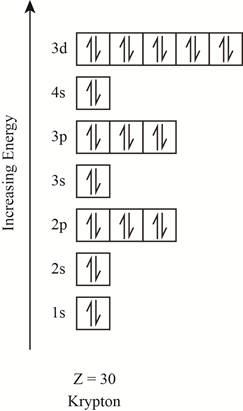
Figure 5
The electronic configuration of the given element can also be written as
Want to see more full solutions like this?
Chapter 11 Solutions
EBK INTRO.CHEMISTRY (NASTA EDITION)
- Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!arrow_forwardCan you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!arrow_forwardCan you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!arrow_forward
- Please answer the questions in the photos and please revise any wrong answers. Thank youarrow_forward(Please be sure that 7 carbons are available in the structure )Based on the 1H NMR, 13C NMR, DEPT 135 NMR and DEPT 90 NMR, provide a reasoning step and arrive at the final structure of an unknown organic compound containing 7 carbons. Dept 135 shows peak to be positive at 128.62 and 13.63 Dept 135 shows peak to be negative at 130.28, 64.32, 30.62 and 19.10.arrow_forward-lease help me answer the questions in the photo.arrow_forward
- For the reaction below, the concentrations at equilibrium are [SO₂] = 0.50 M, [0] = 0.45 M, and [SO3] = 1.7 M. What is the value of the equilibrium constant, K? 2SO2(g) + O2(g) 2SO3(g) Report your answer using two significant figures. Provide your answer below:arrow_forwardI need help with this question. Step by step solution, please!arrow_forwardZn(OH)2(s) Zn(OH)+ Ksp = 3 X 10-16 B₁ = 1 x 104 Zn(OH)2(aq) B₂ = 2 x 1010 Zn(OH)3 ẞ3-8 x 1013 Zn(OH) B4-3 x 1015arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





